
Chemistry & Chemical Reactivity
10th Edition
ISBN: 9781337399074
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 29PS
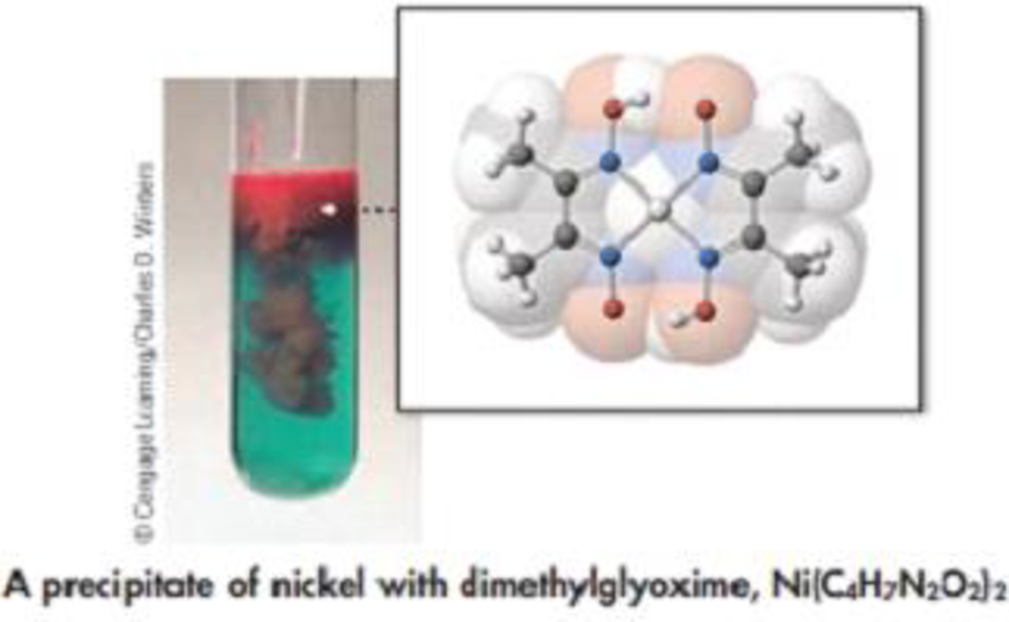
Nickel(II) sulfide, NiS, occurs naturally as the relatively rare mineral millerite. One of its occurrences is in meteorites. To analyze a mineral sample for the quantity of NiS, the sample is dissolved in nitric add to form a solution of Ni(NO3)2.
NiS(s) + 4 HNO3(aq) → Ni(NO3)2(aq) + 2 NO2(g) + 2 H2O(l) + S(s)
The aqueous solution of Ni(NO3)2 is then reacted with the organic compound dimethylglyoxime (C4H8N2O2) to give the red solid Ni((C4H7N2O2)2.
Ni(NO3)2(aq) + 2 (C4H8N2O2)(aq) → Ni(C4H7N2O2)2 + 2 HNO3(aq)
Suppose a 0.468-g sample containing millerite produces 0.206 g of red, solid Ni(C4H7N2O2)2
What is the mass percent of NiS in the sample?
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 4 Solutions
Chemistry & Chemical Reactivity
Ch. 4.1 - What mass of oxygen, O2, is required to completely...Ch. 4.2 - The thermite reaction produces iron metal and...Ch. 4.4 - One method for determining the purity of a sample...Ch. 4.4 - A 0523-g sample of the unknown compound CxHy was...Ch. 4.4 - Prob. 4.5CYUCh. 4.5 - Sodium bicarbonate, NaHCO3, is used in baking...Ch. 4.5 - An experiment calls for you to use 250. mL of 1.00...Ch. 4.6 - (a) What is the pH of a solution of HCI in which...Ch. 4.7 - Prob. 4.9CYUCh. 4.8 - A 25.0-mL sample of vinegar (which contains the...
Ch. 4.8 - Hydrochloric acid. HCl, with a concentration of...Ch. 4.8 - An unknown monoprotic acid reacts with NaOH...Ch. 4.8 - Vitamin C, ascorbic acid (C6HgO6)(molar mass 176.1...Ch. 4.9 - Prob. 4.14CYUCh. 4.9 - Prob. 1.1ACPCh. 4.9 - Excess KI is added to a 100.0-mL sample of a soft...Ch. 4.9 - Prob. 3.1ACPCh. 4.9 - Identify the factor labeled 4 in the strategy...Ch. 4.9 - Identify the factor labeled 3 in this strategy...Ch. 4.9 - Prob. 4.3ACPCh. 4.9 - Prob. 4.4ACPCh. 4 - The reaction of iron(III) oxide with aluminum to...Ch. 4 - What mass of HCI, in grams, is required to react...Ch. 4 - Like many metals, aluminum reacts with a halogen...Ch. 4 - The balanced equation for the reduction of iron...Ch. 4 - Methane, CH4, burns in oxygen. (a) What are the...Ch. 4 - The formation of water-Insoluble silver chloride...Ch. 4 - The metals industry was a major source of air...Ch. 4 - Prob. 8PSCh. 4 - Chromium metal reacts with oxygen to give...Ch. 4 - Ethane, C2H6, burns in oxygen. (a) What are the...Ch. 4 - Prob. 11PSCh. 4 - Ammonia gas can be prepared by the reaction of a...Ch. 4 - The compound SF6 is made by burning sulfur in an...Ch. 4 - Disulfur dichloride, S2Cl2, is used to vulcanize...Ch. 4 - The reaction of methane and water is one way to...Ch. 4 - Aluminum chloride AlCl3, is made by treating scrap...Ch. 4 - In the thermite reaction, iron(III) oxide is...Ch. 4 - Aspirin, C6H4 (OCOCH3) CO3H, is produced by the...Ch. 4 - In Example 4.2, you found that a particular...Ch. 4 - Ammonia gas can be prepared by the following...Ch. 4 - The deep blue compound Cu(NH3)4S04 is made by the...Ch. 4 - Black smokers are found in the depths of the...Ch. 4 - The reaction of methane and water is one way to...Ch. 4 - Methanol, CH3OH, can be prepared from carbon...Ch. 4 - A mixture of CuSO4 and CuSO4.5 H2O has a mass of...Ch. 4 - A 2.634-g sample containing impure CuCl2 2 H2O was...Ch. 4 - Prob. 27PSCh. 4 - Prob. 28PSCh. 4 - Nickel(II) sulfide, NiS, occurs naturally as the...Ch. 4 - The aluminum in a 0.764-g sample of an unknown...Ch. 4 - Prob. 31PSCh. 4 - Mesitylene is a liquid hydrocarbon Burning 0.115 g...Ch. 4 - Naphthalene is a hydrocarbon that once was used in...Ch. 4 - Azulene is a beautiful blue hydrocarbon. If 0.106...Ch. 4 - An unknown compound has the formula CxHyOz. You...Ch. 4 - An unknown compound has the formula CxHyOz. You...Ch. 4 - Nickel forms a compound with carbon monoxide,...Ch. 4 - To find the formula of a compound composed of iron...Ch. 4 - If 6.73 g of Na2CO3 is dissolved in enough water...Ch. 4 - Some potassium dichromate (K2Cr2O7), 2.335 g, is...Ch. 4 - What is the mass of solute, in grams, in 250, mL...Ch. 4 - Prob. 42PSCh. 4 - What volume of 0123 M NaOH, in milliliters,...Ch. 4 - What volume of 2.06 M KMnO4, in liters, contains...Ch. 4 - Identify the ions that exist in each aqueous...Ch. 4 - Identify the ions that exist in each aqueous...Ch. 4 - An experiment in your laboratory requires 500. mL...Ch. 4 - What mass of oxalic acid, H2C2O4, is required to...Ch. 4 - If you dilute 25.0 mL of 1.50 M hydrochloric acid...Ch. 4 - If 4.00 mL of 0.0250 M CuSO4 is diluted to 10.0 mL...Ch. 4 - Which of the following methods would you use to...Ch. 4 - Which of the following methods would you use to...Ch. 4 - You have 250. mL of 0.136 M HCl. Using a...Ch. 4 - Prob. 54PSCh. 4 - A table wine has a pH of 3.40. What is the...Ch. 4 - A saturated solution of milk of magnesia, Mg(OH)2,...Ch. 4 - Prob. 57PSCh. 4 - Prob. 58PSCh. 4 - Prob. 59PSCh. 4 - Prob. 60PSCh. 4 - Prob. 61PSCh. 4 - What mass of Na2CO3, in grams, is required for...Ch. 4 - When an electric current is passed through an...Ch. 4 - Hydrazine, N2H4, a base like ammonia, can react...Ch. 4 - In the photographic developing process, silver...Ch. 4 - You can dissolve an aluminum soft drink can in an...Ch. 4 - What volume of 0.750 M Pb(NO3)2, in milliliters,...Ch. 4 - What volume of 0.125 M oxalic acid, H2C2O4, is...Ch. 4 - What volume of 0.812 M HCI, in milliliters, is...Ch. 4 - What volume of 0.955 M HCl, in milliliters, is...Ch. 4 - If 38.55 mL of HCI is required to titrate 2.150 g...Ch. 4 - Potassium hydrogen phthalate, KHCgH4O4, is used to...Ch. 4 - You have 0.954 g of an unknown acid, H2A, which...Ch. 4 - An unknown solid acid is either citric acid or...Ch. 4 - To analyze an iron-containing compound, you...Ch. 4 - Vitamin C has the formula C6H8O6. Besides being an...Ch. 4 - Prob. 77PSCh. 4 - Suppose 16.04 g of benzene, C6H6, is burned in...Ch. 4 - The metabolic disorder diabetes causes a buildup...Ch. 4 - Your body deals with excess nitrogen by excreting...Ch. 4 - The reaction of iron metal and chlorine gas to...Ch. 4 - Prob. 83GQCh. 4 - The reaction of 750. g each of NH3 and O2 was...Ch. 4 - Sodium azide, an explosive chemical used in...Ch. 4 - Prob. 86GQCh. 4 - Prob. 87GQCh. 4 - Prob. 88GQCh. 4 - Prob. 89GQCh. 4 - A Menthol, from oil of mint, has a characteristic...Ch. 4 - Benzoquinone, a chemical used in the dye industry...Ch. 4 - Aqueous solutions of iron(II) chloride and sodium...Ch. 4 - Sulfuric acid can be prepared starting with the...Ch. 4 - Prob. 94GQCh. 4 - An unknown metal reacts with oxygen to give the...Ch. 4 - Titanium(IV) oxide, TiO2, is heated in hydrogen...Ch. 4 - Potassium perchlorate is prepared by the following...Ch. 4 - A Commercial sodium "hydrosulfite" is 90.1%...Ch. 4 - What mass of lime, CaO, can be obtained by heating...Ch. 4 - The elements silver, molybdenum, and sulfur...Ch. 4 - A mixture of butene, C4Hg, and butane, is burned...Ch. 4 - Cloth can be waterproofed by coating it with a...Ch. 4 - Copper metal can be prepared by roasting copper...Ch. 4 - Prob. 104GQCh. 4 - Sodium bicarbonate and acetic acid react according...Ch. 4 - A noncarbonated soft drink contains an unknown...Ch. 4 - Sodium thiosulfate, Na2S2O3, is used as a fixer in...Ch. 4 - You have a mixture of oxalic acid, H2C2O4, and...Ch. 4 - (a) What is the pH of a 0.105 M HCl solution? (b)...Ch. 4 - A solution of hydrochloric acid has a volume of...Ch. 4 - One half liter (500. mL) of 2.50 M HCl is mixed...Ch. 4 - A solution of hydrochloric acid has a volume of...Ch. 4 - Prob. 113GQCh. 4 - Prob. 115GQCh. 4 - Prob. 116GQCh. 4 - Gold can be dissolved from gold-bearing rock by...Ch. 4 - You mix 25.0 mL of 0.234 M FeCl3 with 42.5 mL of...Ch. 4 - Prob. 119GQCh. 4 - ATOM ECONOMY: Ethylene oxide, C2H4O, is an...Ch. 4 - Suppose you dilute 25.0 mL of a 0.110 M solution...Ch. 4 - Prob. 122ILCh. 4 - Oyster beds in the oceans require chloride ions...Ch. 4 - You wish to determine the weight percent of copper...Ch. 4 - Prob. 126ILCh. 4 - Chromium(III) chloride forms many compounds with...Ch. 4 - Thioridazine, C21H26N2S2, is a pharmaceutical...Ch. 4 - A herbicide contains 2,4-D...Ch. 4 - Sulfuric acid is listed in a catalog with a...Ch. 4 - Two beakers sit on a balance; the total mass is...Ch. 4 - A weighed sample of iron (Fe) is added to liquid...Ch. 4 - Let us explore a reaction with a limiting...Ch. 4 - Two students titrate different samples of the same...Ch. 4 - ATOM ECONOMY: Benzene, C6H6, is a common compound,...Ch. 4 - ATOM ECONOMY: Maleic anhydride, C4H2O3, can be...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Determine the volume of sulfuric acid solution needed to prepare 37.4 g of aluminum sulfate, Al2(SO4)3, by the reaction 2Al(s)+3H2SO4(aq)Al2(SO4)3(aq)+3H2(g) The sulfuric acid solution, whose density is 1.104 g/mL, contains 15.0% H2SO4 by mass.arrow_forwardThe final step in the manufacture of platinum metal (for use in automotive catalytic converters and other products) is the reaction 3 (NH4)2PtCl6(s) 3 Pt(s) + 2 NH4Cl(s) + 2 N2(g) + 16 HCl(g) Complete this table of reaction quantities for the reaction of 12.35 g (NH4)2PtCl6.arrow_forwardA soluble iodide was dissolved in water. Then an excess of silver nitrate, AgNO3, was added to precipitate all of the iodide ion as silver iodide, AgI. If 1.545 g of the soluble iodide gave 2.185 g of silver iodide, how many grams of iodine are in the sample of soluble iodide? What is the mass percentage of iodine, I, in the compound?arrow_forward
- A solution of scandium chloride was treated with silver nitrate. The chlorine in the scandium compound was converted to silver chloride, AgCl. A 58.9-mg sample of scandium chloride gave 167.4 mg of silver chloride. What are the mass percentages of Sc and Cl in scandium chloride? What is its empirical formula?arrow_forwardSilicon is produced for the chemical and electronics industries by the following reactions. Give the balanced equation for each reaction. a. SiO2(s)+C(s)arefurnaceElectricSi(s)+CO(g) b. Liquid silicon tetrachloride is reacted with very pure solid magnesium, producing solid silicon and solid magnesium chloride. c. Na2SiF6(s) + Na(s) Si(s) + NaF(s)arrow_forward4.28 One of the steps in the manufacture of nitric acid is the oxidation of ammonia shown in this equation: 4NH3(g)+5O2(g)4NO(g)+6H2O(g) If 43.0 kg of NH3 reacts with 35.4 kg of O2, what mass of NO forms?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY