
A fuel cell is an electrochemical device in which hydrogen reacts with oxygen to produce water and DC electricity. A 1-watt proton-exchange membrane fuel cell (PEMFC) could be used for portable applications such as cellular telephones, and a 100-kW PEMFC could be used to power an automobile
The following reactions occur inside the PEMFC:
Anode:
Cathode:
Overall:
A ?owchart of a single cell of a PEMFC is shown below. The complete cell would consist of a stack of such cells in series, such as the one shown in Problem 9.19.
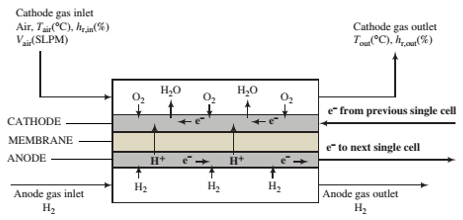
The cell consists of two gas channels separated by a membrane sandwiched between two ?at carbon-paper electrodes—the anode and the cathode—that contain imbedded platinum particles. Hydrogen ?ows into the anode chamber and contacts the anode, where H2molecules are catalyzed by the platinum to dissociate and ionize to form hydrogen ions (protons) and electrons. The electrons are conducted through the carbon ?bers of the anode to an external circuit, where they pass to the cathode of the next cell in the stack. The hydrogen ions permeate from the anode through the membrane to the cathode.
Humid air is fed into the cathode chamber, and at the cathode O2molecules are catalytically split to form oxygen atoms, which combine with the hydrogen ions coming through the membrane and electrons coming from the external circuit to form water. The water desorbs into the cathode gas and is carried out of the cell. The membrane material is a hydrophilic polymer that absorbs water molecules and facilitates the transport of the hydrogen ions from the anode to the cathode. Electrons come from the anode of the cell at one end of the stack and ?ow through an external circuit to drive the device that the fuel cell is powering, while the electrons coming from the device ?ow back to the cathode at the opposite end of the stack to complete the circuit.
It is important to keep the water content of the cathode gas between upper and lower limits. lf the content reaches a value for which the relative humidity would exceed 100%, condensation occurs at the cathode (?ooding), and the entering oxygen must diffuse through a liquid water ?lm before it cart react. The rate of this diffusion is much lower than the rate of diffusion through the gas film normally adjacent to the cathode, and so the performance of the fuel cell deteriorates. On the other hand, if there is not enough water in the cathode gas (less than 85% relative humidity), the membrane dries out and cannot transport hydrogen efficiently, which also leads to reduced performance.
A 400-cell 300—volt PEMFC operates at steady state with a power output of 36 kW. The air fed to the cathode side is at 200°C and roughly 1.0 atm (absolute) with a relative humidity of 70.0% and a volumetric ?ow rate of
(a) Explain in your own words what happens in a single cell of a PEMFC.
(b) The stoichiometric hydrogen requirement for a PEMFC is given by
(c) Use the expression of Part (b) to determine the molar rates of oxygen consumed and water generated in the unit with the given speci?cations, both in units of mol/min. (Remember that power = voltage × current.) Then determine the relative humidity of the cathode exit stream,
(d) Determine the minimum cathode inlet ?ow rate in SLPM to prevent the fuel cell from ?ooding
Learn your wayIncludes step-by-step video

Chapter 6 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Basic Engineering Circuit Analysis
Absolute Java (6th Edition)
Elementary Surveying (14th Edition)
Starting out with Visual C# (4th Edition)
- Another type of battery is the alkaline zinc-mercury cell, in which the cell reaction is Zn(s) + HgO(s) Hg() + ZnO(s) E = + 1.35 V (a) What is the standard free energy change for this reaction? (b) The standard free energy change in a voltaic cell is the maximum electrical energy that the cell can produce. If the reaction in a zinc-mercury cell consumes 1.00 g mercury oxide, what is the standard free energy change? (c) For how many hours could a mercury cell produce a 10-mA current if the limiting reactant is 3.50 g mercury oxide?arrow_forwardCalculate the standard cell potential of the cell corresponding to the oxidation of oxalic acid, H2C2O4, by permanganate ion. MnO4. 5H2C2O4(aq)+2MnO4(aq)+6H+(aq)10CO2(g)+2Mn2+(aq)+8H2O(l) See Appendix C for free energies of formation: Gf for H2C2O4(aq) is 698 kJ.arrow_forwardA voltaic cell is constructed in which one half-cell consists of a silver wire in an aqueous solution of AgNO3.The other half cell consists of an inert platinum wire in an aqueous solution containing Fe2+(aq) and Fe3+(aq). (a) Calculate the cell potential, assuming standard conditions. (b) Write the net ionic equation for the reaction occurring in the cell. (c) Which electrode is the anode and which is the cathode? (d) If [Ag+] is 0.10 M, and [Fe2+] and [Fe3+] are both 1.0 M, what is the cell potential? Is the net cell reaction still that used in part (a)? If not, what is the net reaction under the new conditions?arrow_forward
- Consider the electrolysis of water in the presence of very dilute H2SO4. What species is produced at the anode? Atthe cathode? What are the relative amounts of the speciesproduced at the two electrodes?arrow_forwardAn aqueous solution of an unknown salt of gold is electrolyzed by a current of 2.75 amps for 3.39 hours. The electroplating is carried out with an efficiency of 93.0%, resulting in a deposit of 21.221 g of gold. a How many faradays are required to deposit the gold? b What is the charge on the gold ions (based on your calculations)?arrow_forwardCalcium metal can be obtained by the direct electrolysis of molten CaCl2, at a voltage of 3.2 V. (a) How many joules of electrical energy are required to obtain 12.0 1b of calcium? (b) What is the cost of the electrical energy obtained in (a) if electrical energy is sold at the rate of nine cents per kilowatt hour?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning





