
Concept explainers
(a)
Interpretation:
To estimate the mass and composition (component mass fractions) of each phase of the mixtures in system 1 and in system 2.
Concept introduction:
A ternary mixture is a mixture with combination of water, acetone and methyl isobutyl ketone (MIBK). A system containing ternary mixture is divided into aqueous phase
The behavior of partially miscible three-component systems may be represented on a triangular phase diagram as follows:
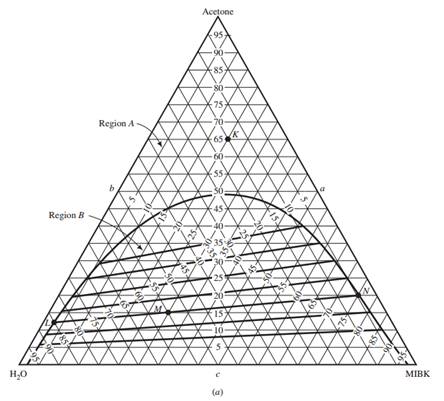
(b)
Interpretation:
To determine the distribution coefficient of acetone in the organic phase relative to the aqueous phase for system-1 and system-2.
Concept introduction:
A ternary mixture is a mixture with combination of water, acetone and methyl isobutyl ketone (MIBK). A system containing ternary mixture is divided into aqueous phase
(c)
Interpretation:
To determine the selectivity,
Concept introduction:
A ternary mixture is a mixture with combination of water, acetone and methyl isobutyl ketone (MIBK). A system containing ternary mixture is divided into aqueous phase
(d)
Interpretation:
To express the selectivity
Concept introduction:
A ternary mixture is a mixture with combination of water, acetone and methyl isobutyl ketone (MIBK). A system containing ternary mixture is divided into aqueous phase
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
- Show that the complete chemical equation, the total ionic equation, and the net ionic equation for the reaction represented by the equation KI(aq)+I2(aq)KI3(aq) give the same expression for the reaction quotient. KI3 is composed of the ions K+ and I3-.arrow_forwardMethanol can be synthesized by means of the equilibriumreaction CO(g)+2H2(g)CH3OH(g) for which the equilibrium constant at 225°C is 6.08103. Assume that the ratio of the pressures of CO(g) and H2(g) is 1:2. What values should they have if the partial pressureof methanol is to be 0.500 atm?arrow_forward1. The decomposition of Cl2Os is allowed to come to equilibrium in a closed container. If the mole fractions are- Cl2:0.243, O2:0.274, Cl2Os:0.483 at equilibrium and the total pressure is 3 atm, what is Ķ»?arrow_forward
- A mixture of 1.00 mol NaHCO;(s) and 1.00 mol Na,CO3(s) is introduced into a 2.50 L flask in which the partial presure of CO, is 2.10 atm and that of H,O (g) is 715 mmHg. When equilibrium is established at 100°C, will the partial pressures of CO(g) and H,0(g) be greater or less than their initial partial pressures? Explain. 2 NaHCO3(s) = Na2CO3(s) + CO2{g) + H2O(g) Kp = 0.23 at 100 °Carrow_forwardBromine monochloride is synthesized using the reaction Br, (g) + Cl, (g) = 2 BrCl(g) K, = 1.1 x 104 at 150 K A 193.0 L flask initially contains 0.956 kg of Br, and 1.014 kg of Cl,. Calculate the mass of BrCI, in grams, that is present i the reaction mixture at equilibrium. Assume ideal gas behavior. 11.45 mass of BrCl: Incorrect What is the percent yield of BrCI? percent yield: 81arrow_forwardFor the reaction CO(g) + 3 H2(g) = H20(g) + CH4(g), Kc = 189 at 1000 K. A vessel is filled with these gases such that the initial concentrations are: [CO] = 0.036 M [H2] = 0.045 %3D [H2O] = 0.020 [CH4] = = 0.031 Is the system at equilibrium? If not, in which direction will a reaction shift to reach equilibrium? toward products because Q = 0.38 toward reactants because Q = 0.24 toward reactants because Q = 61 it is at equilibrium because Q = 189 toward products because Q = 4.1arrow_forward
- Write the expression for the equilibrium constant K, for the following reaction. (Enclose pressures in parentheses and do NOT write the chemical formula as a subscript. For example, enter (PNH, as (P NH3). If either the numerator or denominator is 1, please enter 1.) 2 OF2(g) + 2 F2(g) + 02(g) + K =arrow_forwardFor the reaction PCl5(g) <=> PCl3(g) + Cl2(g), the equilibrium constantKc = 1.1 * 10-2 at 400 K. What is the equilibrium constant for the reactionPCl3(g) + Cl2(g) <=> PCl5(g) at 400 K?arrow_forwardThe equilibrium constant, Ke, for the following reaction is 10.5 at 350 K. 2CH,Cl2(g)CH,(g) + CC14(g) =CH¾(g) + CC1¼(g) Calculate the equilibrium concentrations of reactant and products when 0.374 moles of CH2C1, are introduced into a 1.00 L vessel at 350 K. [CH2C2] =| M [CH4] M [CCL4] M ||arrow_forward
- Suppose 1.000 mol CO and 3.000 mol H2 are putted in a 1.00 L vessel at 1200 k. The equilibrium constant for kc for reverse reaction of CO(g) + 3H2(g) produces CH4(g) + H2O(g) is equals 3.92. find the equilibrium composition of the mixturearrow_forwardSuppose a mixture was prepared containing 0.0100 mol H2, 0.0150 mol F2, and 0.0180 mol HF in a 1.25 L reaction vessel. What will the concentrations of all the gases be when equilibrium is reached? The reaction is 2HF(g) H2(g) + F2(g) for which the equilibrium constant Kc = 1.2 × 10-13. If any value is actually very small, put zero in your answer. Concentration of HF at equilibrium? ____M Concentration of H2 at equilibrium? ___M Concentration of F2 at equilibrium? ___Marrow_forward1. OBJECTIVES Calculate equilibrium constant and the pressure or concentration of reactants or products in an equilibrium mixture II. GENERAL INSTRUCTIONS Read and analyze carefully each problem, Show your complete solution. [Hz] www.the following = 3.7x Whe GAPIDURIVIT 2. N₂(g) + 3H₂(g) + 3. Consider the following reaction where Kc = 3900 at 2000 K 2SO2 (g) + O2(g) = 2503(g) a Write the equilibrium constant expression arearrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,





