
(a)
Interpretation:
The expected ground-state electron configuration of
Concept Introduction:
The fundamental principles that are followed to write an electronic configuration include three rules as follows:
Electron in a
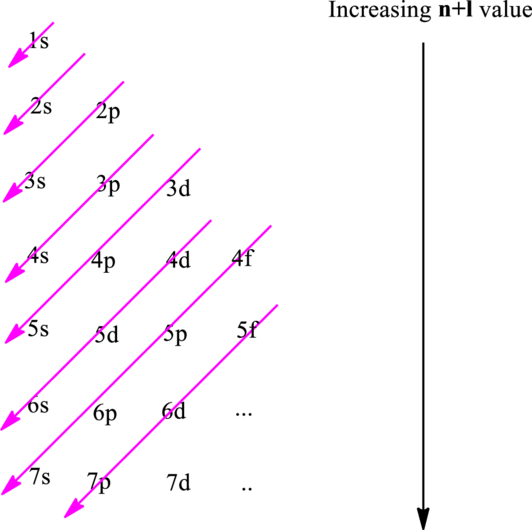
Hund’s rule suggests electrons are not allowed to be paired up until each degenerate set of orbital has got at least one electron.
Pauli Exclusion Principle states two electrons within the same orbital cannot possess same set for four possible quantum numbers.
In
The convention followed to remove or add electrons is electrons of largest principal quantum number are lost first. In case of subshells of the same
(b)
Interpretation:
The expected ground-state electron configuration of
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The expected ground-state electron configuration of
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The ground-state electronic configuration of
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- Draw the Lewis structures of cyanate (OCN-) and fulminate (CNO-) ions and calculate their formal charges. Discuss their stability by giving resonance structures.arrow_forwardWould you expect BrF3 to be as reactive with LiF(s) as it is with CsF(s)? Why (not)?arrow_forwardhow would I identify which of these Lewis structures (A, B, C or D) is correct for PO33- and what is the VSEPR shape of the compound?arrow_forward
- Write the ground-state electron configuration of O2 and calculate the bond order.arrow_forwardDescribe the structure of a P2 molecule in valence-bond terms. Why is P4 a more stable form of molecular phosphorus than P2?arrow_forwardIf the dipole moment of a diatomic molecule is found to be 1.04 D, and its bond length is found to be 124 pm, what is the fractional charge on the atoms of the molecule in Coulombs (C)?arrow_forward
- Write the electron configurations for (a) Ca2+, (b) Co3+, and (c) S2-.arrow_forwardpredict the valence orbital electron molecular orbital configurations for the following, and state whether they vill be stable or unstable ions: d)Si22+ e) P22+ f) S22+ g) F22+ h) Ar22+arrow_forwardGive the ground-state electron configurations of (i) CO, (ii) NO, and (iii) CN−.arrow_forward
- What are the favored geometrical arrangements for ABn molecules for which the A atom has 2, 3, 4, 5, and 6 pairs of electrons in its valence shell?arrow_forwardAlthough the peroxide ion, O22-, and the acetylide ion, C22-, have long been known, the diazenide ion N22- has only been prepared much more recently. By comparison with the other diatomic species, predict the bond order, bond distance, and number of unpaired electrons for N22-.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

