
A bituminous coal is burned with air in a boiler furnace. The coal is fed at a rate of 40,000 kg/h and has an ultimate analysis of 76wt% C, 5% H. 8% O, negligible amounts of N
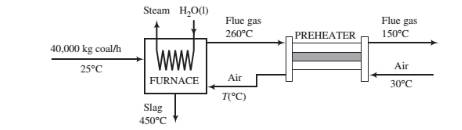
- Prove that the air-to-fuel ratio is 16.1 standard cubic meters/kg coal and that the flue gas contains 4.6% H2O by volume.
- Calculate the rate of cooling required to cool the flue gas from 260°C to 150°C and the temperature to which the air is preheated. {Note: A trial-and-error calculation is required.)
- If 60% of the heat transferred from the furnace goes into producing saturated steam at 30 bar from liquid boiler feedwater at 50°C, at what rate (kg/h) is steam generated?
Trending nowThis is a popular solution!

Chapter 9 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Structural Analysis (10th Edition)
Starting Out with Python (3rd Edition)
Software Engineering (10th Edition)
Web Development and Design Foundations with HTML5 (9th Edition) (What's New in Computer Science)
- consider the balance to reaction 6 A + 9 B -> 3C + 5D If 6.448 moles of A (MW = 39.34) are consumed, how many moles of B (MW = 42.66) are also consumed?arrow_forwardConstruct enthalpy cycles; use Hess's law and the following data to calculate the enthalpy of formation of ethane (from carbon and hydrogen gas). Cis) + Ozig) + CO2(g) AHa = -394kJmol H2g) + %02(a) – H20 m AH°. = -286kJmol1 + 3%O2(g) → 2002(9) + 3H20 m AH = -1560kJmol1arrow_forwardFor the reaction bellow: C„H(2n+2) + a(02 + 3.76 N2) → nC02 + (n+ 1)H,0 + 3.76a N2 If the heat of combustion writing as: Qc = 618.49 n+ 187. 83 And the mole fraction of CO, in products is Xco, 1. Type of fuel. 2. Heat of combustion (KJ/mole of fuel) 25 find: 227 3. If n=4, find the mole fractions of Co, H,0 and N2arrow_forward
- For the reaction bellow: CnH(2n+2) + a(O₂ +3.76 N₂) → nCO₂ + (n + 1)H₂0 +3.76a N₂ If the heat of combustion writing as: Qc = 618.49 n + 187.83 (KJ/mole of fuel) 25 And the mole fraction of CO₂ in products is Xco₂ 227 1. Type of fuel. 2. Heat of combustion 3. If n=3, find the mole fractions of CO2, H₂O and N₂ 3 find:arrow_forwardWhat is the standard enthalpy of reaction per mole of KOH when 50.00 cm3 of 1.0 moldm-3 of Nitric Acid (HNO3) and 50.00 cm3 of 1.0 moldm-3 of Potassium Hydroxide (KOH) are mixed? According to your simulation, the temperature rose by __6.81__ oC. (Density of water = 1gcm-3, Specific heat capacity of water = 4.18 J g-1 oC-1) Show your working below: HNO3 (aq) + KOH (aq) à KNO3 + H2Oarrow_forwardWhat is the standard enthalpy of reaction per mole of either HCl or NaOH when 50.00 cm3 of 0.1 moldm-3 of hydrochloric acid (HCl) and 50.00 cm3 of 0.1 moldm-3 of sodium hydroxide (NaOH) are mixed? According to your simulation, the temperature rose by ___0.68__ oC. (Density of water = 1gcm-3, Specific heat capacity of water = 4.18 J g-1 oC-1) Show your working below: HCl (aq) + NaOH(aq) à NaCl(aq) + H2O(l)arrow_forward
- Carbon dioxide from the atmosphere "weathers," or dissolves, limestone (CACO3) by the reaction C&CO3 (s) + CO2 (g) + H,0(1) → Ca+ (aq) +2HCO3 (ag) Obtain AH° for this reaction using the following data: AH;(CACO, (s))=-1206.9 kJ/mol %3D AH;(CO2 (9)) = -393.5 kJ/mol AH;(H,0(1)) = -285.8 kJ/mol %3D AH;(Ca+ (ag)) = -542.8 kJ/mol %3D AH (HCO, (ag)) = -692.0 kJ/mol AH = kJarrow_forwardThe oxidation of ethylene to produce ethylene oxide proceeds according to the equation2C 2H 4 +O 2 ! 2C 2H 4OThe feed to a reactor contains 100 kmol C 2H 4 and 100 kmol O 2. Which reactant is limiting?arrow_forwardGasoline having a chemical formula C3H17 is burned in an engine at a fuel-air equivalence ratio ø=1.19 and a temperature of 1000K. a) Write the stoichiometric combustion equation of the fuel; b) Write the reaction equation corresponding to the given fuel-air equivalence ratio.arrow_forward
- A cylinder of compressed neon gas is opened to the atmosphere in a storage closed where P closet = 1010, 066 Pa, the gas then fills the closet. Volume of the gas Ne(g) cylinder = 0.7 m^3 and closrt is 1 m. Calculate the work usd in KJarrow_forwarda) What is the standard enthalpy of reaction per mole of either HCl or NaOH when 50.00 cm3 of 0.1 moldm-3 of hydrochloric acid (HCl) and 50.00 cm3 of 0.1 moldm-3 of sodium hydroxide (NaOH) are mixed? According to your simulation, the temperature rose by 0.68 oC. (Density of water = 1gcm-3, Specific heat capacity of water = 4.18 J g-1 oC-1) b) What is the standard enthalpy of reaction per mole of KOH when 50.00 cm3 of 1.0 moldm-3 of Nitric Acid (HNO3) and 50.00 cm3 of 1.0 moldm-3 of Potassium Hydroxide (KOH) are mixed? According to your simulation, the temperature rose by __6.81__ oC. (Density of water = 1gcm-3, Specific heat capacity of water = 4.18 J g-1 oC-1)arrow_forwardnitrogen and oxygen gases. Calculate the enthalpy of the reaction if the process occurs in the dioxide (NO2) gas Write the balanced equation for the production of the brown nitrogen from 1. sequence of reactions given below: AH=180 kJ AH = 112 kJ N,(g) + 0,(8) %3D → 2NO(g) 2NO,(g) %3D → 2NO(g) + 0,(g) 2. Methylhydrazine fiels Iook for thearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





