
Concept explainers
(a)
Interpretation:
The given expanded structural formula of alkane has to be converted into condensed structural formula where parentheses and subscript is used to denote the number of
Concept Introduction:
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
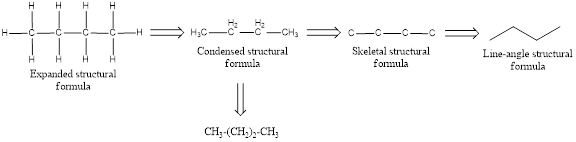
In condensed structural formula for
(b)
Interpretation:
The given expanded structural formula of alkane has to be converted into condensed structural formula where parentheses and subscript is used to denote the number of
Concept Introduction:
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.

In condensed structural formula for alkanes, the repeating
Trending nowThis is a popular solution!

Chapter 12 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Sketch the titration curve for the amino acid alanine with pka1 = 2.34 and pka2 = 9.69.arrow_forwardHemoglobin is considered to be a tetrameric complex with a 64 kDa (α β)2. When attempting to purify hemoglobin, we must first purify the α and β monomers (about 16 kDa each) to prepare the tetramer. This is formed from the dimer intermediate: 2 α + 2 β -> 2 αβ -> (α β)2. 1. The graph given represents a size-exclusion chromatogram after the refolding of the hemoglobin tetramer. This process is not 100% efficient, so we may have leftovers of dimers and monomers. In the graph given, can we label the peaks given as a tetramer, dimer, or monomer?arrow_forwardFollowing are Fischer projection for a group of five carbon sugars,all of which are aldopentoses. Identify the pairs that are enantiomers and the pairs that are epimers.(The sugar shown herebare not all of the possible five carbon sugars.)arrow_forward
- LEFT:You have the following titration curve of an amino acid with a non-polar R-group with arrows on curve from (1) to (5). RIGHT: This amino acid has the following structures (a) to (e)) at different points of the titration curve. Match each numbered arrow on the curve (from (1) to (5)) with the appropriate amino acid structure (from (a) to (e)) 12 (5) H.NCHCOH + HNCHCO (a) 10 (4) CH3 CH Isoelectrin HŃCHCO + (b) ppint 43) HNCHCO CH3 CH3 (2) HaNCHCOH H. NCHCO (e) (d CH3 CH3 0.0 0.5 1.0 15 20 NaOH equivalents. H-NCHCO (e) CH3arrow_forwardFollowing are structural formulas for cytosine and thymine. Draw two additional tautomeric forms for cytosine and three additional tautomeric forms for thymine.arrow_forwardVitamin D3, the most abundant of the D vitamins, is synthesized from 7- dehydrocholesterol, a compound found in milk and fatty fish such as salmon and mackerel. When the skin is exposed to sunlight, a photochemical electrocyclic ring opening forms provitamin D3, which is then converted to vitamin D3 by a sigmatropic rearrangement. Draw the structure of provitamin D3.arrow_forward
- Dihedral angles of Phe165 of 4KW4 betasheet values are consistent with what is expected from the Ramachandran Plot?arrow_forwardFollowing are Fischer projections for a group of five-carbon sugars, all of which are aldopentoses. Identify the pairs that are enantiomers and the pairs that are epimers. (The sugars shown here are not all of the possible five-carbon sugars.) 1 СНО 2 СНО СНО Н-С—ОН Н-С—ОН Н—С—ОН Н-С—ОН НО —С—Н Н-С—ОН Н- С—ОН НО—С— Н НО—С—Н CH̟OH CH̟OH ČH̟OH в iоchemistry |61 STATE ATION Republic of the Philippines Romblon State University Romblan, Philippines 4 СНО 5 СНО 6 СНО НО—С—Н Н-С—ОН НО -С—Н Н—С—ОН НО -С—Н НО -С—Н H-C–OH Н-С—ОН НО—С—Н CH,OH ČH,OH CH,OHarrow_forwardHemoglobin is considered to be a tetrameric complex with a 64 kDa (α β)2. When attempting to purify hemoglobin, we must first purify the α and β monomers (about 16 kDa each) to prepare the tetramer. This is formed from the dimer intermediate: 2 α + 2 β -> 2 αβ -> (α β)2. The graph given represents a size-exclusion chromatogram after the refolding of the hemoglobin tetramer Using the size-exclusion chromatogram given, 1. Draw an SDS-Page Gel with a reducing agent such as BME using the three peaks listed on the graph.arrow_forward
- Oxytocin, a hormone peptide of nine amino acids, is widely used in obstetrics to induce uterine contractions. There is an intramolecular disulfide bond which must be reduced before sequencing. Reduced oxytocin has the composition Asn 2Cys Gln Gly Ile Leu Pro Tyr . Partial hydrolysis of reduced oxytocin led to the following fragments: (a) AsnCys (b) CysTyr (c) TyrIleGln (d) CysProLeu (e) IleGln (f) LeuGly (g) GlnAsnCys . Reaction of reduced oxytocin with carboxypeptidase showed glycine as the first liberated amino acid. What is the amino acid sequence of oxytocin?arrow_forwardWrite a structural formula for a tetrapeptide composed of phenyl-alanine, glycine, aspartic acid, and histidine in which the phenylalanine is N-terminal and the histidine is C-terminal. How many different tetrapeptides could be written that fit the criteria in this question?arrow_forwardImagine a trisaccharide that has D-Altrose, D-Gulose, and D-Ribose. D-Altrose is bonded to D-Gulose in an α(1→4) glycosidic bond and D-Ribose is bonded to D-Altrose in an α(1→6) glycosidic bond. Draw the imaginary trisaccharide using Haworth projection with the appropriate representation of each monosaccharide.arrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
