
(a)
Interpretation:
The formula unit of strontium iodide has to be drawn.
Concept Introduction:
To write formula unit from
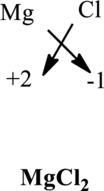
Once the chemical formula is known, each metallic element is suitably accommodated by the non-metallic element. For instance, in magnesium chloride the formula unit is written as follows:
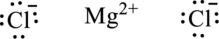
(b)
Interpretation:
The formula unit of potassium phosphide has to be drawn.
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The formula unit of magnesium nitride has to be drawn.
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- If the dipole moment of a diatomic molecule is found to be 1.04 D, and its bond length is found to be 124 pm, what is the fractional charge on the atoms of the molecule in Coulombs (C)?arrow_forwardPhosgene, a substance used in poisonous gas warfare during World War I, is so named because it was first prepared by the action of sunlight on a mixture of carbon monoxide and chlorine gases. Its name comes from the Greek words phos (light) and genes (born of). Phosgene has the following elemental composition: 12.14% C, 16.17% O, and 71.69% Cl by mass. Its molar mass is 98.9 g/mol. (d) Using average bond enthalpies, estimate H for the formation of gaseous phosgene from CO(g) and Cl2(g).arrow_forwardWrite the Lewis structures of both isomers with the formula C2H7N.arrow_forward
- An oxide of nitrogen is 25.9% N by mass, has a molar mass of 108 g/mol, and contains no nitrogen-nitrogen or oxygenoxygen bonds. Draw its Lewis structure, and name it.arrow_forwardWhat is the formal charge on the central chlorine atom in the molecular ion [ClO4]- ? Assume that all of the Cl-O bonds are single bonds.arrow_forwardHow many resonance structures can be drawn for the hydrogen tellurate ion (HTeO4–) in which the central tellurium atom bears a –1 formal charge and the oxygens bear formal charges of either zero or –1? Enter your answer as a whole number.arrow_forward
- For the molecule GaI3, give the lewis structure, the valence shell electron pairs, the bonding electron pairs, the nonbonding electron pairs, the VSEPR formula, bond angle, and the molecular geometry.arrow_forwardWrite a Lewis structure for HC2─ and assign any formal charges to the correct atom.arrow_forwardAluminum oxide (Al₂ O₃) is a widely used industrial abrasive(emery, corundum), for which the specific application depends onthe hardness of the crystal. What does this hardness imply about the magnitude of the lattice energy? Would you have predictedfrom the chemical formula that Al₂ O₃ is hard? Explain.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
