
Concept explainers
(a)
Interpretation:
Taking a basis of 1 mol of feed gas, the moles of each component of the feed product mixture and extent of reaction should be calculated.
Concept Introduction
The flow chart for the given situation would be helpful to solve the problem.
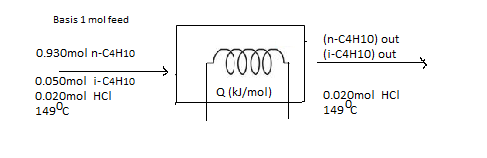
(b)
Interpretation:
First, the calculation of the standard heat of the isomerization reaction (kJ) should be found. Then, taking the feed and product species at 25? as references, an inlet-outlet enthalpy table should be prepared and component amounts (mol) and specific enthalpies (kJ/mol) should be calculated and filled in.
Concept introduction:
The standard heat of formation will be helpful to solve the problem as:
(c)
Interpretation:
The amount of heat transferred should be calculated in kJ and the required heat transfer for a reactor feed of 325 kJ/h should be determined in kW.
Concept introduction:
The heat transferred is calculated using formula:
(d)
Interpretation:
The calculated results should be used to estimate the heat of the isomerization reaction at 149? and the assumptions built into the estimation should be listed.
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
- When the following gas reaction reaches equilibrium, at a certain temperature, the moles fractions of the four reactive species satisfy the following relation (we will use "y" for mole fractions since all of these reagents and products are gases): CO+H20 CO2+H2 K= [yCO2][yH2] [yco][yH20] Where K= 1. Assume that the feed introduced to the reactor is 2 mol of CO and 4 mol of H20 only. Also assume that the reaction reaches equilibrium. a) Calculate the extent of reaction in equilibrium (Šeg) b) Calculate the equilibrium composition, for this you need to calculate the mole fraction for all reactants and products.arrow_forwardFeed gas containing of 78.5mol % H₂, 21% of N₂ & 0.5% of Ar is mixed with recycle gas and enters a reactor where 15% N₂ is converted to NH3 as per the reaction. Ammonia from the exit of the reactor is completely separated from unconverted gases. To avoid the buildup of inerts, a small fraction (5%) of the unreacted gases purged and the balance recycled. USING ASPEN/HYSYS Draw the process flow sheet Product rate and Purge rate Basis:100mol/hrarrow_forwardEthane is chlorinated in a continuous reactor: C2H6 + Cl2 à C2H5Cl + HCl Some of the product monochloroethane is further chlorinated in an undesired side relation: C2H5Cl + Cl2 à C2H4Cl2 + HCl Take a basis of 100 mol C2H5Cl produced. Assume that the feed contains only ethane and chlorine and that all of the chlorine is consumed and carry out a degree-of-freedom analysis based on atomic species balances. The reactor is designed to yield a 15% conversion of ethane and a selectivity of 14 mol C2H5Cl/mol C2H4Cl2. Calculate the feed ratio (mol Cl2/mol C2H6) and the fractional yield of monochloroethane.arrow_forward
- The synthesis of methanol from carbon monoxide and hydrogen is carried out in a continuous vapor-phase reactor at 5.00 atm absolute. The feed contains cO and H2 in stoichiometric proportion and enters the reactor at 25.0°C and 5.00 atm at a rate of 31.1 m³/h. The product stream emerges from the reactor at 157°C. The rate of heat transfer from the reactor is 21.0 kW. Calculate the fractional conversion (0 to 1) of carbon monoxide achieved and the volumetric flow rate (m3/h) of the product stream. f = Vout ! m3/harrow_forwardMethane is burned with air in a continuous combustion reactor that is at steady state.The feed to the reactor contains 7.80 mole % CH4, 19.4 mole % O2, and 72.8 mole % N4. The percentage conversion of methane is 90.0%, and the gas leaving the reactor contains 8 mol CO2/mol CO. Carry out a degree of freedom analysis of the process, and calculate the molar composition of the product stream.arrow_forwardTo prepare a delicious banana milk on an industrial scale, there is an experimental process that consists of seven unit operations. A stream of banana (whose components are fruit, peel, and moisture) is fed to a peeler, where 99.9% of the peel is removed. This stream should become pulp, so it is mixed with an additive Q to avoid pulp oxidation. This mixture is then sent to an extruder press wherein a stream of a 50:50 mixture of additives Q and R is added. The resulting flow of 30 kg/h has a composition of 2% additive R and 5% additive Q. This flow is then fed to an agitated mixer. To the same mixer is fed a dairy stream of 250 kg/h that contains 70% milk (the rest is water) and a sweet aqueous stream that contains 30% sugar. For each 25 kg/h of dairy flow, 2 kg/h of the sweet flow is fed. The stream that leaves the agitated mixer has 5% fruit and 0.005% peel and enters a series of two thermal processes in which each operation allows 5% of the water and 2% of the milk to evaporate. The…arrow_forward
- Consider the combustion of one mole of methane gas:CH4(gas) + 2O2(gas) --> CO2 (gas) + 2H2O(gas).The system is at standard temperature (298 K) and pressure (105 Pa) both before and after the reaction. 1.) First imagine the process of converting a mole of methane into its elemental consituents (graphite and hydrogen gas). Use the data at the back of this book to find ΔH for this process. 2.) Now imagine forming a mole of CO2 and two moles of water vapor from their elemental constituents. Determine ΔH for this process. Question: What is ΔH for the actual reaction in which methane and oxygen form carbon dioxide and water vapor directly? Explain.arrow_forwardConsider the combustion of one mole of methane gas:CH4(gas) + 2O2(gas) --> CO2 (gas) + 2H2O(gas).The system is at standard temperature (298 K) and pressure (105 Pa) both before and after the reaction. 1.) First imagine the process of converting a mole of methane into its elemental consituents (graphite and hydrogen gas). Use the data at the back of this book to find ΔH for this process. 2.) Now imagine forming a mole of CO2 and two moles of water vapor from their elemental constituents. Determine ΔH for this process. Question: How much heat is given off during this reaction, assuming that no "other" forms of work are done?arrow_forwardOne gram-mole each of CO2, 02, and N2 are fed to a batch reactor and heated to 3000 K and 5.0 atm. The two reactions given here proceed to equilibrium (also shown are the equilibrium constants at 3000 K). CO2 =CO+02 1/2 K1 = (PcoPo,)/Pco; = 0.3272 atm'/2 02 +N2 =NO K2 = PNo/(Po,PN,)"/² = 0.1222arrow_forward
- In the combustion of heptane, CO, is produced. Assume that you want to produce 500 kg of dry ice per hour as shown in Fig. How many kilograms of heptane must be burned per hour? Other Products co, Solid 500 kg/hr Reactor C,H15 + 11 O2 7 CO, + 8 H;0 >arrow_forwardethylene oxide is produced by oxidation of ethylene 100 kmol of ethylene and 100 kmol of o2 are charged to a reactor.the % conversion of ethylene is 85 and%yield of ethylene oxide is 94.12 .calculate the composition of product stream leaving the reactor .the reactions takes place arearrow_forwardethylene oxide is prepared by oxidation of ethylene. 100Kmol of ethylene and 100Kmol of O2 are charged to a reactor.The percent conversion of ethylene is 85 and percent yield of C2H4O is 94.12. Calculate the composition of product steam leaving the reactor .The reactions taking place are: C2H4+0.5O2=C2H4O C2H4+3O2= 2CO2+2H2Oarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





