
Chemistry & Chemical Reactivity
10th Edition
ISBN: 9781337399074
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 98PS
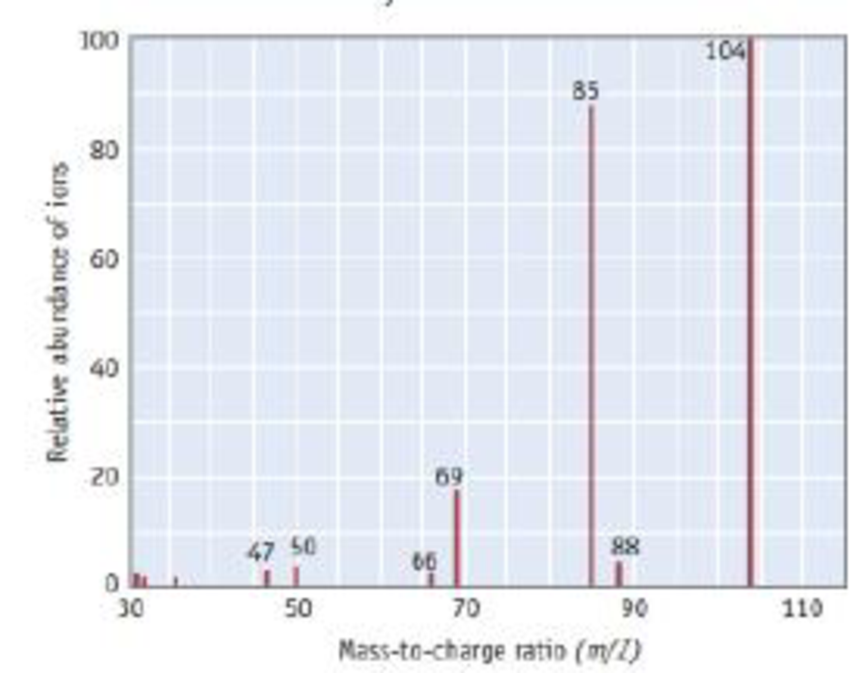
The mass spectrum of phosphoryl chloride. POF3, is illustrated here.
- (a) Identify the cation fragment at a m/Z ratio of 85.
- (b) Identify the cation fragment at a m/Z ratio of 69.
- (c) Which two peaks in the mass spectrum provide evidence that the oxygen atom is connected to the phosphorus atom and is not connected to any of the three fluorine atoms?
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 2 Solutions
Chemistry & Chemical Reactivity
Ch. 2.1 - 1. What is the mass of an iron atom with 30...Ch. 2.2 - Verify that the atomic weight of chlorine is...Ch. 2.2 - Neon has three stable isotopes, one with a small...Ch. 2.5 - Give the number and identity of the constituent...Ch. 2.5 - (a) Write the formulas of all neutral ionic...Ch. 2.6 - What mass of gold, Au, contains 2.6 1024 atoms?Ch. 2.6 - If you have 454 g of citric acid (H3C6H5O7), what...Ch. 2.7 - 1. Express the composition of ammonium carbonate,...Ch. 2.7 - 1. What is the empirical formula of naphthalene,...Ch. 2.7 - Gallium oxide, GaxOy forms when gallium is combine...
Ch. 2.7 - Hydrated nickel(II) chloride is a beautiful green,...Ch. 2.8 - Prob. 2.12CYUCh. 2.8 - Prob. 1.1ACPCh. 2.8 - Prob. 1.2ACPCh. 2.8 - Prob. 2.1ACPCh. 2.8 - Salvarsan was long thought to be a single...Ch. 2.8 - To determine the density of atmospheric nitrogen....Ch. 2.8 - The density of a mixture of gases may be...Ch. 2.8 - Atmospheric argon is a mixture of three stable...Ch. 2.8 - Given that the density of argon is 1.78 g/L under...Ch. 2 - Prob. 1PSCh. 2 - Define mass number. What is the difference between...Ch. 2 - An atom has a very small nucleus surrounded by an...Ch. 2 - A gold atom has a radius of 145 pm. If you could...Ch. 2 - Give the complete symbol(ZAX), including atomic...Ch. 2 - Give the complete symbol(ZAX), including atomic...Ch. 2 - Prob. 7PSCh. 2 - Atomic structure. (a) The synthetic radioactive...Ch. 2 - Prob. 9PSCh. 2 - In 1886 Eugene Goldstein observed positively...Ch. 2 - Marie Curie was born in Poland but studied and...Ch. 2 - Prob. 12PSCh. 2 - The mass of an 16 O atom is 15.995 u. What is its...Ch. 2 - What is the mass of one 16O atom, in grams? (The...Ch. 2 - Cobalt has three radioactive isotopes used in...Ch. 2 - Naturally occurring silver exists as two isotopes...Ch. 2 - Name and describe the composition of the three...Ch. 2 - Which of the following are isotopes of element X,...Ch. 2 - Thallium has two stable isotopes, 203TIand 205Tl....Ch. 2 - Strontium has four stable isotopes. Strontium-84...Ch. 2 - Verify that the atomic weight of lithium is 6.94,...Ch. 2 - Verify that the atomic weight of magnesium is...Ch. 2 - Gallium has two naturally occurring isotopes, 69Ga...Ch. 2 - Europium has two stable isotopes, 151Eu and 153Eu,...Ch. 2 - Titanium and thallium have symbols that are easily...Ch. 2 - In Groups 4A-6A, there are several elements whose...Ch. 2 - How many periods of the periodic table have 8...Ch. 2 - Prob. 28PSCh. 2 - Prob. 29PSCh. 2 - Prob. 30PSCh. 2 - Classify the following elements as metals,...Ch. 2 - Prob. 32PSCh. 2 - Prob. 33PSCh. 2 - Prob. 34PSCh. 2 - What is the charge on the common monatomic ions of...Ch. 2 - What is the charge on the common monatomic ions of...Ch. 2 - Prob. 37PSCh. 2 - Prob. 38PSCh. 2 - When a potassium atom becomes a monatomic ion, how...Ch. 2 - When oxygen and sulfur atoms become monatomic...Ch. 2 - Prob. 41PSCh. 2 - Prob. 42PSCh. 2 - Give the formula and the number of each ion that...Ch. 2 - Give the formula and the number of each ion that...Ch. 2 - Prob. 45PSCh. 2 - Prob. 46PSCh. 2 - Prob. 47PSCh. 2 - Prob. 48PSCh. 2 - Prob. 49PSCh. 2 - Prob. 50PSCh. 2 - Prob. 51PSCh. 2 - Prob. 52PSCh. 2 - Write the formulas for the four ionic compounds...Ch. 2 - Write the formulas for the four ionic compounds...Ch. 2 - Sodium ions, Na+, form ionic compounds with...Ch. 2 - Consider the two ionic compounds NaCl and CaO. In...Ch. 2 - Prob. 57PSCh. 2 - Name each of the following binary, nonionic...Ch. 2 - Prob. 59PSCh. 2 - Prob. 60PSCh. 2 - Calculate the mass, in grams, of each the...Ch. 2 - Calculate the mass, in grams, of each the...Ch. 2 - Calculate the amount (moles) represented by each...Ch. 2 - Calculate the amount (moles) represented by each...Ch. 2 - You are given 1.0-g samples of He, Fe, Li, Si, and...Ch. 2 - You are given 0.10-g samples of K, Mo, Cr, and Al....Ch. 2 - Analysis of a 10.0-g sample of apatite (a major...Ch. 2 - A semiconducting material is composed of 52 g of...Ch. 2 - Calculate the molar mass of each of the following...Ch. 2 - Calculate the molar mass of each of the following...Ch. 2 - Calculate the molar mass of each hydrated...Ch. 2 - Prob. 72PSCh. 2 - What mass is represented by 0.0255 mol of each of...Ch. 2 - Assume you have 0.123 mol of each of the following...Ch. 2 - Sulfur trioxide, SO3, is made industrially in...Ch. 2 - How many ammonium ions and how many sulfate ions...Ch. 2 - Acetaminophen, whose structure is drawn below, is...Ch. 2 - An Alka-Seltzer tablet contains 324 mg of aspirin...Ch. 2 - Calculate the mass percent of each element in the...Ch. 2 - Calculate the mass percent of each element in the...Ch. 2 - Calculate the mass percent of copper in CuS,...Ch. 2 - Calculate the mass percent of titanium in the...Ch. 2 - Succinic acid occurs in fungi and lichens. Its...Ch. 2 - An organic compound has the empirical formula...Ch. 2 - Prob. 85PSCh. 2 - Complete the following table:Ch. 2 - Acetylene is a colorless gas used as a fuel in...Ch. 2 - A large family of boron-hydrogen compounds has the...Ch. 2 - Cumene, a hydrocarbon, is a compound composed only...Ch. 2 - In 2006, a Russian team discovered an interesting...Ch. 2 - Mandelic acid is an organic acid composed of...Ch. 2 - Nicotine, a poisonous compound found in tobacco...Ch. 2 - A compound containing xenon and fluorine was...Ch. 2 - Elemental sulfur (1.256 g) is combined with...Ch. 2 - Epsom salt is used in tanning leather and in...Ch. 2 - You combine 1.25 g of germanium, Ge, with excess...Ch. 2 - The mass spectrum of nitrogen dioxide is...Ch. 2 - The mass spectrum of phosphoryl chloride. POF3, is...Ch. 2 - The mass spectrum of CH3Cl is illustrated here....Ch. 2 - Prob. 100PSCh. 2 - Fill in the blanks in the table (one column per...Ch. 2 - Potassium has three naturally occurring isotopes...Ch. 2 - Crossword Puzzle: In the 2 2 box shown here, each...Ch. 2 - The following chart shows a general decline in...Ch. 2 - Copper atoms. (a) What is the average mass of one...Ch. 2 - Prob. 106GQCh. 2 - Prob. 107GQCh. 2 - Identify two nonmetallic elements that have...Ch. 2 - Prob. 109GQCh. 2 - Prob. 110GQCh. 2 - Prob. 111GQCh. 2 - When a sample of phosphorus burns in air, the...Ch. 2 - Although carbon-12 is now used as the standard for...Ch. 2 - A reagent occasionally used in chemical synthesis...Ch. 2 - Prob. 115GQCh. 2 - Prob. 116GQCh. 2 - Which of the following compounds has the highest...Ch. 2 - Which of the following samples has the largest...Ch. 2 - The structure of one of the bases in DNA, adenine,...Ch. 2 - Prob. 120GQCh. 2 - A drop of water has a volume of about 0.050 mL....Ch. 2 - Capsaicin, the compound that gives the hot taste...Ch. 2 - Prob. 123GQCh. 2 - Write the molecular formula and calculate the...Ch. 2 - Malic acid, an organic acid found in apples,...Ch. 2 - Your doctor has diagnosed you as being anemicthat...Ch. 2 - A compound composed of iron and carbon monoxide,...Ch. 2 - Ma huang, an extract from the ephedra species of...Ch. 2 - Saccharin, a molecular model of which is shown...Ch. 2 - Prob. 130GQCh. 2 - Write the formula for each of the following pounds...Ch. 2 - Complete the table by placing symbols, formulas,...Ch. 2 - Empirical and molecular formulas. (a)...Ch. 2 - Cacodyl, a compound containing arsenic, was...Ch. 2 - The action of bacteria on meat and fish produces a...Ch. 2 - In the laboratory you combine 0.125 g of nickel...Ch. 2 - A compound called MMT was once used to boost the...Ch. 2 - Elemental phosphorus is made by heating calcium...Ch. 2 - Chromium is obtained by heating chromium(III)...Ch. 2 - Stibnite, Sb2S3, is a dark gray mineral from which...Ch. 2 - Direct reaction of iodine (I2) and chlorine (Cl2)...Ch. 2 - In a reaction, 2.04 g of vanadium combined with...Ch. 2 - Iron pyrite, often called fools gold, has the...Ch. 2 - Which of the following statements about 57.1 g of...Ch. 2 - The formula of barium molybdate is BaMoO4. Which...Ch. 2 - A metal M forms a compound with the formula MCl4....Ch. 2 - Pepto-Bismol, which can help provide relief for an...Ch. 2 - The weight percent of oxygen in an oxide that has...Ch. 2 - The mass of 2.50 mol of a compound with the...Ch. 2 - The elements A and Z combine to produce two...Ch. 2 - Polystyrene can be prepared by heating styrene...Ch. 2 - A sample of hemoglobin is found to be 0.335% iron....Ch. 2 - Consider an atom of 64Zn. (a) Calculate the...Ch. 2 - Estimating the radius of a lead atom. (a) You are...Ch. 2 - A piece of nickel foil, 0.550 mm thick and 1.25 cm...Ch. 2 - Uranium is used as a fuel, primarily in the form...Ch. 2 - In an experiment, you need 0.125 mol of sodium...Ch. 2 - Mass spectrometric analysis showed that there are...Ch. 2 - If Epsom salt, MgSO4 x H2O, is heated to 250 C,...Ch. 2 - The alum used in cooking is potassium aluminum...Ch. 2 - Tin metal (Sn) and purple iodine (I2) combine to...Ch. 2 - When analyzed, an unknown compound gave these...Ch. 2 - Two general chemistry students working together in...Ch. 2 - To find the empirical formula of tin oxide, you...Ch. 2 - Prob. 165SCQCh. 2 - Prob. 166SCQCh. 2 - The photo here depicts what happens when a coil of...Ch. 2 - A jar contains some number of jelly beans. To find...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 2.75 Chlorine has only two isotopes, one with mass 35 and the other with mass 37. One is present at roughly 75% abundance, and the atomic weight of chlorine on a periodic table is 35.45. Which must be the correct mass spectrum for chlorine?arrow_forwardThe element bromine is Br2, so the mass of a Br2 molecule is the sum of the mass of its two atoms. Bromine has two isotopes. The mass spectrum of Br2 produces three peaks with relative masses of 157.836, 159.834, and 161.832, and relative heights of 6.337, 12.499. and 6.164, respectively. (a) What isotopes of bromine are present in each of the three peaks? (b) What is the mass of each bromine isotope? (c) What is the average atomic mass of bromine? (d) What is the abundance of each of the two bromine isotopes?arrow_forwardThe mass spectrum of bromine (Br2) consists of three peaks with the following characteristics: Mass (u) Relative Size 157.84 0.2534 159.84 0.5000 161.84 0.2466 How do you interpret these data?arrow_forward
- The mass spectrum of CH3Cl is illustrated here. You know that carbon has two stable isotopes, and 13C with relative abundances of 98.9% and 1.1%, respectively, and chlorine has two isotopes, 35a and 37CI with abundances of 75.77% and 24.23%, respectively. (a) What molecular species gives rise to the lines at m/Z of 50 and 52? Why is the line at 52 about 1/3 the height of the line at 50? (b) What species might be responsible for the line at m/Z = 51?arrow_forwardA cube of sodium has length 1.25 in. How many atoms are in that cube? (Note: dNa=0.968 g/cm3.)arrow_forwardThe mass spectrum of nitrogen dioxide is illustrated here. (a) Identify the cations present for each of the four peaks in the mass spectrum. (b) Does the mass spectrum provide evidence that the two oxygen atoms are attached to a central nitrogen atom (ONO), or that an oxygen atom is at the center (NOO)? Explain.arrow_forward
- Chlorine has two isotopes, Cl-35 and Cl-37. Their abundances are 75.53% and 24.47%, respectively. Assume that the only hydrogen isotope present is H-1. (a) How many different HCI molecules are possible? (b) What is the sum of the mass numbers of the two atoms in each molecule? (c) Sketch the mass spectrum for HCI if all the positive ions are obtained by removing a single electron from an HCI molecule.arrow_forwardGallium arsenide, GaAs, has gained widespread use in semiconductor devices that convert light and electrical signals in fiber-optic communications systems. Gallium consists of 60.% 69Ga and 40.% 71Ga. Arsenic has only one naturally occurring isotope, 75As. Gallium arsenide is a polymeric material, but its mass spectrum shows fragments with the formulas GaAs and Ga2As2. What would the distribution of peaks look like for these two fragments?arrow_forwardStrontium has four stable isotopes. Strontium-84 has a very low natural abundance, but 86Sr, 87Sr, and 88Sr are all reasonably abundant. Knowing that the atomic weight of strontium is 87.62, which of the more abundant isotopes predominates?arrow_forward
- Two elements, R and Q, combine to form two binary compounds. In the first compound, 14.0 g of R combines with 3.00 g of Q. In the second compound, 7.00 g of R combines with 4.50 g of Q. Show that these data are in accord with the law of multiple proportions. If the formula of the second compound is RQ, what is the formula of the first compound?arrow_forward2.92 A candy manufacturer makes chocolate-covered cherries. Although all of the products look roughly the same, 3% of them are missing the cherry. The mass of the candy with a cherry is 18.5 g; those missing the cherry weigh only 6.4 g. (a) How would you compute the average mass of a box of 100 of these chocolate covered cherries from this manufacturer? (b) I low is this question analogous to the determination of atomic weights?arrow_forwardBoron consists of two isotopes, 10B and 11B. Chlorine also has two isotopes, 35Cl and 37Cl. Consider the mass spectrum of BC13. How many peaks would be present, and what approximate mass would each peak correspond to in the BCl3 mass spectrum?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Step by Step Stoichiometry Practice Problems | How to Pass ChemistryMole Conversions Made Easy: How to Convert Between Grams and Moles; Author: Ketzbook;https://www.youtube.com/watch?v=b2raanVWU6c;License: Standard YouTube License, CC-BY