
Concept explainers
Ethylbenzene is converted to styrene in the catalytic dehydrogenation reaction C«H10(g) -
A flowchart of a simplified version of the commercial process is shown here.
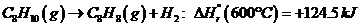
Fresh and recycled liquid ethylbenzene combine and are heated from 25°C to 500°C (A). and the heated ethylbenzene is mixed adiabatically with steam at 700°C (B) to produce the feed to the reactor at 600°C. (The steam suppresses undesired side reactions and removes carbon deposited on the catalyst surface.) A once-through conversion of 35% is achieved in the reactor (C), and the products emerge at 560°C. The product stream is cooled to 25°C (D), condensing essentially all of the water, ethylbenzene, and styrene and allowing hydrogen to pass out as a recoverable by-product of the process.
The water and hydrocarbon liquids are immiscible and are separated in a settling tank decanter (£). The water is vaporized and healed (F) to produce the steam that mixes with the ethylbenzene feed to the reactor. The hydrocarbon stream leaving the decanter is fed to a distillation tower(C) (actually, a series of towers), which separates the mixture into essentially pure styrene and ethylbenzene, each at 25°C after cooling and condensation steps have been carried out. The ethylbenzene is recycled to the reactor preheater, and the styrene is taken off as a product.
- On a basis of 100 kg/h styrene produced, calculate the required fresh ethylbenzene feed rale, the flow rate of recycled ethylbenzene, and the circulation rate of water, all in mol/h. (Assume P = 1 atm.)
- Calculate the required rates of heat input or withdrawal in kJ/h for the ethylbenzene preheater (A), steam generator (F), and reactor (C).
Physical Property Data
Learn your wayIncludes step-by-step video

Chapter 9 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Process Dynamics and Control, 4e
Digital Fundamentals (11th Edition)
Foundation Design: Principles and Practices (3rd Edition)
Experiencing MIS
Structural Analysis (10th Edition)
- a) What is the standard enthalpy of reaction per mole of either HCl or NaOH when 50.00 cm3 of 0.1 moldm-3 of hydrochloric acid (HCl) and 50.00 cm3 of 0.1 moldm-3 of sodium hydroxide (NaOH) are mixed? According to your simulation, the temperature rose by 0.68 oC. (Density of water = 1gcm-3, Specific heat capacity of water = 4.18 J g-1 oC-1) Show your working below:arrow_forwardConstruct enthalpy cycles; use Hess's law and the following data to calculate the enthalpy of formation of ethane (from carbon and hydrogen gas). Cis) + Ozig) + CO2(g) AHa = -394kJmol H2g) + %02(a) – H20 m AH°. = -286kJmol1 + 3%O2(g) → 2002(9) + 3H20 m AH = -1560kJmol1arrow_forwarda) What is the standard enthalpy of reaction per mole of either HCl or NaOH when 50.00 cm3 of 0.1 moldm-3 of hydrochloric acid (HCl) and 50.00 cm3 of 0.1 moldm-3 of sodium hydroxide (NaOH) are mixed? According to your simulation, the temperature rose by 0.68 oC. (Density of water = 1gcm-3, Specific heat capacity of water = 4.18 J g-1 oC-1) b) What is the standard enthalpy of reaction per mole of KOH when 50.00 cm3 of 1.0 moldm-3 of Nitric Acid (HNO3) and 50.00 cm3 of 1.0 moldm-3 of Potassium Hydroxide (KOH) are mixed? According to your simulation, the temperature rose by __6.81__ oC. (Density of water = 1gcm-3, Specific heat capacity of water = 4.18 J g-1 oC-1)arrow_forward
- Calculate the standard enthalpy change for the reaction 2C8H18() + 1702(g) → 16CO(g) + 18H2O(). Given 2C3H15(1)+ 2502(g) →16CO2(g) + 18H2O(I) AH°=-11,020 kJ/mol 2Cog) + O2(g) → 2CO2(g) AH° =-566.0 kJ/mol Multiple Choice 10,450 kJ/mol -10.450 kJ/mol -6,492 kJ/mol 6,492 kJ/mol 15,550 kJ/molarrow_forwardGiven the standard enthalpy changes for the following two reactions:(1) 2C(s) + 2H2(g)C2H4(g)...... ΔH° = 52.3 kJ(2) 2C(s) + 3H2(g)C2H6(g)......ΔH° = -84.7 kJwhat is the standard enthalpy change for the reaction:(3) C2H4(g) + H2(g)C2H6(g)......ΔH° = ?______ kJarrow_forwardThe specific heat of compound AB(s) was determined using coffee-cup calorimeter. When 1.750 g of AB(s) was mixed with 15.00 mL deionized distilled water at room temperature, only 0.850 grams of the compound was dissolved. The temperature of the heterogenous mixture was decreased by 1.70K.Prior to this, the calorimeter was calibrated using a 15mL aqueous reaction mixture that initially contains 0.070 moles each of HBr and KOH. The recorded ΔT is +5.75K. Note: H+(aq) + OH -(aq) → H2O(l) ΔH = -55.85 kJ/molAB(s) ⇌ A+(aq) + B-(aq) ΔH = 88.75 kJ/molspecific heat (H2O) = 4.184 J/g°CMM of AB = 65 g/mol What is the specific heat of solid AB compound (in J/g°C)?arrow_forward
- Calculate the standard enthalpy change (in kJ/mol) for the reaction 2C3H18(1) + 2102(g) – 8CO(g) + 8CO2(g) + 18H20(1). Given that: 2C3H18(1) + 2502(9) → 16CO2(g) + 18H20(1) AH° = -11,020 kJ/mol 2Co(g) + O2(g) → 2CO2(g) AH° = -566.0 kJ/mol Answer in 4 significant figures. Exclude the unit/sarrow_forwardSuppose that 125 mL of 0.1125 M HCI is reacted with 155 mL of 0.1010 M NaOH in a coffee-cup calorimeter. HCl(aq) + NaOH H20(1) + NaCl(aq) -56 KJ mol ΔΗ If the initial temperature of the mixture is 22.1°C, calculate the final temperature of the mixture given the following information. Ssolution = 4.1 g.°C %3D Dsolution = 1.02 mL 25.5 °C 24.3 °C 22.8 °C O 19.8 °Carrow_forwarda) What is the standard enthalpy of reaction per mole of either HCI or NaOH when 100.00 cm³ of 0.1 moldm-³ of hydrochloric acid (HCI) and 100.00 cm³ of 0.1 moldm-3 of sodium hydroxide (NaOH) are mixed? According to your simulation, the temperature rose by 0.68 Specific heat capacity of water = 4.18 Jg-¹ °C-1) Show your working below: HCI (ag) + NaOH(aq) → NaCl(aq) + H₂Om °C. (Density of water = 1gcm-³, Iarrow_forward
- 4.38 g of a gas (28.1 g mol–1) was dissolved in liquid water in a calorimeter with a calorimeter constant of 76.0 J K–1. Prior to mixing everything was equilibrated to 21.2 °C. The resulting solution with a mass of 23.4 g was observed to be at a temperature of -6.8 °C and have a specific heat capacity of 4.61 J g–1 K–1. Determine q per mole for this dissolution process. a. -5150 J/mol b. -1.94 × 104 J/mol c. 2.89 × 105 J/mol d. -3.39 × 105 J/mol e. 3.30 × 104 J/molarrow_forwardEthane is flamed off in abundance from oil wells, because it is unreactive and difficult to use commercially, but would it make a good fuel? The standard enthalpy of reaction for 2 C2H6(g) + 7 O2(g) → 4 CO2(g) + 6 H2O(1) is-3120 kJ mol-1. (a) What is the standard enthalpy of combustion of ethane? (b) What is the specific enthalpy of combustion of ethane? (c) Is ethane a more or less efficient fuel than methane?arrow_forwardUsing the standard formation enthalpies that follow, calculate the standard enthalpy change for this reaction. CO₂(g) + H₂(g) →→→ CO(g) + H₂O(g) A,H° = kJ Species AfH° (kJ/mol) CO₂(g) CO(g) H₂O(g) -393.5 -110.5 -241.8 Submit The enthalpy change for the reaction is given by, AH° => AH° (products) - A#° (reactants) What is the sum of the formation enthalpies of the products? kJ/molarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





