
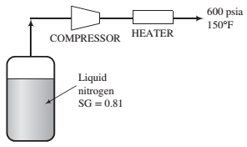
Approximately 150 SCFM (standard cubic feet per minute) of nitrogen is required by a process facility. As shown in the diagram below, plans call for supplying the facility from a tank of liquid nitrogen (SG = 0.81) at its normal boiling point (−350°F) and 1 atm. Nitrogen vapor leaves the tank and is compressed and heated to obtain the desired conditions. 150°F and 600 psia.
(a) Using the generalized compressibility charts, determine the volumetric flow rate of nitrogen delivered from the heater.
(b) What would the required minimum tank size be if deliveries are made to the site no more frequently than every two weeks?
Trending nowThis is a popular solution!

Chapter 5 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Database Concepts (7th Edition)
Starting Out with Java: From Control Structures through Data Structures (3rd Edition)
Experiencing MIS
Computer Systems: A Programmer's Perspective (3rd Edition)
- A steel cylinder containing compressed air is stored in a fermentation laboratory ready to provide aeration gas to a small-scale bioreactor. The capacity of the cylinder is 48 litres, the absolute pressure is 0.35 MPa and the temperature is 22°C. One day in mid-summer when the air conditioning breaks down, the temperature in the laboratory rises to 33°C and the valve at the top of the cylinder is accidentally left open. Estimate the proportion of air that will be lost. What assumptions will you make? 2 24 C.arrow_forwardPressure (A) Explain the term pressure and state its S.I. unit. (B) Explain Henry’s law. (C)A bottle of H2 has just been received by the technicians in DkIT for use in the instrumentation lab. It is a 47 litre cylinder at a pressure of 50 atmospheres. The normal working pressures is 2 bar. (i) To what volume of gas will that equate at the working pressure? (ii) For how many hours will the gas last if it used at the rate of 0.5dm3 per hour? D)You see your best friend at the bar and you walk up behind her. You accidentally startle her and she takes a step backwards. Unfortunately, she is wearing high heels and her heels come down on your foot. She weighs only 55kg but the size of her heel is 6mm by 6mm.Determine the pressure that she applies on your foot. (E) If a diver dives to a depth of 35 m what will be: (i) the pressure in Pascals due the water column? (3 marks) (ii) the pressure of the air in…arrow_forwardProducer gas is a fuel that is manufactured from the reaction of coal, steam and air. A producer gas has the following composition by volume (dry basis): 20.35% CO, 11.03%, CO2, 15% H2, 51.43% N2, 1.98%O2 and 0.21 % CH4. The gas is flowing at 6000 cfm. It has a temperature of 200℃ , a pressure of 21 cm Hg and a partial pressure of 50 mm Hg for H2O. The atmospheric pressure is 758 mm Hg. The gas is delivered from the source to the furnace by means of a pipe. It cools down to 50 ℃ and is compressed to 2 atm pressure. At this condition, calculate:1. the volumetric flow ate2. the mass flow rate3. the partial pressure of each component/including water)4. complete analysis of the mixturearrow_forward
- A solid fuel, described by the chemical structure below, is combusted in 10% excess air. If necessary, clearly state any assumptions required to develop your solution. H Н —с — N — с — s — н | || 0 = C S H (a) Determine the mass feed rate (g/min) of the fuel such that 10,000 m³/min of total flue gas is generated at 1500 °C and 1 atm. (b) Determine the effluent SO2 concentration (ppm,) at STP and dry conditions.arrow_forwardThe combustion gases from an industrial furnace using a hydrocarbon fuel and dry air enter the stack gas at normal barometric pressure and 3750F and have the following Orsat analysis: 12.2% CO2, 3.1% O2, 1.2% CO, and 83.5% N2. Determine the following: (a) The percent excess air (b) The volume of gases entering the stack, expressed as cubic feet per pound of carbon burnt in the furnace (c) The dew point of the stack gas (d) The atomic ratio of hydrogen to carbon in the fuel (e) The mol of stack gas per atom of carbon burntarrow_forwardP1A.6 The molar mass of a newly synthesized fluorocarbon was measured in a gas microbalance. is device consists of a glass bulb forming one end of a beam, the whole surrounded by a closed container. The beam is pivoted, and the balance point is attained by raising the pressure of gas in the container, so increasing the buoyancy of the enclosed bulb. In one experiment, the balance point was reached when the fluorocarbon pressure was 327.10Torr; for the same setting of the pivot, a balance was reached when CHF3 (M = 70.014 g mol−1) was introduced at 423.22 Torr. A repeat of the experiment with a di erent setting of the pivot required a pressure of 293.22 Torr of the uorocarbon and 427.22 Torr of the CHF3. What is the molar mass of the fluorocarbon? Suggest a molecular formula.arrow_forward
- Q.2: Assume the following gas mixture behaves like an ideal gas. Calculate the mass and specific gravity of the gas mixture which is contained in a 1.5 ft³ container at 100 °F and 200 psia. Component: Composition(mole fraction) Methane (C₁) 0.80 Ethane (C₂) 0.10 Propane (C3) 0.10arrow_forwardP1A.6 The molar mass of a newly synthesized fluorocarbon was measured in a gas microbalance. is device consists of a glass bulb forming one end of a beam, the whole surrounded by a closed container. e beam is pivoted, and the balance point is attained by raising the pressure of gas in the container, so increasing the buoyancy of the enclosed bulb. In one experiment, the balance point was reached when the fluorocarbon pressure was 327.10Torr; for the same setting of the pivot, a balance was reached when CHF3 (M = 70.014 g mol−1) was introduced at 423.22 Torr. A repeat of the experiment with a di erent setting of the pivot required a pressure of 293.22 Torr of the uorocarbon and 427.22 Torr of the CHF3. What is the molar mass of the fluorocarbon? Suggest a molecular formula.arrow_forward1. 0.45 kg of carbon monoxide (28) and 1 kg of air at 15°C are contained in a vessel of volume 0.4 m'. Calculate the partial pressure of each constituent and the total pressure in the vessel. The gravimetric analysis of air is to be taken as 23.3% oxygen (32) and 76.7% nitrogen (28). IAns. Po, = 0.4359 bar; PN, = 1.64 bar, Poo = 0.962 bar]arrow_forward
- 20. A mixture consisting of 6 kg of O, and 9 kg of N, has a pressure of 3 bar and temperature of 20°C. For the mixture determine the following : (i) The mole fraction of each component ; (iii) The specific gas constant; (v) The partial pressures and partial volumes. (ii) The average molecular weight ; (iv) The volume and density; [Ans. (i) 0.3684, 0.6315 ; (iüi) 29.475 ; (iii) 0.282 kJ/kg K ; (iv) 4.13 m?, 3.629 kg/m² ; (v) 1.1 bar, 1.894 bar ; 1.52 m², 2.61 m²)arrow_forward3. Given: Soda and lime are added to a glass batch in the form of soda ash (Na,CO,) and limestone (CaCO). During heating, these two ingredients decompose to give off carbon dioxide (CO₂), the resulting products being soda and lime. Find: Compute the weight of soda ash and limestone that must be added to 110 lb of quartz (SiO₂) to yield a glass of composition 72 wt% SiO₂, 16 wt% Na₂O, and 12 wt% Cao. Solution:arrow_forwardA 500 m2 botanical garden which harvest 1000 flowers per day of 15 variations, in a year the average rainfall is 170 cm/yr. With the rainfall 30% percolates into the ground and the rest are harvested in a spherical cistern. 35% of the cistern water is used for watering that percolates into the ground and the rest of water evaporate. Every day they sprinkled water from the cistern and dug wells. Of the dug well water, 75% evaporates and the remainder percolates back into the ground. (Steady State Condition) What is volume of rainfall in the botanical garden? Volume of water in the cistern Volume of water from the well Volume of water that evapotranspiration from the well Total Volume of water that repercolates into the groundarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





