
Concept explainers
(a)
Interpretation:
The most likely charge for ion formed from
Concept Introduction:
The fundamental principles that are followed to write an electronic configuration include three rules as follows:
Electron in a
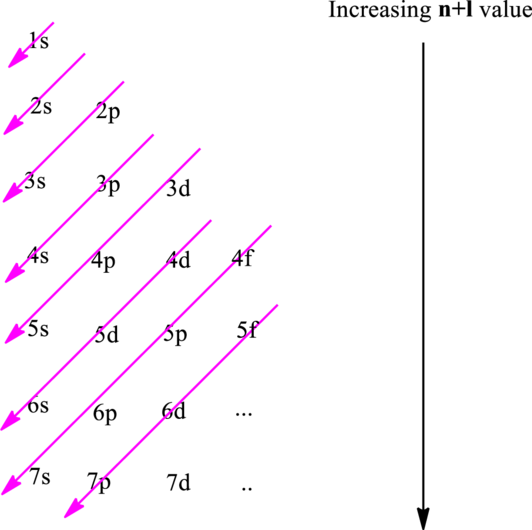
Hund’s rule suggests electrons are not allowed to be paired up until each degenerate set of orbital has got at least one electron.
Pauli Exclusion Principle states two electrons within the same orbital cannot possess same set for four possible quantum numbers.
In
The convention followed to remove or add electrons is electrons of largest principal quantum number are lost first. In case of subshells of the same
(b)
Interpretation:
The most likely charge for ion formed from
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The most likely charge for ion formed from
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The most likely charge for ion formed from
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The most likely charge for ion formed from
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- 1) How much energy (in MJ) is required to ionize 21.14 moles of hydrogen atoms?arrow_forwardThe dipole moment of chlorine monofluoride, ClF(g), is 0.88D. The bond length of the molecule is 1.63 Å. (a) Which atomis expected to have the partial negative charge? (b) What is thecharge on that atom in units of e?arrow_forward3 What is the biggest difference between the Lewis and the AO-model in terms of the localisation of electrons in atoms?arrow_forward
- What is the relationship between the tendency of a main-group element to form a monatomic ion and its position in the periodic table? In what part of the table are the main-group ele-ments that typically form cations? Anions?arrow_forwardThe first three ionization energies of an element X are 900, 1745, and 14912 kJ·mol–1. What is the most likely formula for the stable ion of X?arrow_forwardTo which group of the periodic table does the general valence electron configuration ns2np4 correspond?arrow_forward
- Explain the horizontal irregularity in size of the most com-mon ions of Period 3 elements.arrow_forwardConsider the forces that keep a Z>1 atom together. a. What are these forces? Describe the subatomic particles each force acts on and how the range over which these forces act result in the sub-atomic structure. b. Plot the potential energy as function of distance from these forces acting on two protons as a function of distance.arrow_forwardConsider the A2X4 molecule depicted here, where A and Xare elements. The A¬A bond length in this molecule is d1,and the four A¬X bond lengths are each d2. (a) In terms ofd1 and d2, how could you define the bonding atomic radii ofatoms A and X? (b) In terms of d1 and d2, what would you predictfor the X¬X bond length of an X2 molecule?arrow_forward
- Predict the chemical formulas of the compounds formed bythe following pairs of ions: (a) Fe3+ and O2- , (b) NH4+ and PO43-(c) Ag and N , (d) Ca and Br, (e) Sr and Clarrow_forwardConsider the Mg2+, Cl-, K+, and Se2- ions. The four spheresbelow represent these four ions, scaled according to ionic size.(a) Without referring to Figure 7.8, match each ion to its appropriatesphere. (b) In terms of size, between which of the sphereswould you find the (i) Ca2+ and (ii) S2- ions?arrow_forward1.) An element X reacts with hydrogen to form a compound of formula HX. In which group in the periodic table is X most likely to be found? a. 1A b. 2A c. 7A d. 8A 2.) Which of the following compounds has the strongest chemical bond? a. salt b. sugar c. water d. ethanolarrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

