
(a)
Interpretation:
Structural formula of the parent acid and parent alcohol for the given ester molecule has to be drawn.
Concept Introduction:
Esters are prepared by condensation of
Esterification reaction is the one in which the carboxylic acid is condensed with an alcohol (or phenol) in presence of strong acid catalyst to produce ester. The general reaction scheme can be given as,
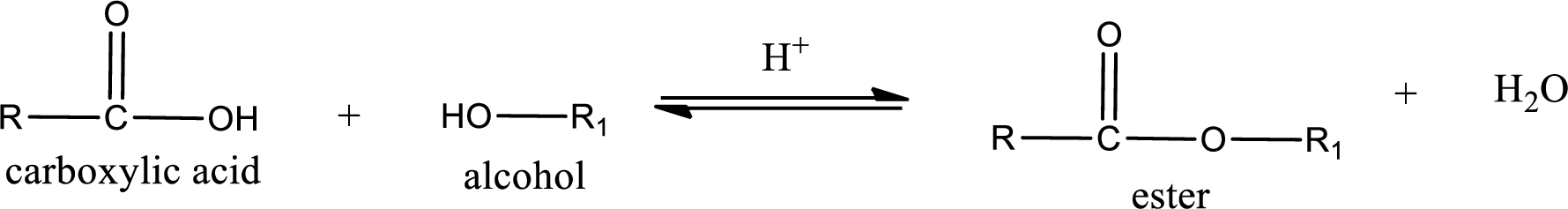
In the above reaction, hydroxyl group is lost from carboxylic acid and hydrogen is lost from alcohol molecule to form water as the byproduct.
(b)
Interpretation:
Structural formula of the parent acid and parent alcohol for the given ester molecule has to be drawn.
Concept Introduction:
Esters are prepared by condensation of carboxylic acid with an alcohol. A molecule of water is lost on this reaction. The reaction that takes place in producing esters is known as esterification reaction.
Esterification reaction is the one in which the carboxylic acid is condensed with an alcohol (or phenol) in presence of strong acid catalyst to produce ester. The general reaction scheme can be given as,
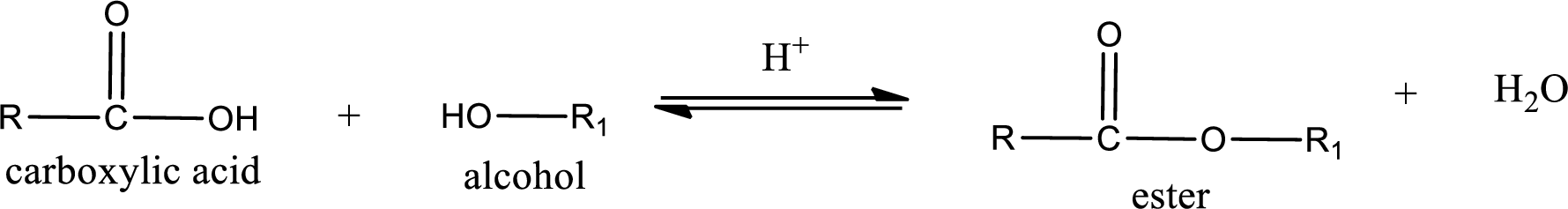
In the above reaction, hydroxyl group is lost from carboxylic acid and hydrogen is lost from alcohol molecule to form water as the byproduct.
(c)
Interpretation:
Structural formula of the parent acid and parent alcohol for the given ester molecule has to be drawn.
Concept Introduction:
Esters are prepared by condensation of carboxylic acid with an alcohol. A molecule of water is lost on this reaction. The reaction that takes place in producing esters is known as esterification reaction.
Esterification reaction is the one in which the carboxylic acid is condensed with an alcohol (or phenol) in presence of strong acid catalyst to produce ester. The general reaction scheme can be given as,
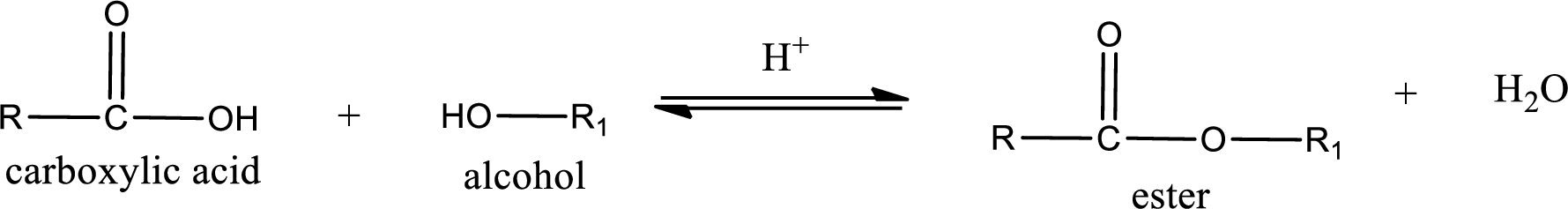
In the above reaction, hydroxyl group is lost from carboxylic acid and hydrogen is lost from alcohol molecule to form water as the byproduct.
(d)
Interpretation:
Structural formula of the parent acid and parent alcohol for the given ester molecule has to be drawn.
Concept Introduction:
Esters are prepared by condensation of carboxylic acid with an alcohol. A molecule of water is lost on this reaction. The reaction that takes place in producing esters is known as esterification reaction.
Esterification reaction is the one in which the carboxylic acid is condensed with an alcohol (or phenol) in presence of strong acid catalyst to produce ester. The general reaction scheme can be given as,
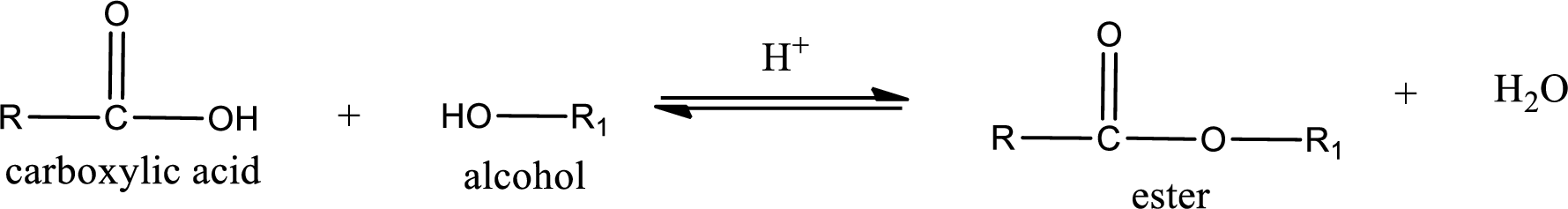
In the above reaction, hydroxyl group is lost from carboxylic acid and hydrogen is lost from alcohol molecule to form water as the byproduct.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- One of the compounds that gives orange oil its unique odor is an ester formed when acetic acid reacts with octan-1-ol. Draw the structure of this ester and name it.arrow_forwardWrite the structure of all organic products formed in the following reaction. NaBH4arrow_forward4. Draw the line structure for the ester that can be formed from the reaction of the acid anhydride and the alcohol given below. :0: :0: HO CH3-CH2-C-o-C-CCH2-CH3 acid anhydride alcoholarrow_forward
- What are the names of the acid and alcohol from which the following ester is formed? acid: alcohol:arrow_forwardH CHỊCHO The IUPAC name for the condensed structural diagram shown above is Select one: O propanoate Ọ propanoic acid FO O ethyl methanoate O methyl ethanoate This organic compound shown in Question 13 is classified as a(n) Select one: Oester O carboxylic acid O halogenated hydrocarbon O alcohol The organic compound shown in Question 13 contains which functional group? Select one: carboxyl Oester linkage O halogen O hydroxyl Select the compound that has the higher boiling point. Select one: O ethanoic acid O pentanoic acid Select the compound that is more soluble in water. Select one: O propyl ethanoate O methyl methanoatearrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





