
(a)
Interpretation:Whether products with reasonable selectivity are obtained should be identified.
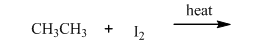
Concept introduction: Tertiary
The phenomenon of hyperconjugation refers to donation of
(b)
Interpretation:Whether products with reasonable selectivity are obtained should be identified.
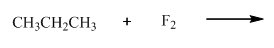
Concept introduction: Tertiary
The phenomenon of hyperconjugation refers to donation of
(c)
Interpretation:Whether products with reasonable selectivity are obtained should be identified.
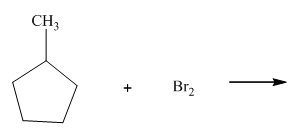
Concept introduction:Tertiary
The phenomenon of hyperconjugation refers to donation of
(d)
Interpretation:Whether products with reasonable selectivity are obtained should be identified.
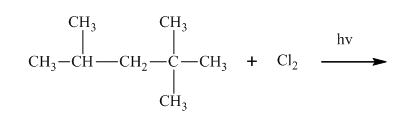
Concept introduction:Tertiary
The phenomenon of hyperconjugation refers to donation of
(e)
Interpretation:Whether products with reasonable selectivity are obtained should be identified.
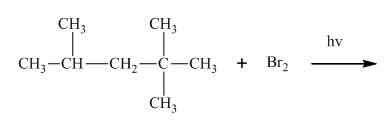
Concept introduction:Tertiary
The phenomenon of hyperconjugation refers to donation of
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Organic Chemistry: Structure and Function
- 66) Which of the following statements is false? Reactions following Markonikov’s Rule, form the less substituted carbocation. In the acid-catalyzed hydration of alkenes, many times a carbocation rearranges to form a more stable structure. Alkoxides, hydroxide anion, and Grignard reagent, are ALL classified as strong nucleophiles. None of the above.arrow_forwardWhich would be the best reagent for carrying out the reaction shown below?arrow_forwardWhat major product is expected from the reaction shown in Image 18?arrow_forward
- 1. Draw a reasonable arrow-pushing mechanism for the transformation shown along. 2. Identify nucleophiles and electrophiles 3. Name any type of reactions taking place like E2 or E1 4. Account for any regio- or stereoselectivityarrow_forwardAny help with this would be great! Explanations definitely welcome, thanks in advance for any help:) What is the major product of the reactions?arrow_forwardWhy is this considered an Elimination E2 reaction? C₃H₈O (l) → C3H6 (g) + H2O (l) propan-1-ol → propene + waterarrow_forward
- Rank these in order of increasing reactivity in an SN1 reactionarrow_forwardThe name for Reaction 1 is _____ while Reaction 2 is called _____. Choices: A. Williamson Ether synthesis, B. Hydration, C. Epoxidation, D. Acidic cleavage The reagent/s for REACTION 1 is/are ______ Choices: A. m-chloroperoxybenzoic acid, B. H2O/H3PO4, C. H3O+, D. HI The reagent/s for REACTION 2 is/are ______ Choices: A. m-chloroperoxybenzoic acid, B. water in acidic medium, C. dilute acid, D. hydroiodic acidarrow_forward1. Which of the following is the strongest nucleophile? Refer to the diagram below with an MeOH, MeSH, etc. 2. Which of the following is true about the stereochemistry of SN1 reaction?a.retention of configuration at the electrophilic centerb.50:50 mixture of retention and inversion of configuration at the electrophilic centerc.inversion of configuration at the electrophilic centerd. slightly more inversion than retention at the electrophilic centerarrow_forward
- Consider the following transformation below, which Prof. Ingoglia conducted during his post-doctoral research. a. Draw the product of the reaction in the box provided. b. Provide an arrow-pushing mechanism for the formation of the product you drew in part (a).arrow_forwardPlease answer the attached question. Give the major products for the appropriate SN1 and E1 reactions and please explain how you calculated your answers. Thanks.arrow_forwardDraw the major organic product of this E1 elimination reaction. Ignore byproducts.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

