
Interpretation:Diagrams drawn for the propagation steps of radical bromination of benzene should be identified as either representative of early or late transition states.
Concept introduction:Hammond postulate states that exothermic reactions are characterized by early transition states that resemble the structure of substrate more than the product. Slow or endothermic processes are characterized by late transition states that resemble the almost formed bonds as in the products.
Analogous to hydrocarbons the benzene can also undergo initiation to generate bromine radicals; propagation of radicals formed and finally termination. This sequence can be outlined as follows:
Step1: Initiation via homolytic cleavage of
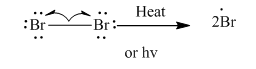
Step2: Propagation: In first of the propagation steps bromine radical from step 1 abstracts hydrogen radical from
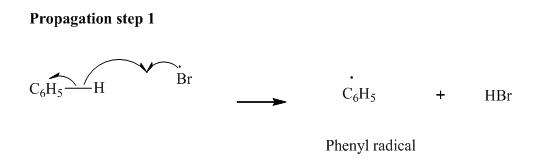
In subsequent propagation step,phenyl radical abstracts
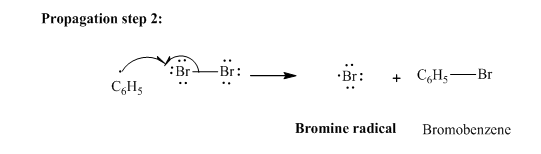
Step3: Termination: Bromine radicals generated in propagation steps get quenched upon combination with one another illustrated as follows:
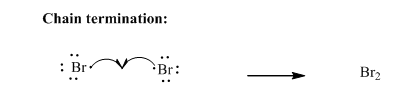
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Organic Chemistry: Structure and Function
- In each of the following, which reaction mechanism assumption is apparently being violated? Explain your answers. a. A reaction takes place more rapidly when the concentration of reactants is decreased. b. A reaction takes place more rapidly when the reaction mixture is cooled. c. The reaction rate of A+BAB increases as the concentration of A is increased but does not change as the concentration of B is increased.arrow_forwardAn elevated level of the enzyme alkaline phosphatase (ALP) in human serum is an indication of possible liver or bone disorder. The level of serum ALP is so low that it is very difficult to measure directly. However. ALP catalyzes a number of reactions, and its relative concentration can be determined by measuring the rate of one of these reactions under controlled conditions. One such reaction is the conversion of p-nitrophenyI phosphate (PNPP) to p-nitrophenoxide ion (PNP) and phosphate ion. Control of temperature during the test is very important; the rate of the reaction increases 1.47 times if the temperature changes from 30 C to 37 C. What is the activation energy for the ALP-catalyzed conversion of PNPP to PNP and phosphate?arrow_forwardThree first-order reactions have the following activation energies: (a) Which reaction is the fastest? (b) Which reaction has the largest half-life? (c) Which reaction has the largest rate?arrow_forward
- n summary, a reaction that forms mostly all products will have a ∆G _______ while a reaction that is mostly unreactive will have a ∆G _______arrow_forwardWhich of the following is true?A. The number of reactant collisions per unit time is equal to the rate constant at a specific temperatureB. The rate constant is lower if the activation energy is higherC. Increasing the temperature increases the activation enrgy of the reactionD. The reaction rate is equal to the number of activated complexes that forms during the reactionarrow_forwardThe 4 molecules are reacting with CH3S- in acetone. Which transition state numbered 1-4 represents A, B, C, and D? Why?arrow_forward
- Find the specific Rate constantarrow_forwardDraw the reaction coordinate of catalyzed and uncatalyzed reactions. Indicate the substrate (S), product (P), activation energy (∆G+), transition state (T), and free energy change (∆G°).arrow_forwardEnzymes are remarkably efficient catalysts that can increase reaction rates by as many as 20 orders of magnitude.(a) How does an enzyme affect the transition state of a reac-tion, and how does this effect increase the reaction rate?(b) What characteristics of enzymes give them this tremendous effectiveness as catalysts?arrow_forward
- I drew out the reaction and the transition states but I am not sure how to draw the energy diagram for compounds with similar mechanismsarrow_forwardAn increase in temperature will Group of answer choices decrease the rate of a reaction. decrease the activation energy for a reaction. increase the amount of collisions in the system. increase the number of elementary steps in the reaction. none of the above.arrow_forwardConsider the following reaction sequence. Identify the missing intermediates.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





