
Concept explainers
Interpretation:
The number of carbon atoms present in the simplest possible ester has to be given.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
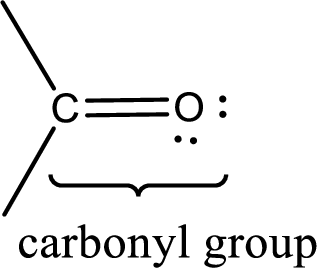
If a hydroxyl group is attached to a carbonyl group means it is known as carboxyl group. This can be represented as shown below,
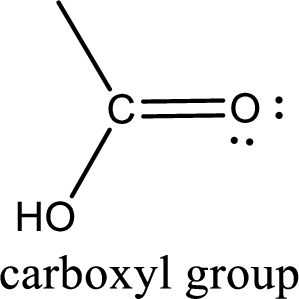
Trending nowThis is a popular solution!

Chapter 16 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- What is the structure of the steroid nucleus found on all sterols? A. It consists of five fused rings, four with six carbons and one with five. B. It consists of three fused rings, two with six carbons and one with five. C. It consists of five fused rings, three with six carbons and two with five. D. It consists of four fused rings, three with six carbons and one with five.arrow_forwardDraw the following compounds: a. α- and β-anomers of D-galactose b. aldonic acid, uronic acid, and aldaric acid derivatives of galactose c. galactitol d. δ-lactone of galactonic acidarrow_forwardThe main difference between a glycerophospholipid and a sphingophospholipid is which component? A. backbone B. phosphate group C. acyl grouparrow_forward
- For D-arabinose: a. Draw its enantiomer. c. Draw a diastereomer that is not an epimer. b. Draw an epimer at C3. d. Draw a constitutional isomer that still contains a carbonyl group.arrow_forwardAfter the removal of all sugar units from a ganglioside, what molecule remains? A. sphingosine B. a ceramide C. a glycerol D. a phosphatearrow_forwardFina How many stereoisomers are possible in Aldopentoses? Select one: a. 6 b. 8 c. 2 d. 4 Whot ie th uinotion and nolycondensaarrow_forward
- What is the molecule below? A. Tetrahydrofolate B. 5-adenosyl methionine C. Pyridoxal phosphate D. Biotinarrow_forwardThe deficiency of which amino acid below will result in the formation of thymine bases that are unable to form a β-glycosidic bond? A. Glu B. Gly C. Asp D. Cysarrow_forwardWhich disaccharide does NOT have an anomeric carbon available for oxidation? O A. Glucose (beta1 1 beta) Galactose O B. Glucose (alpha 14) Galactose OC. Glucose (alpha 1-1 alpha) Galactose O D. Glucose (alpha 1 1 alpha) Mannosearrow_forward
- Given below is the structure of tallose.Answer the following questiona. what is the maximum number of stereoisomers can tallose have?b. how many chiral carbons does it have?c. what is the configuration of tallose is it D or L sugar?d. what type of monosaccharide is it?arrow_forwardWhat is the common structural feature of terpenes? A. All terpenes contain the isoprene unit B. Terpenes function by helping many physiological processes, such as light transducing reactions, absorption of minerals, synthetic reactions, etc. C. Terpenes have the same structure as fatty acids except for the absence of the carboxyl functional group D. Both A and B E. All of these.arrow_forwardWhich functional groups are present in digitoxin? a. To what lipid family does the complex ring drawn to the right of the structure belong? b. Identify the type of glycosidic bond that joins each of the monosaccharide residue in the molecule c. Draw the open form of the monosaccharide used to produce the trisaccharide portion of digitoxin.arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





