
Concept explainers
(a)
Interpretation:
The bond length of
Concept Introduction:
The bond length is estimated to be average of covalent radii of two atoms within a bond. Each ion contributes to the bond length as illustrated as follows:
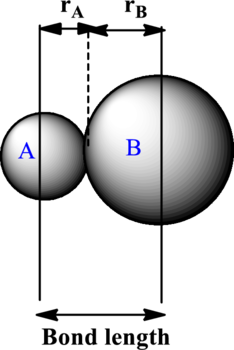
The expression to calculate the bond length is as follows:
Here,
(b)
Interpretation:
The bond length of
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The bond length of nitrogen- nitrogen triple bond in
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- What atomic or hybrid orbitals make up the bond between C and O in carbon dioxide, CO2 ?arrow_forwardDescribe the structure of a P2 molecule in valence-bond terms. Why is P4 a more stable form of molecular phosphorus than P2?arrow_forwardDraw the Lewis dot structure and calculate the formal charges of the oxygen and phosphorus atoms of PO4 3- . What is the molecular geometry of the molecule?arrow_forward
- The cations O2+ and N2+ are formed when molecules of O2 and N2 are subjected to intense, high-energy solar radiation in Earths upper atmosphere. Write the electron configuration for O2+. Predict its bond order and magnetic behavior.arrow_forwardWhat are the hybridizations of nitrogen in NF3 and NH3 respectively?arrow_forwardCompounds such as NaBH4, Al(BH4)3, and LiAlH4 are complexhydrides used as reducing agents in many syntheses.(a) Give the oxidation state of each element in these compounds.(b) Write a Lewis structure for the polyatomic anion in NaBH4, and predict its shape.arrow_forward
- Iodine and oxygen form a complex series of ions, among them IO4^- and IO5^3-. Draw the Lewis structures for these ions, and specify their electron-pair geometries and the shapes of the ions. What is the hybridization of the I atom in these ions?arrow_forwardAccount for the ability of phosphorus to form five bonds, as in PF5.arrow_forwardUse the VSEPR method to predict the structure of the CS2 molecule.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


