
Concept explainers
(a)
Interpretation: Major organic product of monobromination of pentane at
Concept introduction: The monobromination performed with ultraviolet light proceeds via radical chain mechanism.
Tertiary
The phenomenon of hyperconjugation refers to donation of
(b)
Interpretation: All Newman projections of major brominated products in staggered conformations should be drawn.
Concept introduction: Various interconvertible forms that result from rotation around the
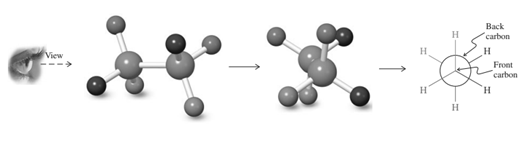
Thus in Newman's projection of simple ethane molecule the “front”
(c)
Interpretation: Qualitative plot of potential energy against torsional angle for
Concept introduction: As rotation is carried out along
The potential energy shows peaks and falls. This corresponds to transition from staggered and eclipsed conformation molecule adopts with each
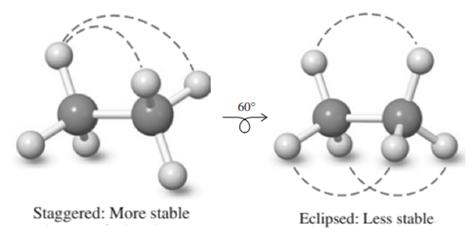
Staggered conformation that has substituents on both carbons farthest apart is regarded most stable due to least torsional strain, whereas the eclipsed has large amount of torsional strain due to steric repulsions. Therefore in potential-energy diagram, peak corresponds to eclipsed while the valley corresponds to stable staggered conformation.
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Organic Chemistry: Structure and Function
- Draw the conformational analysis of 1-bromobutane looking at the C1-C2 bond. Provide the appropriate names for each conformation.arrow_forwardIn addition to more highly fluorinated products, fluorination of 2-methylbutane yields a mixture of compounds with the formula C5H10F2. Draw the structures of all the isomers with the formula C5H10F2 that would be produced and label with a star all the chiral centers present in their structures.arrow_forwardFrom studies of the dipole moment of 1,2-dichloroethane in the gas phase at room temperature (25°C), it is estimated that the ratio of molecules in the anti conformation to gauche conformation is 7.6 to 1. Calculate the difference in Gibbs free energy between these two conformations.arrow_forward
- Draw structural formulas for the cis and trans isomers of hydrindane. Show each ring in its most stable conformation. Which of these isomers is more stable?arrow_forwardgive the degree of unsaturation of the compounds: Vanillin(C8H8O3) and ethyl ethocyacetate (C6H12O3). and draw their structuresarrow_forwardDraw and name the six isomeric cyclopentane of molecular formula C7H14. These will include four constitutional isomers, of which two show geometric (cis-trans) stereoisomerism.arrow_forward
- A difficult problem in the synthesis of PGF2α is the introduction of the OH group at C15 in the desired conguration.a. Label this stereogenic center as R or S.b. A well known synthesis of PGF2α involves reaction of A with Zn(BH4)2, a metal hydride reagent similar in reactivity to NaBH4, to form two isomeric products, B and C. Draw their structures and indicate their stereochemical relationship.c. Suggest a reagent to convert A to the single stereoisomer X.arrow_forwardHow would you best describe the C-C bonds lengths in benzene relative to cyclohexane? Hypothesize why these results are observed.arrow_forwardA difficult problem in the synthesis of PGF2α is the introduction of the OH group at C15 in the desired configuration. a. Label this stereogenic center as R or S. b. A well-known synthesis of PGF2α involves reaction of A with Zn(BH4)2, a metal hydride reagent similar in reactivity to NaBH4, to form two isomeric products, B and C. Draw their structures and indicate their stereochemical relationship. d. Suggest a reagent to convert A to the single stereoisomer X.arrow_forward
- Draw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that shows: Q.)E,Z isomerism but not chiralityarrow_forwarda) When (Z)-3-methylhex-3-ene undergoes hydroboration–oxidation, two isomeric products are formed. Give their structures, and label each asymmetric carbon atom as (R) or (S). What is the relationship between these isomers?arrow_forwardWhat is the degree of unsaturation and structure for C8H5NO2 ?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

