
Concept explainers
(a)
Interpretation:
The bond length of
Concept Introduction:
The bond length is estimated to be average of covalent radii of two atoms within a bond. Each ion contributes to the bond length as illustrated below:
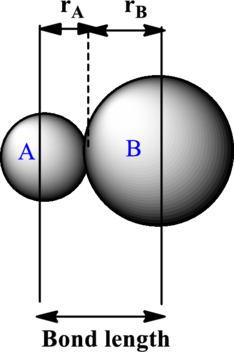
The expression to calculate the bond length is as follows:
Here,
(b)
Interpretation:
The bond length of
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The bond length of
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The bond length of
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- Describe the structure of a P2 molecule in valence-bond terms. Why is P4 a more stable form of molecular phosphorus than P2?arrow_forwardDraw the Lewis structures of cyanate (OCN-) and fulminate (CNO-) ions and calculate their formal charges. Discuss their stability by giving resonance structures.arrow_forwardprovide the following information including the lewis structure and the 3D sketch for CH2Cl2 and SCl2arrow_forward
- Based on average bond enthalpies, would you expect a photon capable ofdissociating a C¬Cl bond to have sufficient energy to dissociate a C¬Br bond?arrow_forwardThe cations O2+ and N2+ are formed when molecules of O2 and N2 are subjected to intense, high-energy solar radiation in Earths upper atmosphere. Write the electron configuration for O2+. Predict its bond order and magnetic behavior.arrow_forwardThe percent ionic character of the bonds in several interhalogen Molecules (as estimated from their measured dipole moments and bond lengths) are ClF (11%), BrF(15%), BrCl (5.6%), ICl(5.8%), and IBr (10%). Estimate the percent ionic characters for each of these molecules, using the equation in Problem 37, and compare them with the given values.arrow_forward
- What are the hybridizations of nitrogen in NF3 and NH3 respectively?arrow_forwardwhat are the bond angles of AsF4Br relative to AsF5? and why? how do the molecule structures differ?arrow_forwardDraw the Lewis structure of phosphate (PO₄³⁻) by following the octet rule on all atoms and then determine the hybridization of the central atom.arrow_forward
- A hypothetical covalent molecule, X–Y, has a dipole moment of 1.12D and a bond length of 101 pm. Calculate the partial charge on a pole of this molecule in terms of e, where e is the charge on an electron.arrow_forwardIn a XeF3Br molecule, why is there a difference in bond length between the Xe-F bond and the Xe-Br bond?arrow_forwardThe existence of compounds of the noble gases was once a great surprise and stimulated a great deal of theoretical work. Sketch the molecular orbital energy level diagram for XeF and deduce its ground-state electron configurations. Is XeF likely to have a shorter or longer bond than XeF+?arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning



