
(a)
Interpretation:
The
Concept Introduction:
The fundamental principles that are followed to write an electronic configuration include three rules as follows:
Electron in a
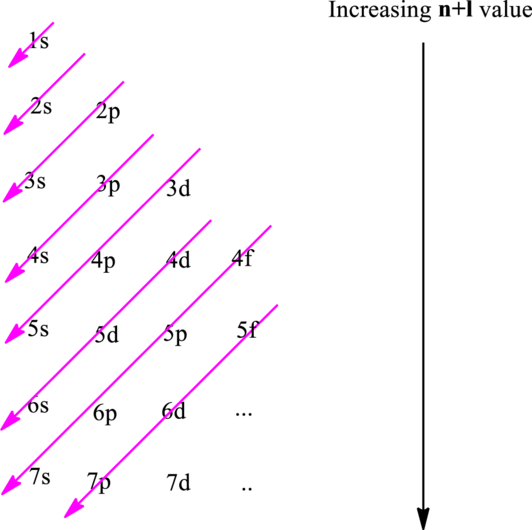
Hund’s rule suggests electrons are not allowed to be paired up until each degenerate set of orbital has got at least one electron.
Pauli Exclusion Principle states two electrons within the same orbital cannot possess same set for four possible quantum numbers. Quantum-mechanical equivalent for this state means that two electrons are not allowed to have the same space simultaneously. Since one electron may certain discrete same set of n, l and m values, however, the fourth spin quantum number should be different. This provides a unique address to every electron. Since the possible magnitudes of spin can be either +1/2 or -1/2 thus at maximum two electrons can occupy any given orbital.
The convention followed to remove or add electrons is electrons of largest principal quantum number are lost first. In case of subshells of the same
(b)
Interpretation:
The
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- Write the ground-state electron configuration for each atomand ion pair-Zr, Zr2+, Co, Co2+, Tc, Tc3+, Os, Os4+?arrow_forwardIs the electron density consistentwith equal contributions from the tworesonance structures for O3? Explain.arrow_forwardGive the ground-state electron configurations and bond orders of(i) Li2, (ii) Be2, and (iii) C2.arrow_forward
- The existence of compounds of the noble gases was once a great surprise and stimulated a great deal of theoretical work. Sketch the molecular orbital energy level diagram for XeF and deduce its ground state electron configurations. Is XeF likely to have a shorter bond length than XeF+?arrow_forwardWhich of the species NP, NP+ and NP2− would be paramagnetic? Briefly explain why (one sentence).arrow_forwardWrite electron configurations for the following ions. Ru3+ As3- Y3+ Pd2+arrow_forward
- Predict the ordering, from shortest to longest, of the bondlengths in CO, CO2, and CO32 - .arrow_forwardHow many valence electron does Br havearrow_forwardThe photoelectron spectrum of HBr has two main groups of peaks. The first has ionization energy 11.88 eV. The next peak has ionization energy 15.2 eV, and it is followed by a long progression of peaks with higher ionization energies. Identify the molecular orbitals corresponding to these two groups of peaks.arrow_forward
- In each of the following molecules, a central atom is surrounded by a total of three atoms or unshared electron pairs: SnCl2, BCl3, SO2. In which of these molecules would you expect the bond angle to be less than 120? Explain your reasoning.arrow_forwardThe B2 molecule is paramagnetic; show how this indicates that the energy ordering of the orbitals in this molecule is given by Figure 6.18a rather than 6.18b.arrow_forwardThe cations O2+ and N2+ are formed when molecules of O2 and N2 are subjected to intense, high-energy solar radiation in Earths upper atmosphere. Write the electron configuration for O2+. Predict its bond order and magnetic behavior.arrow_forward

 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





