
Interpretation: Potential-energy versus reaction-coordinate diagrams for the two propagation steps for monobromination of methane should be sketched.
Concept introduction: The mechanism for monobromination comprises of three stages illustrated as follows:
Step1: Initiation via homolytic cleavage of
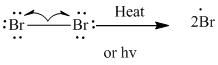
Step2: Propagation: In first of the propagation step bromine radical from step 1 abstracts hydrogen radical from methane as follows:
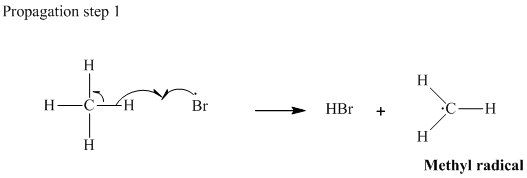
In subsequent propagation step methyl radical abstracts
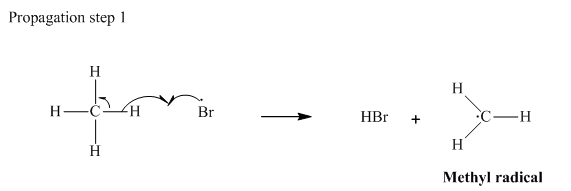
Step3: Termination: Radicals generated in propagation steps get quenched upon combination with one another. Thus termination steps are essentially the radical− radical combination illustrated as follows:
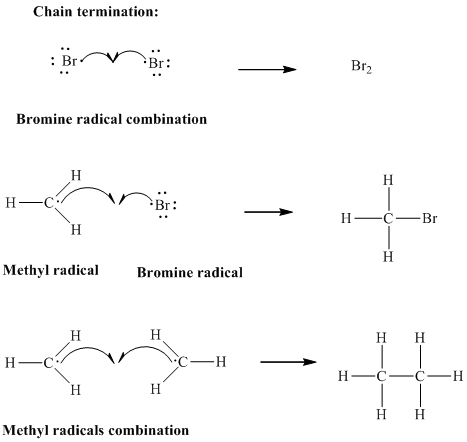
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Organic Chemistry: Structure and Function
- Does increasing the energy barrier for an SN2 reaction increase or decrease the magnitude of the rate constant for the reactionarrow_forwardAlcohols are important for organic synthesis, especially in situations involving alkenes. The alcohol might be the desired product, or the OH group might be transformed into another functional group via halogenation, oxidation, or perhaps conversion to a sulfonic ester derivative. Formation of an alcohol from an alkene is particularly powerful because conditions can be chosen to produce either the Markovnikov or non-Markovnikov product from an unsymmetrical alkene. Using your reaction roadmap as a guide, show how to convert 4-methyl-1-pentene into 5-methylhexanenitrile. You must use 4-methyl-1-pentene and sodium cyanide as the source of all carbon atoms in the target molecule. Show all reagents needed and all molecules synthesized along the way.arrow_forwardDraw the electron-pushing mechanism for the propagation steps of the allylic bromination reactions below. You may omit NBS in your mechanism, and use Br and Br2.arrow_forward
- Consider the following statement in reference to SN1, SN2, E1, and E2 reactions of haloalkanes. To which mechanism(s), if any, does the statement apply? Order of reactivity of haloalkanes is methyl . 1° > 2° > 3°.arrow_forwardPlease show the reaction mechanisms of the [3 + 2] cycloadditions of (A) Ozone and (B) OsO4 to cis-2-butene to form their respective 5-membered rings.arrow_forward5c) Write the rate equation for the following SN1 reaction:arrow_forward
- i need help filling out the following SN2 reactions with appopiate reactants, products, or reagents,arrow_forwardDetermine the structure of the major monochlorination product of 2,4- dimethylpentane shown below using the reactivity order 5 : 3.5 : 1 for tertiary : secondary : primary hydrogens respectively.arrow_forwardDoes the data provide evidence for the claim that the reaction of Br2 with alkenes proceeds via anti‐addition? The mleting point of the product was found to be 234.5-235.5C chemical reaction: trans-stilbene+pyrimindine tribromide --->acetic acid 1,2-di bromo-1,2-diphenylethanearrow_forward
- Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- ΔH=-75 kJ/mol Rate = k [CH3CH2Br][CH3COO-] Which reaction energy profile would be the best representative of the data provided?arrow_forward1. i.What are the various ways by which alkenes may be synthesized? ii. Give two examples each of Unsymmetrical alkenes and reagents. iii. Give two examples of reactions of alkenes that result in Anti-Markonikov’s addition productsarrow_forwardSimilar to alkanes, hydrogen gas can undergo radical bromination according to the reactionbelow. Propose a chain-reaction mechanism for this reaction, including an initiation step, propagation steps, and two plausible termination steps. The homolytic bond dissociation energy for Br-Br is 46 kcal*mole^-1, for H-Br is 88 kcal*mole^-1 and for H-H is 104 kcal*mole^-1arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


