
The wastewater treatment plant at the Ossabaw Paper Company paper mill generates about 24 tonnes of sludge per day. The consistency of the sludge is 35%, meaning that the sludge contains 35 wt% solids and the balance liquids. The mill currently spends $40/tonne to dispose of the sludge in a landfill. The plant environmental engineer has determined that if the sludge consistency could be increased to 75%, the sludge could be incinerated (burned) to generate useful energy and to eliminate the environmental problems associated with landfill disposal.
A flowchart for a preliminary design of the proposed sludge-treatment process follows. For simplicity, we will assume that the liquid in the sludge is just water.
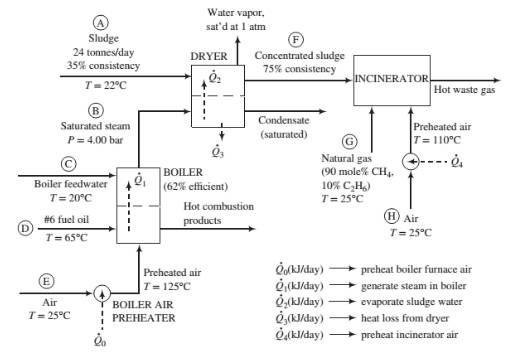 Process description: The sludge from the wastewater treatment plant (Stream (A) passes through a dryer where a portion of the water in the sludge is vaporized. The heat required for the vaporization comes from condensing saturated steam at 4.00 bar (Stream (B)). The steam fed to the dryer is produced in the plant’s oil-fired boiler from feedwater at 20°C (Stream (C)). The heat required to produce the steam is transferred from the boiler furnace, where fuel oil (Stream (D)) is burned with 25% excess air (Stream (E)). The concentrated sludge coming from the dryer (Stream (F)), which has a consistency of 75%, is fed to an incinerator. The heating value of the sludge is insufficient to keep the incinerator temperature high enough for complete combustion, so natural gas (Stream (G)) is used as a supplementary fuel. A stream of outside air at 25°C (Stream (H)) is heated to 110°C and fed to the incinerator along with the concentrated sludge and natural gas. The waste gas from the incinerator is discharged to the atmosphere.
Process description: The sludge from the wastewater treatment plant (Stream (A) passes through a dryer where a portion of the water in the sludge is vaporized. The heat required for the vaporization comes from condensing saturated steam at 4.00 bar (Stream (B)). The steam fed to the dryer is produced in the plant’s oil-fired boiler from feedwater at 20°C (Stream (C)). The heat required to produce the steam is transferred from the boiler furnace, where fuel oil (Stream (D)) is burned with 25% excess air (Stream (E)). The concentrated sludge coming from the dryer (Stream (F)), which has a consistency of 75%, is fed to an incinerator. The heating value of the sludge is insufficient to keep the incinerator temperature high enough for complete combustion, so natural gas (Stream (G)) is used as a supplementary fuel. A stream of outside air at 25°C (Stream (H)) is heated to 110°C and fed to the incinerator along with the concentrated sludge and natural gas. The waste gas from the incinerator is discharged to the atmosphere.
Fuel oil: The oil is a low-sulfur No. 6 fuel oil. Its ultimate (elemental) analysis on a weight basis is 87% C, 10% H, 0.84% S, and the balance oxygen, nitrogen, and nonvolatile ash. The higher heating value of the oil is 3.75 × 104kJ/kg and the heat capacity is Cp= 1.8 kJ/(kg·°C).
Boiler: The boiler has an efficiency of 62%, meaning that 62% of the heating value of the fuel oil burned is used to produce saturated steam at 4.00 bar from boiler feedwater at 20°C. Fuel oil at 65°C and dry air at 125°C are fed to the boiler furnace. The air feed rate is 25% in excess of the amount theoretically required for complete consumption of the fuel.
Sludge: The sludge from the wastewater treatment plant contains 35% w/w solids (S) and the balance liquids (which for the purposes of this problem may be treated as only water) and enters the dryer at 22°C. The sludge includes a number of volatile organic species, some of which may be toxic, and has a terrible odor. The heat capacity of the solids is approximately constant at 2.5 kJ/(kg·°C).
Dryer: The dryer has an efficiency of 55%, meaning that the heat transferred to the sludge, 02, is 55% of the total heat lost by the condensing steam, and the remainder,
Incinerator: The concentrated sludge has a heating value of 19,000 kJ/kg dry solids. For a feed sludge of 75% consistency, the incinerator requires 195 SCM natural gas/tonne wet sludge [ 1 SCM = 1 m3(STP)]. The theoretical air requirement for the sludge is 2.5 SCM air/10.000 kJ of heating value. Air is fed in 100% excess of the amount theoretically required to bum the sludge and the natural gas.
Use material and energy balances to calculate the mass flow rates (tonnes/day) of Streams (B), (C), (D), (E), (F), (G) and (H), and heat flows
- The money saved by implementing this process will be the current cost of disposing of the wastewater plant sludge in a landfill. Two major costs of implementing the process are the installed costs of the new dryer and incinerator. What other costs must be taken into account when determining the economic feasibility of the process? Why might management decide to go ahead with the project even if it proves to be unprofitable?
- What opportunities exist for improving the energy economy of the process? (Hint: Think about the need to preheat the fuel oil and the boiler and incinerator air streams and consider heat exchange possibilities.)
- The driving force for the introduction of this process is to eliminate the environmental cost of sludge disposal. What is that cost—that is, what environmental penalties and risks are associated with using landfills for hazardous waste disposal? What environmental problems might incineration introduce?
Learn your wayIncludes step-by-step video

Chapter 9 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Objects First with Java: A Practical Introduction Using BlueJ (6th Edition)
Starting Out With Visual Basic (7th Edition)
Introduction to Heat Transfer
- 2. In an ethanol production plant, a separator produces a 99% ethanol product from a feedstock stream containing 85% water and 15% ethanol at a rate of 450 Ib/min. The separator has two outlet streams: the ethanol product outlet stream (99% ethanol) and a residual water stream. 30% of the feedstock is bypassed and mixed with the residual water stream leaving the separator. If 60% of the ethanol entering the separator is recovered in the product stream, determine the composition of the residual water stream just after leaving the separator and the composition of the residual stream after mixing with the bypass stream.arrow_forwardethylene oxide is produced by oxidation of ethylene 100 kmol of ethylene and 100 kmol of o2 are charged to a reactor.the % conversion of ethylene is 85 and%yield of ethylene oxide is 94.12 .calculate the composition of product stream leaving the reactor .the reactions takes place arearrow_forwardFeed gas containing of 78.5mol % H₂, 21% of N₂ & 0.5% of Ar is mixed with recycle gas and enters a reactor where 15% N₂ is converted to NH3 as per the reaction. Ammonia from the exit of the reactor is completely separated from unconverted gases. To avoid the buildup of inerts, a small fraction (5%) of the unreacted gases purged and the balance recycled. USING ASPEN/HYSYS Draw the process flow sheet Product rate and Purge rate Basis:100mol/hrarrow_forward
- - = T C 54°F Humid % Explanation O CHEMICAL REACTIONS Percent yield of chemical reactions 2 W |HC|HL| (@F11218HCHIHUSI https://www-awu.aleks.com/alekscgi/x/Isl.exe/10_u-IgNslkr7j8P3jH-IBMBkpcnaFu0F7Uj... FAX S Check بهر 3 * E $ X Gaseous ethane (CH₂CH3) reacts with gaseous oxygen gas (O₂) to produce gaseous carbon dioxide (CO₂) and gaseous water (H₂O). If 0.469 g of water is produced from the reaction of 0.90 g of ethane and 5.2 g of oxygen gas, calculate the percent yield of water. Round your answer to 2 significant figures. 4 0 d R DE C Ś 96 % 5 T 6 G H "4+ & 7 B N U 144 * 00 C | 141 | a A 8 1 D-II ( 9 r K M || a ✓ DDI O s ) B Ⓒ2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | | O F ▬▬▬▬▬▬ fo 1 11² a P MC - MC insert C + 3/5 = prt sc } ♡ 4 Privacy Center Privacy Center | Acces 1 0 Chisom Seve E d 11:10 10/22/20arrow_forwardThe production of cement results in significant GHG emissions. The emissions are caused by the decomposition of calcium carbonate (limestone) to calcium oxide (lime) and carbon dioxide. This process known as calcination, is carried out in kilns. Therefore, additional GHGs are emitted as a result of heating the kilns. A large cement manufacturer uses a mix of coal, natural gas, and biomass in its boiler. This mix produces the following amounts of GHGs per Million BTUs of energy: • 250 lbs CO2/MMBtu • 5 lbs CH4/MMBtu • 0.36 lbs N2O/MMBtu Process carbon emissions from the calcination process produce 925 lbs of carbon dioxide per metric ton of cement. In addition, the plant uses 1.7 Million BTUs of energy per metric ton of cement. If the plant produces 180,000 metric tons of cement annually, how many metric tons of CO2e does it emit?arrow_forwardMethanol (CH3OH) is produced in the reaction of carbon dioxide and hydrogen via the reaction and process given below: CO2+3H2→CH3OH+H2O The fresh feed to the process contains hydrogen, carbon dioxide, and 0.400 mol% inerts (I). The reactor effluent passes through a condenser that removes essentially all of the methanol and water formed and none of the reactants or inerts. The unreacted reactants and inerts are recycled to the reactor. To avoid buildup of the inerts in the system, a purge stream is withdrawn from the recycle. 100 mol/h of feed to the reactor (not the fresh feed to the process) contains 29.5 mol% CO2, 68.0 mol% H2, and 2.50 mol% inerts. The single-pass conversion of hydrogen is 46.71%. What is the recycle-to-fresh feed ratio?arrow_forward
- The oxidation of ethylene to produce ethylene oxide proceeds according to the equation2C 2H 4 +O 2 ! 2C 2H 4OThe feed to a reactor contains 100 kmol C 2H 4 and 100 kmol O 2. If the reaction proceeds to completion, how much of the excess reactant will be left; how much C 2H 4O will be formed; and what is the extent of reaction?arrow_forwardHematite is an iron ore with the following composition: Fe2O3 [MW=159.70]. To make steel, carbon [At. Wt. = 12.01] in the form of coke is used to reduce Fe2O3 to iron metal [At. Wt. = 55.85] as shown below: 3 C + 2 Fe2O3 → 3 CO2 + 4 Fe How many grams of carbon are needed to produce 2,500 grams of iron? Relative to the problem the processing of 798.5 g of hematite ore produced 508.2 g of iron metal. Determine the percent yield of pure iron for this batch?arrow_forward.edu bryuitra/courses roee_re Question Completion Status: The following process describes the complete combustion reaction of Toluene fuel C7HB (SG 0.866) with air. Liquid fuel (n1) enters the combustion chamber at 200mL/min, where it evaporated into a stream of air (n2) 15% in excess. Complete combustion reaction in which only fraction of the fuel is burned. The combustion products go to a condenser, where the unreacted fuel and water are liquified. The mass flowrate of the fuel in this steam (ma) is 50. The uncondensed gas leaves the condenser at 60 C and 0.5 atm gauge pressure. Calculate the fractional conversion of the fuel Calculate the SCMH of the air entering the combustion chamber Calculate the volumetric flow rate of the gas leaving the condenser (m³/min). a. b. C. ri4 CO2 n3 Fuel ris N2 Fuel ri1 ni4 CO2 nig O2 Combustion nig N2 Condenser air riz Chamber ng O2 ni, H,0 n3 Fuel niz H,0arrow_forward
- The oxidation of ethylene to produce ethylene oxide proceeds according to the equation2C 2H 4 +O 2 ! 2C 2H 4OThe feed to a reactor contains 100 kmol C 2H 4 and 100 kmol O 2. Which reactant is limiting?arrow_forward3. Suppose you are to measure the BOD removal rate for a primary wastewater treatment plant. You take 2 samples of raw sewage on its way into the plant and two samples of the effluent leaving the plant. Standard 5-day BOD tests are run on the four samples, with no seeding producing the following data: Sample Dilution Doi (mg/L) DOf (mg/L) Source 1 Raw 1:30 9.0 2.2 2 Raw 1:20 9.0 ? 3 Treated 1:15 8.5 1.5 4 Treated ? 9.0 1 a. Find the BOD5 for the raw and treated sewage, and the percent removal of BOD in the treatment plant. b. Find the DO that would be expected in sample 2 at the end of the test. c. What would be the maximum volume of treated sewage for sample 4 that could be put into the 300 mL BOD bottle and still have the DO after 5 days remain above zero?arrow_forwardSolid calcium (CaF2) reacts with sulfuric acid to form solid calcium sulfate and gaseous hydrogen fluoride. The HF is then dissolved in water to form hydrofluoric acid. A source of calcium fluoride is fluorite ore containing 96 wt% CaF2 and 4% SiO2. In a typical hydrofluoric acid manufacturing process, fluorite ore is reacted with 93 wt% aqueous sulfuric acid, supplied 15% in excess of the stoichiometric amount. Ninety-five percent of the ore dissolves in the acid. Some of the HF formed reacts with the dissolved silica in the reaction 6 HF + SIO2 (aq) – H2SİF6 (9) + 2 H2O (1) The hydrogen fluoride exiting from the reactor is subsequently dissolved in enough water to produce 60 wt% hydrofluoric acid. Calculate the quantity of fluorite ore needed to produce a metric ton of acid. Express your answer into three significant figures.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
