
(a)
Interpretation:
The
Concept Introduction:
The fundamental principles that are followed to write an electronic configuration include three rules as follows:
Electron in a
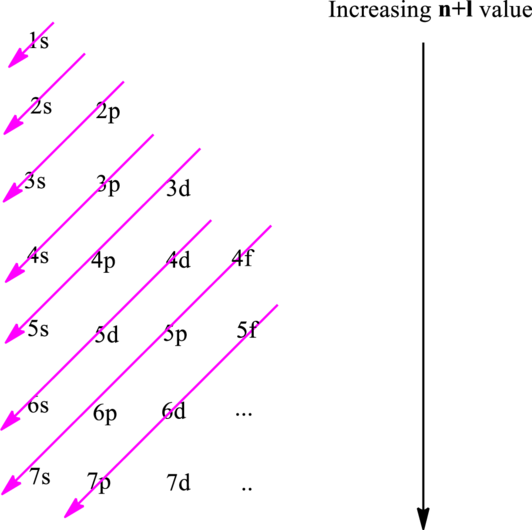
Hund’s rule suggests electrons are not allowed to be paired up until each degenerate set of orbital has got at least one electron.
Pauli Exclusion Principle states two electrons within the same orbital cannot possess same set for four possible quantum numbers. Quantum-mechanical equivalent for this state means that two electrons are not allowed to have the same space simultaneously. Since one electron may certain discrete same set of n, l and m values, however, the fourth spin quantum number should be different. This provides a unique address to every electron. Since the possible magnitudes of spin can be either +1/2 or -1/2 thus at maximum two electrons can occupy any given orbital.
The convention followed to remove or add electrons is electrons of largest principal quantum number are lost first. In case of subshells of the same
(b)
Interpretation:
The
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemical Principles: The Quest for Insight
- 3. Arrange the following iso-electronic species in order of increasing ionic radius: Br –, Sr2+, As3–. Give a brief explanation why you chose this arrangement.arrow_forwardHow many valence electron does Br havearrow_forwardWrite the electron configurations for the following ions, anddetermine which have noble-gas configurations: (a) Co2+ ,(b) Sn2+ , (c) Zr4+ , (d) Ag+, (e) S2- .arrow_forward
- What is the most probable position of a 1s electron in Li2+?arrow_forwardHow are measurements of paramagnetism used to support electron configurations derived spectroscopically? Use Cu(I) and Cu(II) chlorides as examples.arrow_forwardWrite electron configurations for the following ions. a. Ni2+ b. Cd2+ c. Zr3+ and Zr4+ d. Os2+ and Os3+arrow_forward
- Identify two ions that have the following ground-state electronconfigurations: (a) 3Ar4, (b) 3Ar43d5, (c) 3Kr45s24d10.arrow_forwarda) what is meant by the term polarizability? (b) Which of the following atoms would you expect to be most polarizable: O, S, Se, or Te? Explain.arrow_forwardTo which group of the periodic table does the general valence electron configuration ns2np4 correspond?arrow_forward
- Write the ground state electron configuration for the following species. (use shorthand notation) Mo2+ _______________________ Ag+ _______________________ P3- ______________________ Fe_______________________ Which of any are paramagnetic?arrow_forwardDefine the term electron domain?arrow_forwardWrite electron configurations for the following ions, anddetermine which have noble-gas configurations: (a) Cd2+,(b) P3-, (c) Zr4+, (d) Ru3+, (e) As3-, (f) Ag+.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

