
Concept explainers
(a)
Interpretation:
Bond angle in
Concept Introduction:
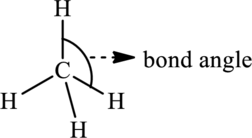
The bond angle is the angle formed between two bonded atoms in a covalent species. It decides the molecular geometry of a molecule. For example in molecule
The table that relates hybridization with % s character is as follows:
Bond angle is directly related to % s character in a molecule.
Hybridization is calculated by the hybrid orbitals and to calculate hybrid orbitals we need to know the steric number that is given by,
The table that relates the steric number with hybridization is as follows:
(b)
Interpretation:
Whether
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- Identify the hybrid orbitals used by boron in BCl3 and in BCl4, the ion formed from the reaction of BCl3 and Cl. Explain your choices.arrow_forwardWhat is the hybridisation of Cl in ClO3-?arrow_forwardWhat atomic or hybrid orbitals make up the bond between C and O in carbon dioxide, CO2 ?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning


