
Bread is typically made by ?rst dissolving preserved yeast (a microscopic biological organism that consumes sugars and emits CO2 as a waste product) in water, then adding other ingredients, including ?our, sugar, fat (usually butter or shortening), and salt. After the ingredients are combined, the dough is "kneaded," or mixed to promote the formation of a protein network from two proteins (gliadin and glutenin)15 present in wheat ?our. This network is what strengthens the dough and allows it to stretch elastically without breaking. The dough is then allowed to rise in a process called "proo?ng," in which the yeast consumes sugar and releases CO2, which in?ates air pockets in the dough that are subsequently ?lled with air. Finally the dough is baked; the gas pockets expand due to the temperature rise and evaporation of water, the starches from the ?our are dehydrated (dried), and the yeast dies.
A good French bread has an open, porous structure. The pores must be stabilized by the protein network until the bread is dried suf?ciently to hold its shape. The bread collapses if the protein network fails prematurely.
(a) Rouille et al.16 investigated the in?uence of ingredients and mixing conditions on the quality of frozen French bread dough. Each loaf was initially formed roughly as a cylinder with a mass of 150 g (including essentially no CO2), a diameter of 2.0 cm. and a length of 25.0cm. Determine the speci?c volume of a bread dough proofed for two hours at 28°C from which 1.20 cm3 gas/min per 100 g dough evolves as bubbles within the dough. State your assumptions.
(b) During proo?ng, the increases in volume of a series of control loaves were monitored along with the mass of CO2 evolved. Rupture of the protein network during proo?ng can be detected when the volume of the dough no longer increases at the same rate as the production of CO2 from the yeast. Data from one of these experiments are shown in the table below. Plot the speci?c volumes of CO2 (per 100g dough) and dough as a function of time. If the preferred proo?ng time is such that the dough achieves 70% of its total volume before collapse, specify the proper proo?ng time for this formula.
(c) The referenced study found that the parameter with the most signi?cant in?uence on dough quality was mixing time, with an extended mixing time producing a stronger protein network. Why might extended mixing times not be desirable in commercial production of bread?
(d) Suggest causes for the following undesirable bread-baking outcomes: (i) a ?at, dense loaf; (ii) an overly large loaf.
(e) Suggest why the period during which the dough rises is called "proo?ng." Remember that yeast is a biological organism.
| t(min) | 0 | 20 | 40 | 60 | 80 | 100 | 120 | 140 | 160 | 180 | 200 | 220 | 240 |
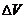 cm3 dough) cm3 dough) |
0 | 0 | 20 | 60 | 80 | 115 | 155 | 198 | 247 | 305 | 322 | 334 | 336 |
| Gas evolved (g CO2) | 0.0 | 37.2 | 63.2 | 68.8 | 126.3 | 192.7 | 234.8 | 315.8 | 385.4 | 515.0 | 578.1 | 657.5 | 745.0 |
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Data Mining for Business Analytics: Concepts, Techniques, and Applications with XLMiner
Starting Out With Visual Basic (7th Edition)
Management Information Systems: Managing the Digital Firm (15th Edition)
Heating Ventilating and Air Conditioning: Analysis and Design
- The first step of the metabolic process known as glycolysis is the conversion of glucose to glucose- 6-phosphate. This process has a positive value for rG' Glucose + Pi Glucose-6-phosphate + H2O rG' = +13.8 kJ/mol-rxn This reaction is coupled to the hydrolysis of ATP ATP + H2O ADP + Pi rG' = -30.5 kJ/mol-rxn What is the sum of these two equations and the value of rG' for the coupled reaction? Is the coupled reaction product-favored at equilibrium?arrow_forward(Chemical Connections 30A) What is a recommendation from ChooseMyPlate.gov regarding dairy products?arrow_forwardWhy are enzymes that are used for laboratory or clinical work stored in refrigerators?arrow_forward
- Some researchers feel that the high experienced by the runners may be due to the brains synthesis of petides. Explain why this theory might be a reasonable explanation.arrow_forwardWhy are so many different enzymes needed?arrow_forwardExplain why monitoring blood lactate levels might be a useful technique to gauge the amount of conditioning in an Olympic runner.arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning





