
Fermentation is a biochemical process in which a carbohydrate—such as the sugar glucose—is converted by bacteria or yeast to an organic compound—such as ethanol—and carbon dioxide. The process is best known for its use in making beer_ wine, and other alcoholic beverages, but it is also used to produce a wide range of other industrially important chemicals.
The diagram below shows a continuous fermentation process. An aqueous solid-liquid slurry containing a carbohydrate and bacteria enters a stirred tank where it undergoes fermentation. Fermentation products, including species condensed from the off-gas, and bacteria are withdrawn from the tank. A mixer with multiple rotating impellers keeps the solids in the tank suspended in the liquid. An air stream enters the fermenter through a sparger (something like an inverted shower head) designed to produce small bubbles that rise through the mixture and remove the CO2 formed in the reaction. Some of the oxygen in the air fed to the fermenter is consumed by the bacteria. The gas products go through a condenser, which condenses most of the water and returns it to the fermenter, and the uncondensed off-gas goes to a dryer and then to a mass spectrometer, where its composition is determined. The ?ow rate of the air feed is measured to be 0.600 SLPM (liters per minute at standard temperature and pressure) and the measured compositions of the air and the off- gas are shown on the diagram. The off-gas is at 28°C and 1.1 atm.
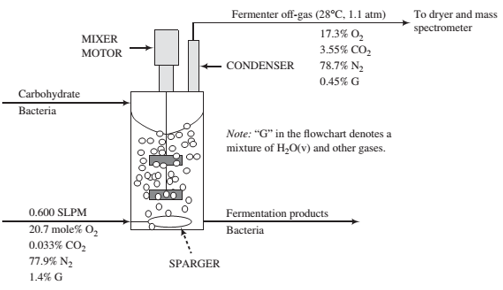
(a) Describe in your own words the purpose of this process and the functions of the mixer, the air, and the sparger.
(b) In a small laboratory fermenter, the mass of the broth in the tank is 0.58 kg, and the broth density is 1.05 kg/L. Calculate the carbon dioxide evolution rate (CER) and the oxygen uptake rate (OUR) in mmol/(h ( L), where the volume unit in the denominator refers to the volume of broth in the tank. Also, calculate the off-gas volumetric ?ow rate in L/min.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Java How To Program (Early Objects)
C++ How to Program (10th Edition)
Using MIS (10th Edition)
Starting Out with C++: Early Objects (9th Edition)
- Please show calculations for the blank spaces from the image.arrow_forwardPart B The solvent for an organic reaction is prepared by mixing 70.0 mL of acetone (C3H6O) with 64.0 mL of ethyl acetate (C4H8O2). This mixture is stored at 25.0 °C. The vapor pressure and the densities for the two pure components at 25.0 °C are given in the following table. What is the vapor pressure of the stored mixture? Vapor pressure Density Compound (mmHg) (g/mL) 230.0 0.791 95.38 0.900 acetone ethyl acetate Express your answer to three significant figures and include the appropriate units. ▸ View Available Hint(s) Psoln: HÅ Units Value ?arrow_forward[1b] How much of the concentrated acid is needed to prepare the diluted solution?arrow_forward
- Pre-lab Assignment 1.) Note from the 'Introduction' that the freezing-point-depression constant, Kfp, for any solvent can be calculated as long as the heat of fusion, AHfus, and freezing point of the solvent is known. Calculate the freezing-point-depression constant (in SI units) for carbon tetrachloride, CC14, given that its heat of fusion is 2.5 kJ/mol and its freezing point is 250.3 K. M solvent & (TF Solvet) AIFUD - molor Mod 1=0,0821 LATM/ molk ~14=153.82glangs Кы = latm (153.82 $7) (0.0821 (ar) (250.5x)² 2 2.5 123/M/G\arrow_forwardExplain the importance of using a thermometer in the simple distillation setup. Enumerate the characteristics of a substance for it to be separated from the heterogeneous mixture by steam distillation. How does steam facilitate the distillation of essential oils? Discuss some applications of simple distillation and steam distillation. Explain why you can use each method for the chosen application. What is the most dominant intermolecular forces of attraction that exist between: a. water and limonene, the major monoterpene component of lemon essential oil? b. water and constituent ions of sodium chloride? 6. Based on your answers in number 5, which pair (water-limonene or water-sodium chloride) exhibits a stronger attraction between their molecules/ions? 7. Describe what happens during the salting-out process in terms of the intermolecular forces of attraction involved as mentioned in your previous answers.arrow_forward1)The production of authentic tequila is restricted to the blue agave and a defined geographical area in west-central Mexico. Two basic categories of tequila can be distinguished: “100% agave” and “mixed” tequila. High-quality “100% agave” tequila uses pure agave juice for fermentation and Mexican law states that these authentic tequilas must be bottled in Mexico. A mixed tequila is manufactured by adding up to 49% (w/v) of sugar from sugar cane for fermentation. The low-quality tequila is usually shipped in bulk containers for diluting and bottling in the importing countries. Due to frequent fraud in the labeled quality of tequila, there is a need to verify the authenticity of a tequila using chemical analysis. Figure 1 shows an ion chromatogram for an authentic 100% agave tequila and a commercial mixed tequila. The difference in anion content is due to the different sources of water used in final dilution and bottling steps. a) In reversed-phase liquid chromatography, a…arrow_forward
- Heat of Reaction, Mole Fraction 1. A group of students perform a series of experiments mixing HCI with an unknown base of the formula M(OH)n. For each trial the total volume of acid and base was 50.0 mL. The concentration of the acid and base solutions were 0.6000 M. The data is plotted in the graph provided. The density of the aqueous solution is assumed to be 1.00 g/mL and the specific heat of water is 4.18 J/g °C in joules 1600 1400 1200 1000 800 600 400 200 0 0.0000 0.6000 M HCI 0 2000 0.6000 M(OH)x 0 4000 0 6000 Mole Fraction of Acid 0 8000 10000 a) What is the limiting reagent at a mole fraction of 0.6 of acid? b) Write a balanced equation for the reaction of M(OH), with HCI. (You must determine "n") c) For one trial, the students measured a temperature change of + 2.9 °C. Calculate q. d) The students calculated a value of q of 1.5 x 10³ J for one of their trials. For this trial 0.015 mol of HCI and 0.015 mol of M(OH), were used. Calculate heat per mole for this reaction.arrow_forwardIf you analyze the solubility curve and determine how much sugar (in grams) can be added to 100 grams of water at 30°C to form a saturated solution.arrow_forwardDraw a temperature-composition diagram for the acetone-chloroform mixture, wherein the boiling points of acetone and chloroform are 329 K and 334 K, respectively. They supposedly form a high boiling azeotropic mixture at Xacetone = 0.34 with boiling point at 430 K.arrow_forward
- General Chemistry ----------------------- (a) A 10.0 percent solution of sucrose (C12H22O11) in water has a density of 1.038 gmL-1. Express the concentration of the sugar as (1) Molality; (2) Molarity; (3) Mole fractionarrow_forwardBased on the following data, determine the molecular weight of the unknown solute. Note the "initial freezing point" refers to the freezing point of the solvent as a pure substance while the "final freezing point" refers to the freezing point of the mixture. FPD Constant of Final Freezing Mass Solvent: Mass Solute Initial Freezing Point: Solvent: Point: 15.000 g 2.350 g 85.3°C 4.9 82.4°C Your answer should follow the format: a) ΔΤ b) Molality of the Solution c) mols of solute d) Molecular weight of Solutearrow_forwardcan you help with sub questions A, B and Carrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





