
Concept explainers
(a)
Interpretation:
A chair conformation of the given molecule with all
Concept introduction:
When the cyclohexane ring is oriented as a chair, all the axial bonds are alternating up and down around the ring and are perfectly vertical. All the equatorial bonds are also alternating around the ring, but are slightly up or slightly down. The structure of cyclohexane in the chair form is shown as:
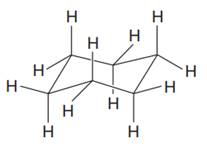
(b)
Interpretation:
A chair conformation of the given molecule with all
Concept introduction:
When the cyclohexane ring is oriented as a chair, all the axial bonds are alternating up and down around the ring and are perfectly vertical. All the equatorial bonds are also alternating around the ring, but are slightly up or slightly down. The structure of cyclohexane in the chair form is shown as:

Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
- myo-Inositol, one of the isomers of 1,2,3,4,5,6-hexahydroxycyclohexane, acts as a growth factor in both animals and microorganisms. Draw the most stable chair conformation of myo-inositol.arrow_forwardFollowing is a planar hexagon representation for one isomer of 1,2,4-trimethylcyclohexane. Draw the alternative chair conformations of this compound and state which of the two is more stable.arrow_forwardFill in the blanks: cis-1,3-Dimethylcyclohexane has two different chair conformations: one withboth methyl groups in __________ positions and one with both methyl groups in ____________ positions.arrow_forward
- Following is a structural formula for cortisol (hydrocortisone). Draw a stereo-representation of this molecule showing the conformations of the five- and six-membered rings.arrow_forwardDraw a Newman projection for two more staggered conformations of this molecule. Which of your conformations is most stable? Assume that -OH and -CH3 are comparable in size.arrow_forwardDraw both chair conformations of cis-1,2-dimethylcyclohexane, and determine which trans isomer is more stable.arrow_forward
- Draw both chair conformers for the following molecule and circle the most stable conformation.arrow_forwardDraw the two possible chair conformations for the below molecule. Clearly indicate whether the substituents are axial (a) or equatorial (e).arrow_forwardwhat is the sawhorse projection of the LEAST STABLE ECLIPSED CONFORMATION OF N-PENTANE ROTATING BETWEEN C2-C3?arrow_forward
- draw the two chair conformers for the following structures. If one is lower energy, circle the one of each pair .arrow_forwardWhat are the two chair conformers for this molecule and which one is more favorable?arrow_forwardHow can you draw a ring flip if given the 2D drawing of a chair conformation?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


