
Concept explainers
Interpretation:
The chemical formula and the electronic configuration of iodide ion have to be predicted.
Concept Introduction:
The fundamental principles that are followed to write an electronic configuration include three rules as follows:
Electron in a
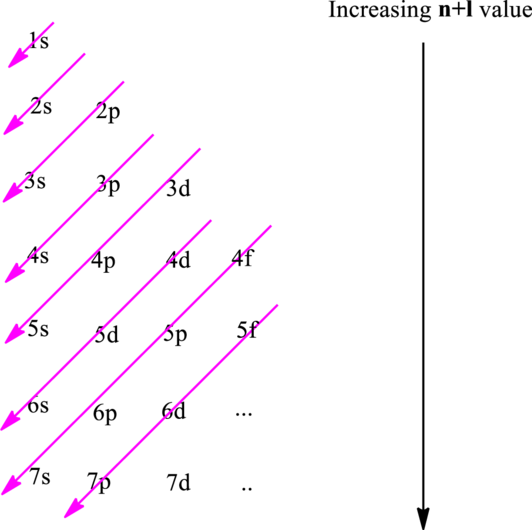
Hund’s rule suggests electrons are not allowed to be paired up until each degenerate set of orbital has got at least one electron.
Pauli Exclusion Principle states two electrons within the same orbital cannot possess same set for four possible quantum numbers.
In
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- Which has the greater bond lengths: NO2 or NO3? Explain.arrow_forwardDraw the Lewis structure of phosphate (PO₄³⁻) by following the octet rule on all atoms and then determine the hybridization of the central atom.arrow_forwardWhat is the state of hybridization of boron and oxygen atoms in boric acid?arrow_forward
- In the formula BH2CI State the hybridization of the boron atom, the polarity and the geometric shape of the moleculearrow_forwardWrite the ground-state electron configuration for each atomand ion pair-Zr, Zr2+, Co, Co2+, Tc, Tc3+, Os, Os4+?arrow_forwardWhat is the molecular and electron configuration for H2Oarrow_forward
- A group of Finnish scientists announced the discovery of the first argon compound, HArF, in 2000. Assuming one of argon's filled 3p orbitals is involved in bond formation, what is the shape of the HArF molecule?arrow_forwardState the polarity, hybridization of the xenon atom and the geometric shape of XeBr4arrow_forwardArrange the following material according to the increasing tendency to lose an electron. (I.) glass, (II.) copper, (III.) fur, (IV.) Silkarrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning



