
(a)
Interpretation:
Condensed structural formula for acetyl chloride has to be drawn.
Concept Introduction:
General structure of acid chloride can be represented as shown below,
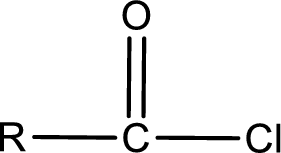
The structure of the acid chloride can be derived from the IUPAC name or common name. “R” group present in the above structure is a part of the parent
(b)
Interpretation:
Condensed structural formula for 2-methylbutanoyl chloride has to be drawn.
Concept Introduction:
General structure of acid chloride can be represented as shown below,
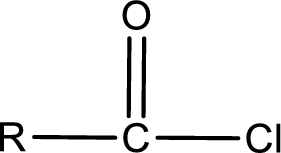
The structure of the acid chloride can be derived from the IUPAC name or common name. “R” group present in the above structure is a part of the parent carboxylic acid from which the acid chloride is obtained. Name of the acid chloride is obtained by replacing “-oic acid” in the parent carboxylic acid name with “-oyl chloride”.
(c)
Interpretation:
Condensed structural formula for propionic anhydride has to be drawn.
Concept Introduction:
General structure of acid anhydride can be represented as shown below,
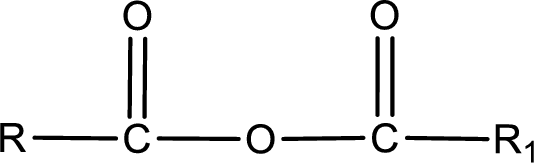
The structure of the acid anhydride can be derived from the IUPAC name or common name. “R, R1” group present in the above structure is a part of the parent carboxylic acid from which the acid anhydride is obtained. If the acid anhydride is a symmetric one, then the name of acid anhydride is given by replacing the acid in the parent carboxylic acid name with anhydride. If the acid anhydride is an asymmetric one, then the name of acid anhydride is given by using the name of individual carboxylic acid in an alphabetical order followed by anhydride.
(d)
Interpretation:
Condensed structural formula for ethanoic methanoic anhydride has to be drawn.
Concept Introduction:
General structure of acid anhydride can be represented as shown below,
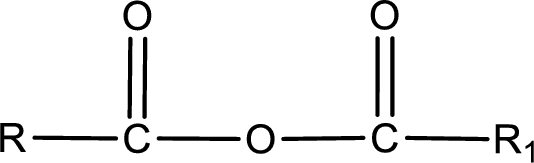
The structure of the acid anhydride can be derived from the IUPAC name or common name. “R, R1” group present in the above structure is a part of the parent carboxylic acid from which the acid anhydride is obtained. If the acid anhydride is a symmetric one, then the name of acid anhydride is given by replacing the acid in the parent carboxylic acid name with anhydride. If the acid anhydride is an mixed one, then the name of acid anhydride is given by using the name of individual carboxylic acid in an alphabetical order followed by anhydride.
Trending nowThis is a popular solution!

Chapter 16 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- للتدة Consider the following compounds. O 2 3 Use the numbers shown to answer the following questions. If none of the structures is applicable enter the word none. An anhydride is represented by the structure numbered O O HC 1 CH3 HC 3 CH3arrow_forwardDraw structural formulas for all of the following. Q.) Alcohols with the molecular formula C4H10Oarrow_forwardDraw the structures of the following acids:(a) 2-Ethyl-3-hydroxyhexanoic acid (b) m-Nitrobenzoic acidarrow_forward
- Determine the weight/volume of the chemicals needed to prepare the following solutions: a) 100 ml of 0.9% (w/v) saline (NaCl) b) 30 ml of 50% glycerol (v/v) c) Electrophoresis requires TAE, which is a specific mixture of Tris base, acetic acid, and EDTA. TAE is normally made as a 50X concentrated stock. Provide a recipe to make 40 ml of 50X TAE. The recipe for one liter of 50X TAE is as follows: 242g Tris base, 57.1 ml glacial acetic acid, 100 ml 0.5 M EDTAarrow_forwardDetermine the pH for the following solutions. {H3O+}=2.4x10^-2 Marrow_forwardScott test is one of the methods used for screening for Cocaine in Forensic Chemistry. The multi step method consists of a) reacting the sample drug with cobaltous thiocyanate (Co(SCN)2) b) dissolving of the formed precipitate by adding HCl and c) extraction of coordination compound with Chloroform d) measurement of absorbance of extracted coordination compound at 627 nm What is the type of chemical equilibria involved in first step of the method? a.Solubility Product equilibria b.Bronsted Lowry acid – Bronsted Lowry base equilibria c.Complex ion formation d.Coordination compound formationarrow_forward
- Name the following compound and indicate whether or not is a reducing sugar:arrow_forwardDraw all possible carboxylic acids with the formula C5H10O2.arrow_forwardIn the reaction between p-aminophenol and acetic anhydride to form acetaminophen, 0.450 mL of water was added. What was the purpose of the water?arrow_forward
- Define the following terms: a. ganglioside b. sphingolipidoses c. isoprenoid d. terpene e. mixed terpenoidarrow_forwardIdentify the functional groups in the following compoundsarrow_forwardThe formula for a sports rub cream is given in the literature as follows:Methylsalicylate 15 mLMenthol 10 gEucalyptus oil 1 gHydrophilic ointment, q.s. ad 100 g If the specific gravity of methyl salicylate is 1.18, how many grams of methylsalicylate would be needed to prepare two dozen 30-g tubes of ointment?arrow_forward
