
Organic Chemistry: Structure and Function
8th Edition
ISBN: 9781319079451
Author: K. Peter C. Vollhardt, Neil E. Schore
Publisher: W. H. Freeman
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 3.7, Problem 3.7E
Interpretation Introduction
Interpretation: Various products formed from the radical monochlorination of butane along with ratios of different products should be written.
Concept introduction: The monochlorination performed with ultraviolet light proceeds via radical chain mechanism. Chlorine transforms
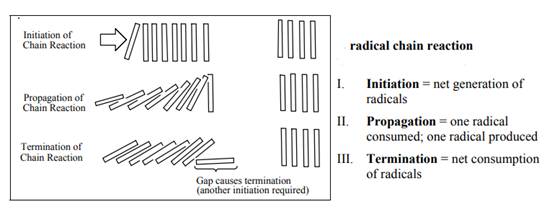
The fundamental radical chain mechanism is summarized in the illustration as follows:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Illustrate how diazomethane and ozone can both behave as 1,3-dipoles. Indicate, by drawing their resonance forms, how they can do so and draw the products of their reaction with styrene
What’s the role of NaBH4 in Oxymercuration-Demercuration of cyclohexene?
2-methyl-2-butanol was mixed with sulfuric acid. Draw out the structures for this reaction and state the limiting reagent.
Chapter 3 Solutions
Organic Chemistry: Structure and Function
Ch. 3.1 - Prob. 3.2TIYCh. 3.1 - Prob. 3.3ECh. 3.4 - Prob. 3.5TIYCh. 3.5 - Prob. 3.6ECh. 3.7 - Prob. 3.7ECh. 3.7 - Prob. 3.9TIYCh. 3.9 - Prob. 3.11TIYCh. 3.11 - Prob. 3.12ECh. 3 - Prob. 15PCh. 3 - Prob. 16P
Ch. 3 - Prob. 17PCh. 3 - Prob. 18PCh. 3 - Prob. 19PCh. 3 - Prob. 20PCh. 3 - Prob. 21PCh. 3 - Prob. 22PCh. 3 - Prob. 23PCh. 3 - Prob. 24PCh. 3 - Prob. 25PCh. 3 - Prob. 26PCh. 3 - Prob. 27PCh. 3 - Prob. 28PCh. 3 - Prob. 29PCh. 3 - Prob. 30PCh. 3 - Prob. 31PCh. 3 - Prob. 32PCh. 3 - Prob. 33PCh. 3 - Prob. 34PCh. 3 - Prob. 35PCh. 3 - Prob. 36PCh. 3 - Prob. 37PCh. 3 - Prob. 38PCh. 3 - Prob. 39PCh. 3 - Prob. 40PCh. 3 - Prob. 41PCh. 3 - Prob. 42PCh. 3 - Prob. 43PCh. 3 - Prob. 44PCh. 3 - Prob. 45PCh. 3 - Prob. 46PCh. 3 - Prob. 47PCh. 3 - Prob. 48PCh. 3 - Prob. 49PCh. 3 - Prob. 50PCh. 3 - Prob. 51P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Why does the addition of an electron from zinc to maleic acid occur to initiate reduction of the double bond while the same reaction does not occur with 2-butene?arrow_forwardWrite the Wurtz – Fittig reaction for the preparation of toluene.arrow_forwardGive the reaction equation for the alkene given to you assuming the reactant was Br2, in the presence ofwater indicating the stereochemistry of all products formed.arrow_forward
- Consider the following reaction. 1,3-butadiene + maleic anhydride ---> Provide the structural formula of the product?arrow_forwardUpon successful bromination of an alkene, what is the molecular weight increase of the compound? The number commonly associated with petroleum products. Often mentioned on refueling stations. The difference in molecular weights of chloropropane and 2-chloropropane When a haloalkane is heated with alcoholic solution of potassium hydroxide, what will be the result? What does Kerosene tends to contain these organic compounds? Wet Filter paper containing iodoform weighs 2.1 grams. Upon drying only 0.6 grams of iodoform is left. Assuming 1 gram of moisture is removed, what is the weight of the filter paper?arrow_forwardHow do you expect the composition of cyclohexane and toluene to change over the course of a distillation?arrow_forward
- Acrolein and 1,3-cyclohexadiene react in a one-step concerted manner to yield a single product. Give the structure of the product. What kind of reaction is this an example of? In terms of this reaction, how would you classify acrolein? How would you classify 1,3-cyclohexadiene? Hint: acrolein is not a systematic name so you may need to look up its structure if you are not already familiar with it.arrow_forwardAccount for the fact that addition of HCl to 1-bromopropene gives exclusively 1-bromo-1-chloropropane.arrow_forwardName, draw and describe the organic product of the reaction between 2-methylbut-1-ene and H2O in the presence of H2SO4 and provide a clear rationale as to why this is the major product of the reaction.arrow_forward
- Give the product for the addition of chlorine when HCl reacts with 3-Hexyne in acetic acid (CH3COOH). Show the correct stereochemistry, (is the product Z or E isomer).arrow_forwardWhat is the role of benzoyl peroxide in addition polymerisation of alkenes? Explain its mode of action with the help of an example.arrow_forwardThe reaction of DBU with 2-bromoheptane gives two alkene products, what are they? Which of these would you expect to be the major product? Explain your answer.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License