
(a)
Interpretation: The
Concept introduction: At extremely elevated temperature, the
The phenomenon of hyperconjugation refers to donation of
(b)
Interpretation: The preferred product formed from attack of
Concept introduction: After the homolytic cleavage radicals may recombine with one another and result in variety of
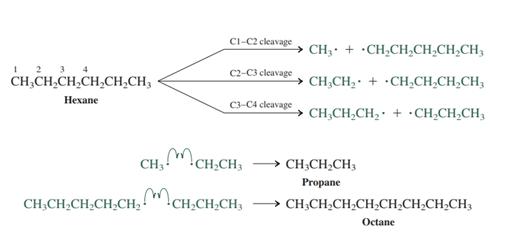
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Organic Chemistry: Structure and Function
- Which is more stable - a phenyl radical, [C6H5]•, or a benzyl radical, [C6H5CH2]• - and why?arrow_forwardWhat’s the role of NaBH4 in Oxymercuration-Demercuration of cyclohexene?arrow_forwardPrimary halogenoalkanes undergo hydrolysis by SN2 mechanism. State the meaning of S, N and 2 in SN2. Using curly arrows to show the movement of electrons give the reaction mechanism for the hydrolysis of 1-bromopropane [CH3CH2CH2Br] with aqueous hydroxide ions (OH-). The description should be detailed and must include the type of bond fission that takes place. You may sketch and insert suitable diagrams to aid your description if you wish.arrow_forward
- Acetylacetone (pentane-2,4-dione) reacts with sodium hydroxide to give water and the sodium salt of a carbanion. Write a complete structural formula for the carbanion, and use resonance forms to show the stabilization of the carbanion.arrow_forwardWrite the propagation steps leading to the formation of dichloromethane (CH2Cl2) from chloromethanearrow_forwardFollowing is a balanced equation for bromination of toluene. (a) Using the values for bond dissociation enthalpies given in Appendix 3, calculate H0 for this reaction. (b) Propose a pair of chain propagation steps and show that they add up to the observed reaction. (c) Calculate H0 for each chain propagation step. (d) Which propagation step is rate-determining?arrow_forward
- butene-1 acts on a combination of three reactants.Write the corresponding reactions. a) H2, Br2, N2 b) Na, NaOH, HCl c) H2O, HBr, KMnO4 d) Cl2, CO2, H2SO4arrow_forwardWrite structural formulas for toluene (C6H5CH3) and for benzoic acid (C6H5CO2H) (a) as resonance hybrids of two Kekulé forms and (b) with the Robinson symbol.arrow_forwardIllustrate how diazomethane and ozone can both behave as 1,3-dipoles. Indicate, by drawing their resonance forms, how they can do so and draw the products of their reaction with styrenearrow_forward
- 3. The reaction of bromine with 3-hexanone produces two isomeric products of formula C6H11BrO. Give the mechanism for obtaining the two isomers.arrow_forwardBromine reacts with alkenes in methanol according to the equation (see image 1). When this reaction was carried out with 4-tert-butylcyclohexene, only one isomer was formed with the molecular formula C12H23BrO (80% yield) a) Which of the following is the structure more reasonable for this compound? (see image 2) b) Explain your reasoning through a corresponding mechanismarrow_forwardRead these directions carefully. For the reaction of 3-methyl-1-propene with Cl2 and H2O shown below, fill in the details of the mechanism. Draw the appropriate chemical structures and use an arrow to show how pairs of electrons are moved to make and break bonds during the reaction. Be sure to write all lone pairs of electrons and all formal charges. Finally, in the boxes provided by the arrows, write which kind of mechanistic element is being indicated, such as "make a bond", "add a proton", etc.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

