
Concept explainers
(a)
Interpretation:
The manner resonance phenomenon occurs in acetate ion has to be shown.
Concept Introduction:
When a single Lewis structure cannot describe all the experimentally observed properties of a molecule more than one Lewis structure that differs only in the position of multiple bonds is drawn. These are termed resonance structures. The actual molecule is represented by the hybrid of each of these resonance structures. For example, for a molecule like ozone
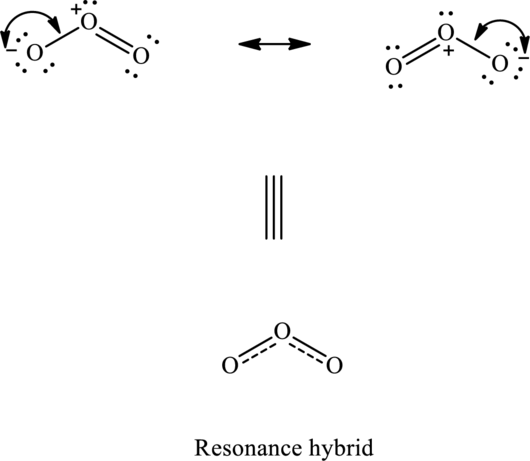
(b)
Interpretation:
The manner resonance phenomenon occurs in enolate ion has to be shown.
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The manner resonance phenomenon occurs in allyl cation has to be shown.
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The manner resonance phenomenon occurs in amidate ion has to be shown.
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- Using the bond dissociation enthalpies in Table 8.8, estimate the enthalpy of combustion of gaseous methane, CH4, to give water vapor and carbon dioxide gas.arrow_forwardDraw Lewis structures for these ions and show which atom in each bears the formal charge. Q.) H3O+arrow_forwardDetermine the number of valence electrons in sulfuric acid (H₂SO₄) and then draw the corresponding Lewis structure (with minimized formal charges).arrow_forward
- We can define average bond enthalpies and bond lengthsfor ionic bonds, just like we have for covalent bonds. Whichionic bond is predicted to have the smaller bond enthalpy,Li—F or Cs—F?arrow_forwardAcetylene 1C2H22 and nitrogen 1N22 both contain a triplebond, but they differ greatly in their chemical properties.(a) Write the Lewis structures for the two substances. (b) Byreferring to Appendix C, look up the enthalpies of formationof acetylene and nitrogen. Which compound is more stable?(c) Write balanced chemical equations for the completeoxidation of N2 to form N2O51g2 and of acetylene to formCO21g2 and H2O1g2. (d) Calculate the enthalpy of oxidationper mole for N2 and for C2H2 (the enthalpy of formationof N2O51g2 is 11.30 kJ>mol). (e) Both N2 and C2H2 possesstriple bonds with quite high bond enthalpies (Table 8.3).Calculate the enthalpy of hydrogenation per mole for bothcompounds: acetylene plus H2 to make methane, CH4;nitrogen plus H2 to make ammonia, NH3.arrow_forwardWrite the Lewis structures for CH2N2, including all resonance forms, and show formal charges.arrow_forward
- 1. Calculate the enthalpy of the following reactionarrow_forwardFor the carbonate ion (CO32-), there are 3 resonance structures. For any given structure what would the formal charge be for an oxygen that has a single bond to the central carbon atom?arrow_forwardIf the dipole moment of a diatomic molecule is found to be 1.04 D, and its bond length is found to be 124 pm, what is the fractional charge on the atoms of the molecule in Coulombs (C)?arrow_forward
- Draw three resonance structures for carbonate ion, CO32-, and assign formal charges on all the atoms.arrow_forwardWhat is the formal charge on carbon in COCl2?arrow_forwardCalculate the following: Standard enthalpy of formation of methane, the enthalpy of atomization of methane and the Bond energy term for the C-H bond.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning




