
Chemistry & Chemical Reactivity
10th Edition
ISBN: 9781337399074
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4.9, Problem 4.1ACP
Identify the factor labeled 4 in the strategy map
- (a) Density H2O(ℓ), 1.00 g/mL
- (b) Density of O2(ℓ), 1.14 g/mL
- (c) molar mass of O2, 32.0 g/mol
- (d) molar mass of H2O, 18.0 g/mol
below. Labels 1 through 5 are the factors used in each step of the calculation, and A is the mass of O2 and B is the amount of H2O.
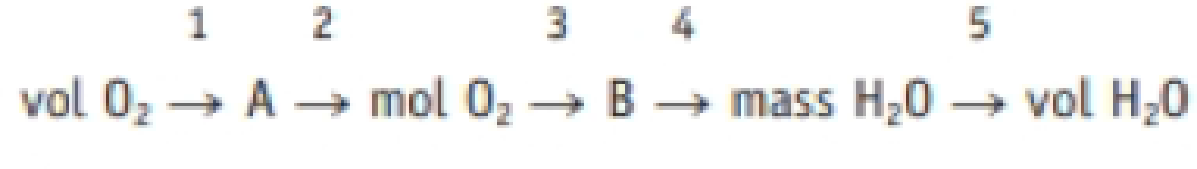
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
2 When corn is allowed to ferment, the fructose in the
corn is converted to ethyl alcohol according to the following
reaction
C,H12O6(aq) –→ 2C,H;OH(1) + 2CO2(g)
(a) What volume of ethyl alcohol (d = 0.789 g/mL) is
produced from one pound of fructose?
(b) Gasohol can be a mixture of 10 mL ethyl alcohol
and 90 mL of gasoline. How many grams of fructose are
required to produce the ethyl alcohol in one gallon of
gasohol?
%3D
62. When corn is allowed to ferment, the fructose in the
corn is converted to ethyl alcohol according to the following
reaction
C,H12O6(aq) →2C,H;OH(1) + 2CO2(g)
(a) What volume of ethyl alcohol (d = 0.789 g/mL) is
produced from one pound of fructose?
(b) Gasohol can be a mixture of 10 mL ethyl alcohol
and 90 mL of gasoline. How many grams of fructose are
required to produce the ethyl alcohol in one gallon of
gasohol?
7
The answers should be rounded off to three (3) significant figures and reported in
scientific notation [enter the coefficient and the exponent in the space provided].
Given 1.82 x 105 mg of diphosphorous pentoxide:
A How many millimoles (mmoles) are there?
mmol of compound
х 10
%3D
B How many molecules are there?
molecules =
х 10
C How many grams of phosphorous are there?
grams of P =
x 10
D How many moles of oxygen atoms are there?
moles of O =
х 10
How many mL of Br2 with a density of 3.12 g/mL must you dispense to have 1.56 mole
(mol) of Br2 ()-
(1)
The answer should be rounded off to three (3) significant figures and reported in decimal
notation
mL of Bromine =
mL
What is the mass of 0.6832 moles of Zinc perbromate? The answers should be rounded off
to the correct number of significant figures and reported in DECIMAL notation. Do not
enter units (already provided for you)
Answer =
grams
Chapter 4 Solutions
Chemistry & Chemical Reactivity
Ch. 4.1 - What mass of oxygen, O2, is required to completely...Ch. 4.2 - The thermite reaction produces iron metal and...Ch. 4.4 - One method for determining the purity of a sample...Ch. 4.4 - A 0523-g sample of the unknown compound CxHy was...Ch. 4.4 - Prob. 4.5CYUCh. 4.5 - Sodium bicarbonate, NaHCO3, is used in baking...Ch. 4.5 - An experiment calls for you to use 250. mL of 1.00...Ch. 4.6 - (a) What is the pH of a solution of HCI in which...Ch. 4.7 - Prob. 4.9CYUCh. 4.8 - A 25.0-mL sample of vinegar (which contains the...
Ch. 4.8 - Hydrochloric acid. HCl, with a concentration of...Ch. 4.8 - An unknown monoprotic acid reacts with NaOH...Ch. 4.8 - Vitamin C, ascorbic acid (C6HgO6)(molar mass 176.1...Ch. 4.9 - Prob. 4.14CYUCh. 4.9 - Prob. 1.1ACPCh. 4.9 - Excess KI is added to a 100.0-mL sample of a soft...Ch. 4.9 - Prob. 3.1ACPCh. 4.9 - Identify the factor labeled 4 in the strategy...Ch. 4.9 - Identify the factor labeled 3 in this strategy...Ch. 4.9 - Prob. 4.3ACPCh. 4.9 - Prob. 4.4ACPCh. 4 - The reaction of iron(III) oxide with aluminum to...Ch. 4 - What mass of HCI, in grams, is required to react...Ch. 4 - Like many metals, aluminum reacts with a halogen...Ch. 4 - The balanced equation for the reduction of iron...Ch. 4 - Methane, CH4, burns in oxygen. (a) What are the...Ch. 4 - The formation of water-Insoluble silver chloride...Ch. 4 - The metals industry was a major source of air...Ch. 4 - Prob. 8PSCh. 4 - Chromium metal reacts with oxygen to give...Ch. 4 - Ethane, C2H6, burns in oxygen. (a) What are the...Ch. 4 - Prob. 11PSCh. 4 - Ammonia gas can be prepared by the reaction of a...Ch. 4 - The compound SF6 is made by burning sulfur in an...Ch. 4 - Disulfur dichloride, S2Cl2, is used to vulcanize...Ch. 4 - The reaction of methane and water is one way to...Ch. 4 - Aluminum chloride AlCl3, is made by treating scrap...Ch. 4 - In the thermite reaction, iron(III) oxide is...Ch. 4 - Aspirin, C6H4 (OCOCH3) CO3H, is produced by the...Ch. 4 - In Example 4.2, you found that a particular...Ch. 4 - Ammonia gas can be prepared by the following...Ch. 4 - The deep blue compound Cu(NH3)4S04 is made by the...Ch. 4 - Black smokers are found in the depths of the...Ch. 4 - The reaction of methane and water is one way to...Ch. 4 - Methanol, CH3OH, can be prepared from carbon...Ch. 4 - A mixture of CuSO4 and CuSO4.5 H2O has a mass of...Ch. 4 - A 2.634-g sample containing impure CuCl2 2 H2O was...Ch. 4 - Prob. 27PSCh. 4 - Prob. 28PSCh. 4 - Nickel(II) sulfide, NiS, occurs naturally as the...Ch. 4 - The aluminum in a 0.764-g sample of an unknown...Ch. 4 - Prob. 31PSCh. 4 - Mesitylene is a liquid hydrocarbon Burning 0.115 g...Ch. 4 - Naphthalene is a hydrocarbon that once was used in...Ch. 4 - Azulene is a beautiful blue hydrocarbon. If 0.106...Ch. 4 - An unknown compound has the formula CxHyOz. You...Ch. 4 - An unknown compound has the formula CxHyOz. You...Ch. 4 - Nickel forms a compound with carbon monoxide,...Ch. 4 - To find the formula of a compound composed of iron...Ch. 4 - If 6.73 g of Na2CO3 is dissolved in enough water...Ch. 4 - Some potassium dichromate (K2Cr2O7), 2.335 g, is...Ch. 4 - What is the mass of solute, in grams, in 250, mL...Ch. 4 - Prob. 42PSCh. 4 - What volume of 0123 M NaOH, in milliliters,...Ch. 4 - What volume of 2.06 M KMnO4, in liters, contains...Ch. 4 - Identify the ions that exist in each aqueous...Ch. 4 - Identify the ions that exist in each aqueous...Ch. 4 - An experiment in your laboratory requires 500. mL...Ch. 4 - What mass of oxalic acid, H2C2O4, is required to...Ch. 4 - If you dilute 25.0 mL of 1.50 M hydrochloric acid...Ch. 4 - If 4.00 mL of 0.0250 M CuSO4 is diluted to 10.0 mL...Ch. 4 - Which of the following methods would you use to...Ch. 4 - Which of the following methods would you use to...Ch. 4 - You have 250. mL of 0.136 M HCl. Using a...Ch. 4 - Prob. 54PSCh. 4 - A table wine has a pH of 3.40. What is the...Ch. 4 - A saturated solution of milk of magnesia, Mg(OH)2,...Ch. 4 - Prob. 57PSCh. 4 - Prob. 58PSCh. 4 - Prob. 59PSCh. 4 - Prob. 60PSCh. 4 - Prob. 61PSCh. 4 - What mass of Na2CO3, in grams, is required for...Ch. 4 - When an electric current is passed through an...Ch. 4 - Hydrazine, N2H4, a base like ammonia, can react...Ch. 4 - In the photographic developing process, silver...Ch. 4 - You can dissolve an aluminum soft drink can in an...Ch. 4 - What volume of 0.750 M Pb(NO3)2, in milliliters,...Ch. 4 - What volume of 0.125 M oxalic acid, H2C2O4, is...Ch. 4 - What volume of 0.812 M HCI, in milliliters, is...Ch. 4 - What volume of 0.955 M HCl, in milliliters, is...Ch. 4 - If 38.55 mL of HCI is required to titrate 2.150 g...Ch. 4 - Potassium hydrogen phthalate, KHCgH4O4, is used to...Ch. 4 - You have 0.954 g of an unknown acid, H2A, which...Ch. 4 - An unknown solid acid is either citric acid or...Ch. 4 - To analyze an iron-containing compound, you...Ch. 4 - Vitamin C has the formula C6H8O6. Besides being an...Ch. 4 - Prob. 77PSCh. 4 - Suppose 16.04 g of benzene, C6H6, is burned in...Ch. 4 - The metabolic disorder diabetes causes a buildup...Ch. 4 - Your body deals with excess nitrogen by excreting...Ch. 4 - The reaction of iron metal and chlorine gas to...Ch. 4 - Prob. 83GQCh. 4 - The reaction of 750. g each of NH3 and O2 was...Ch. 4 - Sodium azide, an explosive chemical used in...Ch. 4 - Prob. 86GQCh. 4 - Prob. 87GQCh. 4 - Prob. 88GQCh. 4 - Prob. 89GQCh. 4 - A Menthol, from oil of mint, has a characteristic...Ch. 4 - Benzoquinone, a chemical used in the dye industry...Ch. 4 - Aqueous solutions of iron(II) chloride and sodium...Ch. 4 - Sulfuric acid can be prepared starting with the...Ch. 4 - Prob. 94GQCh. 4 - An unknown metal reacts with oxygen to give the...Ch. 4 - Titanium(IV) oxide, TiO2, is heated in hydrogen...Ch. 4 - Potassium perchlorate is prepared by the following...Ch. 4 - A Commercial sodium "hydrosulfite" is 90.1%...Ch. 4 - What mass of lime, CaO, can be obtained by heating...Ch. 4 - The elements silver, molybdenum, and sulfur...Ch. 4 - A mixture of butene, C4Hg, and butane, is burned...Ch. 4 - Cloth can be waterproofed by coating it with a...Ch. 4 - Copper metal can be prepared by roasting copper...Ch. 4 - Prob. 104GQCh. 4 - Sodium bicarbonate and acetic acid react according...Ch. 4 - A noncarbonated soft drink contains an unknown...Ch. 4 - Sodium thiosulfate, Na2S2O3, is used as a fixer in...Ch. 4 - You have a mixture of oxalic acid, H2C2O4, and...Ch. 4 - (a) What is the pH of a 0.105 M HCl solution? (b)...Ch. 4 - A solution of hydrochloric acid has a volume of...Ch. 4 - One half liter (500. mL) of 2.50 M HCl is mixed...Ch. 4 - A solution of hydrochloric acid has a volume of...Ch. 4 - Prob. 113GQCh. 4 - Prob. 115GQCh. 4 - Prob. 116GQCh. 4 - Gold can be dissolved from gold-bearing rock by...Ch. 4 - You mix 25.0 mL of 0.234 M FeCl3 with 42.5 mL of...Ch. 4 - Prob. 119GQCh. 4 - ATOM ECONOMY: Ethylene oxide, C2H4O, is an...Ch. 4 - Suppose you dilute 25.0 mL of a 0.110 M solution...Ch. 4 - Prob. 122ILCh. 4 - Oyster beds in the oceans require chloride ions...Ch. 4 - You wish to determine the weight percent of copper...Ch. 4 - Prob. 126ILCh. 4 - Chromium(III) chloride forms many compounds with...Ch. 4 - Thioridazine, C21H26N2S2, is a pharmaceutical...Ch. 4 - A herbicide contains 2,4-D...Ch. 4 - Sulfuric acid is listed in a catalog with a...Ch. 4 - Two beakers sit on a balance; the total mass is...Ch. 4 - A weighed sample of iron (Fe) is added to liquid...Ch. 4 - Let us explore a reaction with a limiting...Ch. 4 - Two students titrate different samples of the same...Ch. 4 - ATOM ECONOMY: Benzene, C6H6, is a common compound,...Ch. 4 - ATOM ECONOMY: Maleic anhydride, C4H2O3, can be...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A solution of 75 mM tartaric acid (C4H,O6) in water is used as a standard in HPLC analysis. How many grams of C4H,O6 ·H2O are needed to make up 10 ml of solution?arrow_forwardThe three sets of units for expressing density (or concentration, as in “mass of solute per unit volume of solvent”) below are equivalent: kg⁄L = g⁄mL =g cm! ⁄a) Using dimensional analysis, show that the above expression is true.b) What is 1.0 kg⁄L = 1.0 g⁄mL = 1.0 g cm! ⁄ expressed in SI base units?arrow_forward5. (optional) Blue turquoise is an example of a hydrate mineral containing copper; the chemical formula is CuAl6(PO4)4(OH)8•4H2Ó. What is the percent water in this semiprecious stone?arrow_forward
- 7. Suppose you make a solution that contains 17.59 g of sodium chloride and 960 g of water. (a) What is the mass % of sodium chloride? (b) If 16.47 mL of your solution weighs 16.651 g,what is the density (in g/cc)?arrow_forward(b) The combustion of octane is represented by the following balanced equation: 2 C3H28(1) + 25 Ozlg) → 16 CO2 (g) + 18 H20 (g) In a brief chemistry or scientific language, describe this reaction:arrow_forward(a) Carbon monoxide (CO) is a poisonous gas because it binds very strongly to the oxygen carrier hemoglobin in blood. A concentration of 8.00 × 102 ppm by volume of carbon monoxide is considered lethal to humans. Calculate the volume in liters occupied by carbon monoxide in a room that measures 17.6 m long, 8.80 m wide, and 2.64 m high at this concentration.(b) Prolonged exposure to mercury (Hg) vapor can cause neurological disorders and respiratory problems. For safe air quality control, the concentration of mercury vapor must be under 0.050 mg/m3 . Convert this number to g/L. (c) The general test for type II diabetes is that the blood sugar (glucose) level should be below 120 mg per deciliter (mg/dL). Convert this number to micrograms per milliliter (μg/mL).arrow_forward
- (b) Potassium nitrate was synthesized in an experiment using 10 mL of nitric acid and excess potassium hydroxide. The mass data obtained from the experiment is shown in Table 1 below. Given that the percentage yield of potassium nitrate was 88.3 %, calculate the concentration (% m/v) of the nitric acid used in the experiment. Table 1: Mass data for the synthesis of potassium nitrate from nitric acid and potassium hydroxide Mass of clock glass and crystallized potassium nitrate/ 45.55 Mass of empty clock glass/ g 39.18arrow_forwardThe production of ammonia nia * (N*H_{3}) from nitrogen and hydrogen, shown below, was an important breakthrough resulting in Nobel prizes in chemistry to Fritz Haber (1918) and Carl Bosch (1931). This compound is widely used in manufacturing fertilizers. 3H 2 (g)+N 2 (g) 2NH 3 (g) Calculate the mass of nitrogen (in kg ) needed to manufacture 1000 kg of ammonia . Assume four significant figures .arrow_forwardThe following ratios of NaOH to HCl were determined in an analysis: T1 = 0.9958, T2 = 0.9929. How much is the precision?arrow_forward
- A power plant is driven by the combustion of a complex fossil fuel having the formula C11H7S. Assume the air supply is composed of only N2 and O2 with a molar ratio of 3.76:1.00, and the N2 remains unreacted. In addition to the water produced, the fuels C is completely combusted to CO2 and its sulfur content is converted to SO2. In order to evaluate gases emitted at the exhaust stacks for environmental regulation purposes, the nitrogen supplied with the air must also be included in the balanced reactions. a Including the N2 supplied m the air, write a balanced combustion equation for the complex fuel assuming 100% stoichiometric combustion (i.e., when there is no excess oxygen in the products and the only C-containing product is CO2). Except in the case of N2, use only integer coefficients. b Including N2 supplied in the air, write a balanced combustion equation for the complex fuel assuming 120% stoichiometric combustion (i.e., when excess oxygen is present in the products and the only C-containing product is CO2). Except in the case of use only integer coefficients c Calculate the minimum mass (in kg) of air required to completely combust 1700 kg of C11H7S. d Calculate the air/fuel mass ratio, assuming 100% stoichiometric combustion. e Calculate the air/fuel mass ratio, assuming 120% stoichiometric combustion.arrow_forwardA soft drink contains an unknown mass of citric acid, C3H5O(COOH)3. It requires 6.42 mL of 9.580 × 10−2-M NaOH to neutralize the citric acid in 10.0 mL of the soft drink. C3H5O(COOH)3(aq) + 3 NaOH(aq) → Na3C3H5O(COO)3(aq) + 3 H2O(ℓ) Determine which step in these calculations for the mass of citric acid in 1 mL soft drink is incorrect? Why? n (NaOH) = (6.42 mL)(1L/1000 mL)(9.580 × 10−2 mol/L) n (citric acid) = (6.15 × 10−4 mol NaOH) × (3 mol citric acid/1 mol NaOH) m (citric acid in sample) = (1.85 × 10−3 mol citric acid) × (192.12 g/mol citric acid) m (citric acid in 1 mL soft drink) = (0.354 g citric acid)/(10 mL soft drink) Determine the correct result.arrow_forward4-77 To convert 1 mol of iron(III) oxide to its elements requires 196.5 kcal: How many grams of iron can be produced if 156.0 kcal of heat is absorbed by a large-enough sample of iron(III) oxide?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning
Step by Step Stoichiometry Practice Problems | How to Pass ChemistryMole Conversions Made Easy: How to Convert Between Grams and Moles; Author: Ketzbook;https://www.youtube.com/watch?v=b2raanVWU6c;License: Standard YouTube License, CC-BY