
Concept explainers
Interpretation:
Lewis structure, hybridization, and bond angle of
Concept Introduction:
Hybridization is calculated by the hybrid orbitals and to calculate hybrid orbitals we need to know the steric number that is given by,
The table that relates the steric number with hybridization is as follows:
The table that relates steric number with geometry and bond angles is as follows:
Answer to Problem 2F.14E
Hybridization of
Explanation of Solution
The molecule
The symbol for xenon is
The symbol for oxygen is
Thus total valence electrons are sum of the valence electrons for each atom in
The skeleton structure
To complete the octet of xenon it forms a double bond with three oxygen atom.
Hence, 14 electrons are allocated as 2 lone pairs on each oxygen atoms and 1 lone pair on xenon atom to complete their octet. The Lewis structure is as follows:
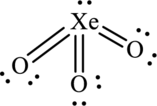
In the structure of
Substitute 3 for number of atoms bonded with central atom and 1 for lone pair on central atom in equation (1) to calculate steric number.
Since, the steric number of the molecule
The molecule
The symbol for xenon is
The symbol for oxygen is
Thus total valence electrons are sum of the valence electrons for each atom in
The skeleton structure
To complete the octet of xenon it forms a double bond with four oxygen atoms.
Hence, 16 electrons are allocated as 2 lone pairs on each oxygen atoms to complete its octet. The Lewis structure is as follows:
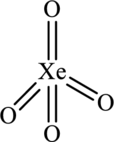
In the structure of
Substitute 4 for atoms bonded with central atom and 0 lone pair on central atom in equation (1) to calculate steric number.
Since, the steric number of the molecule
The molecule
The symbol for xenon is
The symbol for oxygen is
Four negative charges on molecule are added up in the total valence count.
Thus total valence electrons are sum of the valence electrons for each atom in
The skeleton structure
To complete the octet of xenon it forms a double bond with two oxygen atoms.
Hence, 32 electrons are allocated as 2 lone pairs on two doubly bonded oxygen atoms and 3 lone pairs on remaining oxygen atoms to complete their octet. The Lewis structure is as follows:
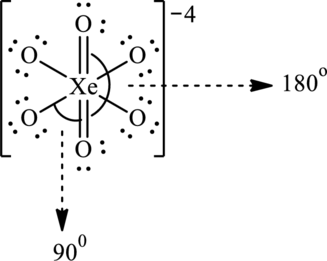
In the structure of
Substitute 6 for number of atoms bonded with central atom and 0 for lone pair on central atom in equation (1) to calculate steric number.
Since the steric number of the molecule
Bond order is the number of electrons involved between the two atoms in a molecule. Bond order is inversely related to bond length.
In
Want to see more full solutions like this?
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- Describe the hybridization around the central atom and the bonding in SCl2 and OCS.arrow_forwardConsider the polyatomic ion IO65-. How many pairs of electrons are around the central iodine atom? What is its hybridization? Describe the geometry of the ion.arrow_forwardThe sulfamate ion, H2NSO3, can be thought of as having been formed from the amide ion, NH2, and sulphur trioxide, SO3. (a) What are the electron-pair and molecular geometries or the amide ion and or SO3? What are the hybridizations of the N and S atoms, respectively? (b) Sketch a structure for the sulfamate ion, and estimate the bond angles. (c) What changes in hybridization do you expect for N and S in the course of the reaction NH2 + SO3 H2NSO3? (d) Is SO3 the donor of an electron pair or the acceptor of an electron pair in the reaction with amide ion? Does the electrostatic potential map shown below confirm your prediction?arrow_forward
- Methylcyanoacrylate is the active ingredient in super glues. Its Lewis structure is (a) How many sigma bonds are in the molecule? (b) How many pi bonds are in the molecule? (c) What is the hybridization of the carbon atom bonded to nitrogen? (d) What is the hybridization of the carbon atom bonded to oxygen? (e) What is the hybridization of the double-bonded oxygen?arrow_forwardAspirin, or acetylsalicylic acid, has the formula C9H8O4 and the skeleton structure (a) Complete the Lewis structure and give the number of bonds and bonds in aspirin. (b) What is the hybridization about the CO2H carbon atom (colored blue)? (c) What is the hybridization about the carbon atom in the benzene-like ring that is bonded to an oxygen atom (colored red)? Also, what is the hybridization of the oxygen atom bonded to this carbon atom?arrow_forwardIt is possible to write a simple Lewis structure for the SO42- ion, involving only single bonds, which follows the octet rule. However, Linus Pauling and others have suggested an alternative structure, involving double bonds, in which the sulfur atom is surrounded by six electron pairs. (a) Draw the two Lewis structures. (b) What geometries are predicted for the two structures? (c) What is the hybridization of sulfur in each case? (d) What are the formal charges of the atoms in the two structures?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning





