Problem 1ALQ: elate Active Learning Question 2 from Chapter 2 to the concepts of chemical stoichiometry. Problem 2ALQ: You are making cookies and are missing a key ingredient—eggs. You have plenty of the other... Problem 3ALQ: Nitrogen (N2) and hydrogen (H2)react to form ammonia (NH3). Consider the mixture of N2 and H2 in a... Problem 4ALQ Problem 5ALQ: ou know that chemical A reacts with chemical B. You react 10.0 g A with 10.0 g B. What information... Problem 6ALQ: f 10.0 g of hydrogen gas is reacted with 10.0 g of oxygen gas according to the equation... Problem 7ALQ Problem 8ALQ Problem 9ALQ: hat happens to the weight of an iron bar when it rusts? l type='a'> There is no change because mass... Problem 10ALQ Problem 11ALQ: What is meant by the term mole ratio? Give an example of a mote ratio, and explain how it is used in... Problem 12ALQ: Which would produce a greater number of moles of product: a given amount of hydrogen gas reacting... Problem 13ALQ: Consider a reaction represented by the following balanced equation :math>2A+3BC+4D u find that it... Problem 14ALQ Problem 15ALQ: Consider the balanced chemical equation :math>A+5B3C+4D en equal masses of A and B are reacted,... Problem 16ALQ: Which of the following reaction mixtures would produce the greatest amount of product, assuming all... Problem 17ALQ: Baking powder is a mixture of cream of tartar (KHC4H4O6)and baking soda (NaHCO3). When it is placed... Problem 18ALQ: You have seven closed containers each with equal masses of chlorine gas (Cl2). You add 10.0 g of... Problem 19ALQ Problem 20ALQ Problem 21ALQ: Consider the reaction between NO(g)and O2(g)represented below. mg... Problem 1QAP: hat do the coefficients of a balanced chemical equation tell us about the proportions in which atoms... Problem 2QAP: he vigorous reaction between aluminum and iodine gives the balanced equation:... Problem 3QAP Problem 4QAP: hich of the following statements is true for the reaction of nitrogen gas with hydrogen gas to... Problem 5QAP: or each of the following reactions, give the balanced equation for the reaction and state the... Problem 6QAP: or each of the following reactions, give the balanced chemical equation for the reaction and state... Problem 7QAP Problem 8QAP Problem 9QAP: onsider the balanced chemical equation :math>4Al(s)+3O2(g)2Al2O3(s). at mole ratio would you use to... Problem 10QAP: Write the balanced chemical equation for the complete combustion of heptene, C7H14. In combustion,... Problem 11QAP: For each of the following balanced chemical equations, calculate how many moles of product(s) would... Problem 12QAP Problem 13QAP: For each of the following balanced chemical equations, calculate how many grams of the product(s)... Problem 14QAP: For each of the following balanced chemical equations, calculate how many moles and how many grams... Problem 15QAP: For each of the following unbalanced equations, indicate how many moles of the second reactant would... Problem 16QAP: For each of the following unbalanced equations, indicate how many moles of the first product are... Problem 17QAP: What quantity serves as the conversion factor between the mass of a sample and how many moles the... Problem 18QAP Problem 19QAP: Using the average atomic masses given inside the front cover of this book, calculate how many moles... Problem 20QAP: Using the average atomic masses given inside the front cover of this book, calculate the number of... Problem 21QAP: Using the average atomic masses given inside the front cover of this book, calculate the mass in... Problem 22QAP: Using the average atomic masses given inside the front cover of this hook, calculate the mass in... Problem 23QAP: For each of the following unbalanced equations, calculate how many moles of the second reactant... Problem 24QAP: For each of the following unbalanced equations, calculate how many moles of the second reactant... Problem 25QAP: For each of the following unbalanced equations, calculate how many grants of each product would be... Problem 26QAP: Boron nitride reacts with iodine monofluoride i trichlorofluoro methane at 30°C to produce pure... Problem 27QAP: “Smelling salts,” which are used to revive someone who has fainted, typically contain ammonium... Problem 28QAP: Calcium carbide, CaC2, can be produced in an electric furnace by strongly heating calcium oxide... Problem 29QAP: When elemental carbon is burned in the open atmosphere, with plenty of oxygen gas present, the... Problem 30QAP: If baking soda (sodium hydrogen carbonate) is heated strongly, the following reaction occurs:... Problem 31QAP: Although we usually think of substances as “burning” only in oxygen gas, the process of rapid... Problem 32QAP: When yeast is added to a solution of glucose or fructose, the sugars are said to undergo... Problem 33QAP: Sulfurous acid is unstable in aqueous solution and gradually decomposes to water and sulfur dioxide... Problem 34QAP: Small quantities of ammonia gas can be generated in the laboratory by heating an ammonium salt with... Problem 35QAP: Elemental phosphorus bums in oxygen with an intensely hot flame, producing a brilliant light and... Problem 36QAP Problem 37QAP: Ammonium nitrate has been used as a high explosive because it is unstable and decomposes into... Problem 38QAP: If common sugars arc heated too strongly, they char as they decompose into carbon ¡md water vapor.... Problem 39QAP: Thionyl chloride, SOCl2, is used as a very powerful drying agent in many synthetic chemistry... Problem 40QAP Problem 41QAP: Which of the following statements is(are) true? l type='a'> A balanced equation relates the numbers... Problem 42QAP: Explain how one determines which reactant in a process is the limiting reactant. Does this depend... Problem 43QAP: Consider the equation: 2A+B5C. If 10.0 g of A reacts with 5.00 g of B. how is the limiting reactant... Problem 44QAP: Balance the following chemical equation, and then answer the question below.... Problem 45QAP: For each of the following unbalanced reactions, suppose exactly 5.00 moles of each reactant are... Problem 46QAP: For each of the following unbalanced chemical equations, suppose that exactly 5.00 g of each... Problem 47QAP: For each of the following unbalanced chemical equations, suppose 10.0 g of each reactant is taken.... Problem 48QAP: For each of the following unbalanced chemical equations, suppose that exactly 1.00 g of each... Problem 49QAP: For each of the following unbalanced chemical equations, suppose 1.00 g of each reactant is taken.... Problem 50QAP: For each of the following unbalanced chemical equations, suppose that exactly 15.0 g of each... Problem 51QAP: Lead(II) carbonate, also called “white lead,” was formerly used as a pigment in white paints.... Problem 52QAP: Copper(II) sulfate has been used extensively as a fungicide (kills fungus) and herbicide (kills... Problem 53QAP: Lead(II) oxide from an ore can be reduced to elemental lead by heating in a furnace with carbon.... Problem 54QAP: If steel wool (iron) is heated until it glows and is placed in a bottle containing pure oxygen, the... Problem 55QAP: A common method for determining how much chloride ion is present in a sample is to precipitate the... Problem 56QAP: Although many sulfate salts are soluble in water, calcium sulfate is not (Table 7. 1). Therefore, a... Problem 57QAP: Hydrogen peroxide is used as a cleaning agent in the treatment of cuts and abrasions for several... Problem 58QAP: Silicon carbide, SIC, is one of the hardest materials known. Surpassed in hardness only by diamond,... Problem 59QAP Problem 60QAP: The text explains that one reason why the actual yield for a reaction may be less than the... Problem 61QAP: According to his prelaboratory theoretical yield calculations, a student’s experiment should have... Problem 62QAP: An air bag is deployed by utilizing the following re tion the nitrogen gas produced inflates the air... Problem 63QAP: The compound sodium thiosutfate pentahydrate. Na2SO35H2O, is important commercially to the... Problem 64QAP: Alkali metal hydroxides are sometimes used to “scrub” excess carbon dioxide from the air in closed... Problem 65QAP: Although they were formerly called the inert gases, at least the heavier elements of Group 8 do form... Problem 66QAP: Solid copper can be produced by passing gaseous ammonia over solid copper (II) oxide al high... Problem 67AP Problem 68AP Problem 69AP Problem 70AP: When the sugar glucose, C6H12O6, is burned in air, carbon dioxide and water vapor are produced.... Problem 71AP: When elemental copper is strongly heated with sulfur, a mixture of CuS and Cu2Sis produced. with CuS... Problem 72AP: Barium chloride solutions are used in chemical analysis for the quantitative precipitation of... Problem 73AP: The traditional method of analysis for the amount of chloride ion present in a sample is to dissolve... Problem 74AP: For each of the following reactions, give the balanced equation for the reaction and state the... Problem 75AP Problem 76AP: Consider the balanced equation :math>C3H8(g)+5O2(g)3CO2(g)+4H2O(g) at mole ratio enables you to... Problem 77AP: For each of the following balanced reactions, calculate how many moles of each product would be... Problem 78AP: For each of the following balanced equations, indicate how many moles of the product could be... Problem 79AP Problem 80AP: Using the average atomic masses given inside the front cover of the text, calculate the mass in... Problem 81AP: For each of the following incomplete and unbalanced equations, indicate how many moles of the second... Problem 82AP Problem 83AP Problem 84AP: It sodium peroxide is added to water, elemental oxygen gas is generated:... Problem 85AP: When elemental copper is placed in a solution of silver nitrate, the following oxidationreduction... Problem 86AP: When small quantities of elemental hydrogen gas are needed for laboratory work, the hydrogen is... Problem 87AP: The gaseous hydrocarbon acetylene, C2H2, is used in welders’ torches because of the large amount of... Problem 88AP: For each of the following unbalanced chemical equations, suppose exactly 5.0 g of each reactant is... Problem 89AP: For each of the following unbalanced chemical equations, suppose 25.0 g of each reactant is taken.... Problem 90AP: Hydrazine N2H4, emits a large quantity of energy when it reacts with oxygen, which has led to... Problem 91AP: Consider the following reaction: 4NH3(g)+5O2(g)4NO(g)+6H2O(g) a container were to have only 10... Problem 92AP: Before going to lab, a student read in his lab manual that the percent yield for a difficult... Problem 93CP: Consider the following unbalanced chemical equation for the combustion of pentane (C5H12):... Problem 94CP Problem 95CP: Consider the following unbalanced chemical equation. :math>LiOH(s)+CO2(g)Li2CO3(s)+H2O(l) 67.4 g of... Problem 96CP: Over the years, the thermite reaction has been used for welding railroad rails, in incendiary bombs,... Problem 97CP: Consider the following unbalanced chemical equation: :math>H2S(g)+O2(g)SO2(g)+H2O(g) termine the... Problem 98CP: Ammonia gas reacts with sodium metal to form sodium amide (NaNH2)and hydrogen gas. The unbalanced... Problem 99CP Problem 100CP: he production capacity for acrylonitrile (C3H3N)in the United States is over 2 billion pounds per... Problem 1CR Problem 2CR: erhaps the most important concept in introductory chemistry concerns what a mole of a substance... Problem 3CR: ow do we know that 16.00 g of oxygen Contains the same number of atoms as does 12.01 g of carbon,... Problem 4CR Problem 5CR: hat is meant by the percent composition by mass for a corn pound? Describe in general terms how this... Problem 6CR Problem 7CR Problem 8CR Problem 9CR Problem 10CR: Consider the unbalanced equation for the combustion of propane: :math>C3H8(g)+O2(g)CO2(g)+H2O(g)... Problem 11CR Problem 12CR: What is meant by a limiting reactant in a particular reaction? In what way is the reaction... Problem 13CR Problem 14CR Problem 15CR Problem 16CR Problem 17CR: A compound was analyzed and was found to have the following percent composition by mass: sodium,... Problem 18CR Problem 19CR Problem 20CR: Solid calcium carbide (CaC2)reacts with liquid water to produce acetylene gas (C2H2)and aqueous... Problem 21CR: A traditional analysis for samples containing calcium ion was to precipitate the calcium ion with... format_list_bulleted

 and
and
 in a closed container as illustrated below:
in a closed container as illustrated below: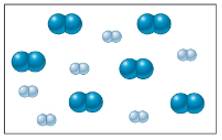


 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning




