
Chemistry & Chemical Reactivity
10th Edition
ISBN: 9781337399074
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 118SCQ
The composition diagram, or alpha plot, for the important acid-base system of carbonic acid, H2CO3, is illustrated. (See Study Question 1.7 for more information on such diagrams.)
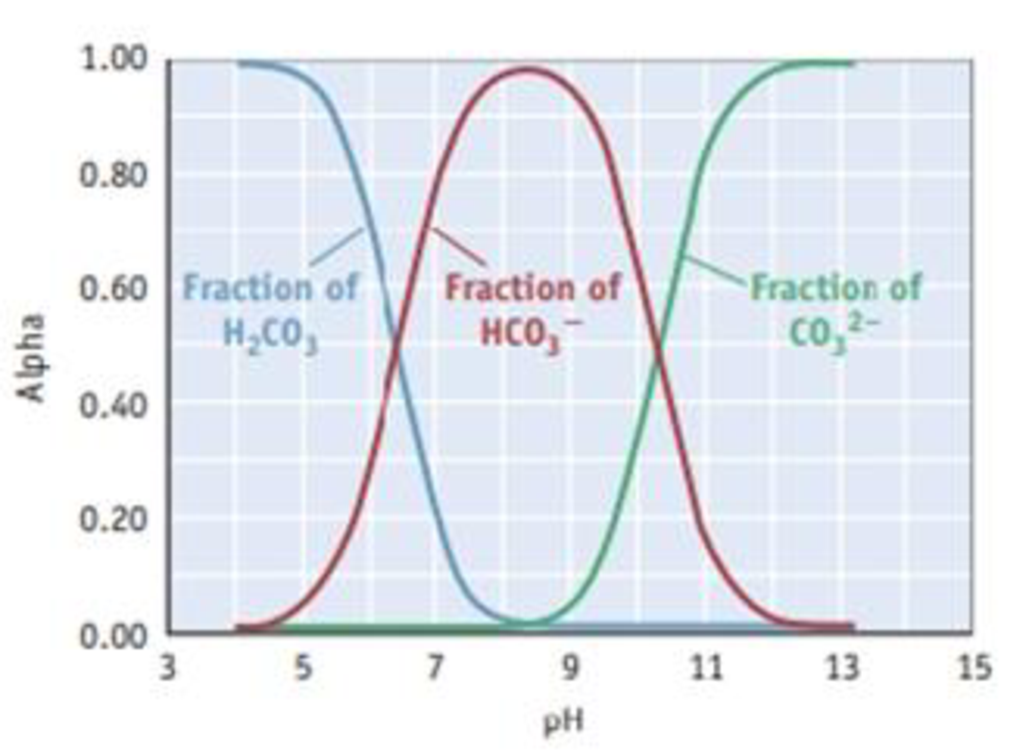
- (a) Explain why the fraction of bicarbonate ion, HCO3−, rises and then falls as the pH increases.
- (b) What is the composition of the solution when the pH is 6.0? When the pH is 10.0?
- (c) If you wanted to buffer a solution at a pH of 11.0, what should be the ratio of HCO3− to CO32−?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Suppose that, instead of using NaOH, a base such as Ba(OH)2 had been used. What changes in the calculations would then have to be made to determine the molar concentrations of the base?
Write the net-ionic reaction that occurs between Na2HPO4 and NaOH (an acid-base reaction)
Write the balanced molecular equation for the neutralization reaction between HIHI and Ba(OH)2Ba(OH)2 in aqueous solution. Include physical states.
Chapter 17 Solutions
Chemistry & Chemical Reactivity
Ch. 17.1 - You have a 0.30 M solution of formic acid (HCO2H)...Ch. 17.2 - What is the pH of a buffer solution composed of...Ch. 17.2 - Use the Henderson-Hasselbalch equation to...Ch. 17.2 - Using an acetic acid/sodium acetate buffer...Ch. 17.2 - Calculate the pH of 0.500 L of a buffer solution...Ch. 17.3 - The titration of 0.100 M acetic acid with 0.100 M...Ch. 17.3 - Calculate the pH after 75.0 mL of 0.100 M HO has...Ch. 17.4 - The barium ion concentration, [Ba2+], in a...Ch. 17.4 - Calculate the solubility of AgCN in moles per...Ch. 17.4 - Calculate the solubility of Ca(OH)2 in moles per...
Ch. 17.4 - Calculate the solubility of BaSO4 (a) in pure...Ch. 17.5 - Solid Pbl2 (Ksp = 9.8 109) is placed in a beaker...Ch. 17.5 - Prob. 17.13CYUCh. 17.5 - Prob. 17.14CYUCh. 17.6 - Silver nitrate (0.0050 mol) is added to 1.00 L of...Ch. 17.6 - Calculate the value of the equilibrium constant,...Ch. 17.6 - Prob. 1.1ACPCh. 17.6 - What is the minimum volume of 0.0071 M NaCN(aq)...Ch. 17.6 - Use the formation constant of [Au(CN)2] in...Ch. 17.6 - Silver undergoes similar reactions as those shown...Ch. 17.6 - Write a balanced chemical equation for the...Ch. 17.6 - Phosphate ions are abundant in cells, both as the...Ch. 17.6 - A typical total phosphate concentration in a cell,...Ch. 17 - Does the pH of the solution increase, decrease or...Ch. 17 - Does the pH of the solution increase, decrease, or...Ch. 17 - What is the pH of a solution that consists of 0.20...Ch. 17 - What is the pH of 0.15 M acetic acid to which 1.56...Ch. 17 - What is the pH of the solution that results from...Ch. 17 - What is the pH of the solution that results from...Ch. 17 - What is the pH of the buffer solution that...Ch. 17 - Lactic acid (CH3CHOHCO2H) is found in sour milk,...Ch. 17 - What mass of sodium acetate, NaCH3CO2, must he...Ch. 17 - What mass of ammonium chloride, NH4Cl, must be...Ch. 17 - Calculate the pH of a solution that has an acetic...Ch. 17 - Calculate the pH of a solution that has an...Ch. 17 - What must the ratio of acetic acid to acetate ion...Ch. 17 - What must the ratio of H2PO4 to HPO42 be to have a...Ch. 17 - A buffer is composed of formic acid and its...Ch. 17 - A buffer solution is composed of 1.360 g of KH2PO4...Ch. 17 - Which of the following combinations would be the...Ch. 17 - Which of the following combinations would be the...Ch. 17 - Describe how to prepare a buffer solution from...Ch. 17 - Describe how to prepare a buffer solution from NH3...Ch. 17 - Determine the volume (in mL) of 1.00 M NaOH that...Ch. 17 - Determine the volume (in mL) of 1.00 M HC1 that...Ch. 17 - A buffer solution was prepared by adding 4.95 g of...Ch. 17 - You dissolve 0.425 g of NaOH in 2.00 L of a buffer...Ch. 17 - A buffer solution is prepared by adding 0.125 mol...Ch. 17 - What is the pH change when 20.0 mL of 0.100 M NaOH...Ch. 17 - Phenol, C6H5OH, is a weak organic acid. Suppose...Ch. 17 - Assume you dissolve 0.235 g of the weak acid...Ch. 17 - You require 36.78 mL of 0.0105 M HCl to reach the...Ch. 17 - A titration of 25.0 mL of a solution of the weak...Ch. 17 - Without doing detailed calculations, sketch the...Ch. 17 - Without doing detailed calculations, sketch the...Ch. 17 - You titrate 25.0 mL of 0.10 M NH3 with 0.10 M HCl....Ch. 17 - Using Figure 17.11, suggest an indicator to use in...Ch. 17 - Using Figure 17.11, suggest an indicator to use in...Ch. 17 - Name two insoluble salts of each of the following...Ch. 17 - Prob. 38PSCh. 17 - Using the solubility guidelines (Figure 3.10),...Ch. 17 - Predict whether each of the fallowing is insoluble...Ch. 17 - For each of the following insoluble salts, (1)...Ch. 17 - Prob. 42PSCh. 17 - When 1.55 g of solid thallium(I) bromide is added...Ch. 17 - At 20 C, a saturated aqueous solution of silver...Ch. 17 - When 250 mg of SrF2, strontium fluoride, is added...Ch. 17 - Calcium hydroxide, Ca(OH)2, dissolves in water to...Ch. 17 - You add 0.979 g of Pb(OH)2 to 1.00 L of pure water...Ch. 17 - You place 1.234 g of solid Ca(OH)2 in 1.00 L of...Ch. 17 - Estimate the solubility of silver iodide in pure...Ch. 17 - What is the molar concentration of Au+(aq) in a...Ch. 17 - Prob. 51PSCh. 17 - Estimate the solubility of lead(II) bromide (a) in...Ch. 17 - The Ksp value for radium sulfate, RaSO4, is 4.2 ...Ch. 17 - If 55 mg of lead(II) sulfate is placed in 250 mL...Ch. 17 - Prob. 55PSCh. 17 - Prob. 56PSCh. 17 - Calculate the molar solubility of silver...Ch. 17 - Calculate the solubility of silver bromide, AgBr,...Ch. 17 - Compare the solubility, in milligrams per...Ch. 17 - What is the solubility, in milligrams per...Ch. 17 - Calculate the solubility, in moles per liter, of...Ch. 17 - Calculate the solubility, in moles per liter, of...Ch. 17 - Which insoluble compound in each pair should be...Ch. 17 - Which compound in each pair is more soluble in...Ch. 17 - You have a solution that has a lead(II) ion...Ch. 17 - Sodium carbonate is added to a solution in which...Ch. 17 - If the concentration of Zn2+ in 10.0 mL of water...Ch. 17 - You have 95 mL of a solution that has a lead(II)...Ch. 17 - Prob. 69PSCh. 17 - Will a precipitate of Mg(OH)2 form when 25.0 mL of...Ch. 17 - Zinc hydroxide is amphoteric (Section 16.10). Use...Ch. 17 - Solid silver iodide, AgI, can be dissolved by...Ch. 17 - What amount of ammonia (moles) must be added to...Ch. 17 - Can you dissolve 15.0 mg of AuCl in 100.0 mL of...Ch. 17 - What is the solubility of AgCl (a) in pure water...Ch. 17 - Prob. 76PSCh. 17 - Prob. 77GQCh. 17 - Prob. 78GQCh. 17 - Prob. 79GQCh. 17 - Calculate the hydronium ion concentration and the...Ch. 17 - Calculate the hydronium ion concentration and the...Ch. 17 - For each of the following cases, decide whether...Ch. 17 - Prob. 83GQCh. 17 - A sample of hard water contains about 2.0 103 M...Ch. 17 - What is the pH of a buffer solution prepared from...Ch. 17 - Prob. 86GQCh. 17 - Describe the effect on the pH of the following...Ch. 17 - What volume of 0.120 M NaOH must be added to 100....Ch. 17 - A buffer solution is prepared by dissolving 1.50 g...Ch. 17 - What volume of 0.200 M HCl must be added to 500.0...Ch. 17 - What is the equilibrium constant for the following...Ch. 17 - Calculate the equilibrium constant for the...Ch. 17 - Prob. 93GQCh. 17 - The solubility product constant for calcium...Ch. 17 - In principle, the ions Ba2+ and Ca2+ can be...Ch. 17 - A solution contains 0.10 M iodide ion, I, and 0.10...Ch. 17 - A solution contains Ca2+ and Pb2+ ions, both at a...Ch. 17 - Prob. 98GQCh. 17 - Prob. 99GQCh. 17 - Prob. 100GQCh. 17 - Each pair of ions below is found together in...Ch. 17 - Each pair of ions below is found together in...Ch. 17 - The cations Ba2+ and Sr2+ can be precipitated as...Ch. 17 - You will often work with salts of Fe3+, Pb2+, and...Ch. 17 - Aniline hydrochloride, (C6H5NH3)Cl, is a weak...Ch. 17 - The weak base ethanolamine. HOCH2CH2NH2, can be...Ch. 17 - For the titration of 50.0 mL of 0.150 M...Ch. 17 - A buffer solution with it pH of 12.00 consists of...Ch. 17 - To have a buffer with a pH of 2.50, what volume of...Ch. 17 - What mass of Na3PO4 must be added to 80.0 mL of...Ch. 17 - You have a solution that contains AgNO3, Pb(NO3)2,...Ch. 17 - Prob. 112ILCh. 17 - Suggest a method for separating a precipitate...Ch. 17 - Prob. 114SCQCh. 17 - Prob. 115SCQCh. 17 - Two acids, each approximately 0.01 M in...Ch. 17 - Composition diagrams, commonly known as alpha...Ch. 17 - The composition diagram, or alpha plot, for the...Ch. 17 - The chemical name for aspirin is acetylsalicylic...Ch. 17 - Prob. 120SCQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Write the acid dissociation reaction for formic acid, HCO2H.arrow_forwardSodium bicarbonate, NaHCO3, is used in baking powder formulations and in the manufacture of plastics and ceramics, among other things. If 26.3 g of the compound is dissolved in enough water to make 200. mL of solution, what is the concentration of NaHCO3? What are the concentrations of the ions in solution?arrow_forwardWhat is pH of a 0.1 Molar solution of oxalic acid in waterarrow_forward
- If the molar concentration of OH- is 1.0 x 10-7, the pH is what?arrow_forwardSolubility of gases in water increases with increasing mass. Explain? What is the relationship between the value of pKa and the strength of a weak acid?arrow_forward4. What is the pH of a solution that contains 0.35 mol/L of the strons base NaOH?arrow_forward
- Calculate the pH of a 0.25 M solution of aluminum chloride, which dissolves completely to give the hydrated aluminum ion [Al(H2O)6]3+ in solution.arrow_forwardThe pH of an aqueous solution of 0.137 M sodium hypochlorite, NaClO (aq), is ________________ This solution is: ________ (ACIDIC, BASIC, OR NEUTRAL?)arrow_forwardWrite the molecular equation and the net ionic equation for the neutralization of hydrocyanic acid, HCN, by lithium hydroxide, LiOH, both in aqueous solution.arrow_forward
- Calculate the pH of the weak acid, lactic acid (HC3H5O3), if the concentration is 0.10 mol/L and the Ka value is 1.4 x 10-4arrow_forwardThe pH of an aqueous solution of acetic acid (CH3COOH) is 2.0. What is the initial molar concentration of CH3COOH, if its acid ionization constant is Ka = 1.8×10–5?arrow_forwardIron in drinking water is removed by precipitation of the Fe3+ ion by reaction with NaOH to produce iron(III) hydroxide. Write the balanced chemical equation and the net ionic equation for this reaction.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Types of Matter: Elements, Compounds and Mixtures; Author: Professor Dave Explains;https://www.youtube.com/watch?v=dggHWvFJ8Xs;License: Standard YouTube License, CC-BY