
Chemistry & Chemical Reactivity
10th Edition
ISBN: 9781337399074
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 76GQ
Vanillin is the flavoring agent in vanilla extract and in vanilla ice cream. Its structure is shown here:
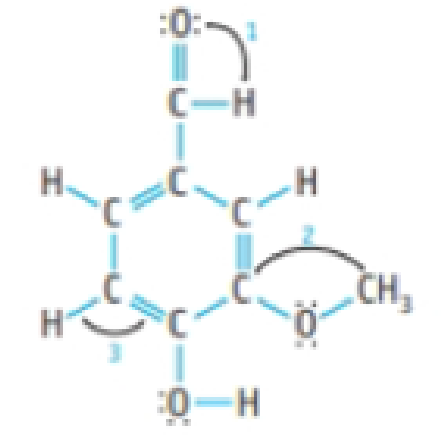
- (a) Give values for the three bond angles indicated.
- (b) Indicate the shortest carbon-oxygen bond in the molecule.
- (c) Indicate the most polar bond in the molecule.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
For hexane, what is the bond length? Are all C—C bonds the same length?
The electron geometry of H2S is ____?
Identify which of these Lewis structures (A, B, C or D) is correct for NOF and what is the VSEPR shape of the compound?
Chapter 8 Solutions
Chemistry & Chemical Reactivity
Ch. 8.2 - Draw Lewis electron dot structures for CH3Cl...Ch. 8.2 - Prob. 8.2CYUCh. 8.2 - Prob. 8.3CYUCh. 8.2 - Prob. 8.4CYUCh. 8.3 - Prob. 8.5CYUCh. 8.4 - Draw resonance structures for the bicarbonate ion,...Ch. 8.5 - Sketch the Lewis structures for CIF2+ and CIF2....Ch. 8.6 - What is the shape of the dichloromethane (CH2C12)...Ch. 8.6 - Give the electron-pair geometry and molecular...Ch. 8.6 - Draw the Lewis structure for lCl2, and then decide...
Ch. 8.7 - For each of the following pairs of bonds, decide...Ch. 8.7 - Draw the resonance structures for SCN. What are...Ch. 8.8 - For each of the following molecules, decide...Ch. 8.8 - The electrostatic potential surface for SOCl2 is...Ch. 8.9 - Using the bond dissociation enthalpies in Table...Ch. 8.10 - Prob. 1.1ACPCh. 8.10 - Do any of the atoms in an ibuprofen molecule have...Ch. 8.10 - What is the most polar bond in the molecule?
Ch. 8.10 - Prob. 1.4ACPCh. 8.10 - Prob. 1.5ACPCh. 8.10 - Prob. 1.6ACPCh. 8.10 - Are there any 120° bond angles in ibuprofen? Any...Ch. 8.10 - Prob. 1.8ACPCh. 8.10 - Prob. 2.2ACPCh. 8.10 - Calculate the difference in electronegativity...Ch. 8.10 - Predict the bond dissociation enthalpy for a...Ch. 8.10 - Prob. 3.3ACPCh. 8 - Give the periodic group number and number of...Ch. 8 - Give the periodic group number and number of...Ch. 8 - For elements in Groups 4A-7A of the periodic...Ch. 8 - Prob. 4PSCh. 8 - Draw a Lewis structure for each of the following...Ch. 8 - Draw a Lewis structure for each of the following...Ch. 8 - Draw a Lewis structure for each of the following...Ch. 8 - Draw a Lewis structure for each of the following...Ch. 8 - Show all possible resonance structures for each of...Ch. 8 - Show all possible resonance structures for each of...Ch. 8 - Prob. 11PSCh. 8 - Draw a Lewis structure for each of the following...Ch. 8 - Determine the formal charge on each atom in the...Ch. 8 - Determine the formal charge on each atom in the...Ch. 8 - Determine the formal charge on each atom in the...Ch. 8 - Determine the formal charge on each atom in the...Ch. 8 - Draw a Lewis structure for each of the following...Ch. 8 - Prob. 18PSCh. 8 - Prob. 19PSCh. 8 - The following molecules or ions all have three...Ch. 8 - Draw a Lewis structure for each of the following...Ch. 8 - Draw a Lewis structure for each of the following...Ch. 8 - Give approximate values for the indicated bond...Ch. 8 - Give approximate values for the indicated bond...Ch. 8 - Phenylalanine is one of the natural amino acids...Ch. 8 - Acetylacetone has the structure shown here....Ch. 8 - For each pair of bonds, indicate the more polar...Ch. 8 - For each of the bonds listed below, tell which...Ch. 8 - Urea, (NH2)2CO, is used in plastics and...Ch. 8 - Considering both formal charges and bond...Ch. 8 - Considering both formal charge and bond...Ch. 8 - Three resonance structures are possible for...Ch. 8 - Three resonance structures are possible for the...Ch. 8 - Compare the electron dot structures of the...Ch. 8 - Compare the electron dot structures of the...Ch. 8 - The chemistry of the nitrite ion and HNO2: (a) Two...Ch. 8 - Draw the resonance structures for the formate ion,...Ch. 8 - Prob. 39PSCh. 8 - Consider the following molecules: (a) CH4 (b)...Ch. 8 - Which of the following molecules is(are) polar?...Ch. 8 - Prob. 42PSCh. 8 - Give the bond order for each bond in the following...Ch. 8 - Prob. 44PSCh. 8 - In each pair of bonds, predict which is shorter....Ch. 8 - In each pair of bonds, predict which is shorter....Ch. 8 - Prob. 47PSCh. 8 - Compare the carbon-oxygen bond lengths in the...Ch. 8 - Consider the carbon-oxygen bond in formaldehyde...Ch. 8 - Compare the nitrogen-nitrogen bond in hydrazine,...Ch. 8 - Ethanol can be made by the reaction of ethylene...Ch. 8 - Methanol can be made by partial oxidation of...Ch. 8 - Hydrogenation reactions, which involve the...Ch. 8 - Phosgene, Cl2CO, is a highly toxic gas that was...Ch. 8 - The compound oxygen difluoride is quite reactive,...Ch. 8 - Oxygen atoms can combine with ozone to form...Ch. 8 - Prob. 57GQCh. 8 - Prob. 58GQCh. 8 - Which of the following compounds or ions do not...Ch. 8 - Prob. 60GQCh. 8 - Draw resonance structures for the formate ion,...Ch. 8 - Prob. 62GQCh. 8 - Prob. 63GQCh. 8 - What is the principle of electroneutrality? Use...Ch. 8 - Prob. 65GQCh. 8 - Draw resonance structures for the SO2 molecule,...Ch. 8 - What are the orders of the NO bonds in NO2 and...Ch. 8 - Which has the greater ONO bond angle, NO2 or NO2+?...Ch. 8 - Compare the FClF angles in CIF2+ and ClF2. Using...Ch. 8 - Draw an electron dot structure for the cyanide...Ch. 8 - Draw the electron dot structure for the sulfite...Ch. 8 - Dinitrogen monoxide, N2O, can decompose to...Ch. 8 - The equation for the combustion of gaseous...Ch. 8 - The cyanate ion, OCN, has the least...Ch. 8 - Vanillin is the flavoring agent in vanilla extract...Ch. 8 - Explain why (a) XeF2 has a linear molecular...Ch. 8 - The formula for nitryl chloride is ClNO2 (in which...Ch. 8 - Hydroxyproline is a less-common amino acid. (a)...Ch. 8 - Amides are an important class of organic...Ch. 8 - Prob. 81GQCh. 8 - The molecule shown here. 2-furylmelhanethiol, is...Ch. 8 - Dihydroxyacetone is a component of quick-tanning...Ch. 8 - It is possible to draw three resonance structures...Ch. 8 - Acrolein is used to make plastics. Suppose this...Ch. 8 - Molecules in space: (a) In addition to molecules...Ch. 8 - 1,2-Dichloroethylene can be synthesized by adding...Ch. 8 - The molecule pictured below is epinephrine, a...Ch. 8 - You are doing an experiment in the laboratory and...Ch. 8 - Prob. 90ILCh. 8 - A paper published in the research Journal Science...Ch. 8 - Uracil is one of the bases in RNA, a close...Ch. 8 - Guanine is present in both DNA and RNA. (a) What...Ch. 8 - Prob. 94ILCh. 8 - Prob. 95SCQCh. 8 - Prob. 96SCQCh. 8 - Bromine-containing species play a role in...Ch. 8 - Acrylamide, H2C=CHCONH2, is a known neurotoxin and...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What aspect of the following Lewis structure indicates that the concept of coordinate covalency is needed to explain the bonding in the molecule?arrow_forwardDefine the term coordinate covalent bond?arrow_forwardFrom this graph, what are the bond length and bond strengthin the H2 molecule?arrow_forward
- For the molecule GaI3, give the lewis structure, the valence shell electron pairs, the bonding electron pairs, the nonbonding electron pairs, the VSEPR formula, bond angle, and the molecular geometry.arrow_forwardWhat's the written lewis structure for C2H5O2N and how many valence electrons?arrow_forwardWhat is the formal charge on the central chlorine atom in the molecular ion [ClO4]- ? Assume that all of the Cl-O bonds are single bonds.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY