
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
4th Edition
ISBN: 9781119192107
Author: BULLARD
Publisher: JOHN WILEY+SONS,INC.-CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 6, Problem 6.79P
A two-unit process is used to separate H2S from a gas containing 96% H2 and 4% H2S by volume. The H2S is absorbed in a solvent, which is then regenerated by air in a stripping column. The Henry’s law constant for the absorption of H2S in the solvent at 0°C is 22 atm/mole fraction.
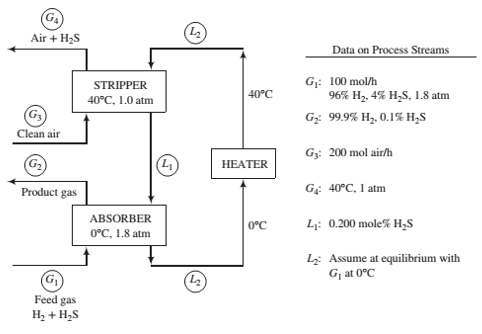
- Brie?y explain in your own words the functions of the three process units. Include in your explanation the purpose of the air in the stripper and the reason the stripper operates at a higher temperature than the absorber.
- Calculate the molar ?ow rate of pure solvent and the volumetric ?ow rate of the gas at G4, neglecting evaporation of solvent in both columns. (See ?owchart.) Exploratory Exercise—Research and Discover (c) The objective of the process described above is to produce puri?ed hydrogen. However, in doing so the process also generates an effluent stream containing H 2S. Identify at least three concerns with simply releasing this stream into the air. Suggest an add-on to the process that allays as many of these concerns as possible. Calculate the mass per 100 mol of G 1 of any reactant required by the add-on. Identify any new concerns created by the add-on.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
4G 72%
4:37
= bartleby
Q&A
Science / Chemistry / Chemic... / 11th Ed...
: A student determined the molar m... A
A student determined the molar mass of an
unknown non-dissociating liquid by the
method described in this experiment. She
found that the equilibrium temperature of a
mixture of ice and pure water was indicated
to be +0.4°C on her thermometer. When she
added 9.9 g of her sample to the mixture, the
temperature, after thorough stirring, fell to
-3.7°C. She then poured off the solution
through a screen into a beaker. The mass of
the solution was 84.2 g.
a. What was the freezing point depression?
°C
b. What was the molality of the unknown
liquid?
m
c. What mass of unknown liquid was in the
decanted solution?
d. What mass of water was in the decanted
solution?
A mixture consisting of 1-mol-% ethane, 5-mol-% propane, 44-mol-% n-butane, and 50-mol-% isobutane is brought to a condition of 294.15 K (21°C) at pressure P. If the molar fraction of the system that is vapor is 0.2, what is pressure P (in bar), and what are the compositions of the vapor and liquid phases?
Consider an ideal mixture of two liquids, A and B. The image below shows the mole fraction of
component A in the vapour plotted against the mole fraction of component A in the solution. The
curve has a slope of 1. Which of the following is necessarily true in such a case?
Mole fraction of A in the solution
O A. A and Bare present in equimolar quantities in the solution.
B. A and B are present in equimolar quantities in the vapour that is in equilibrium with the
solution.
O C. The vapour pressure of A, pa is equal to the vapour pressure of B, p'B.
D. The mole fraction of A in the solution, xA =1
E. The mole fraction of B in the solution, xg = 1
moden au v joue aow
Chapter 6 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Ch. 6 - Ten mL of pure liquid water in a cylinder with a...Ch. 6 - A quantity of methyl acetate is placed in an open,...Ch. 6 - Ethyl alcohol has a vapor pressure of 20.0 mm Hg...Ch. 6 - Prob. 6.4PCh. 6 - Prob. 6.5PCh. 6 - Prob. 6.6PCh. 6 - Prob. 6.7PCh. 6 - Prob. 6.8PCh. 6 - Prob. 6.9PCh. 6 - Prob. 6.10P
Ch. 6 - Prob. 6.11PCh. 6 - Prob. 6.12PCh. 6 - Prob. 6.13PCh. 6 - Air at 50% relative humidity is cooled...Ch. 6 - Prob. 6.15PCh. 6 - Prob. 6.16PCh. 6 - Air at 90°C and 1.00 atm (absolute) contains 10.0...Ch. 6 - When fermentation units are operated with high...Ch. 6 - When you step out of a shower, the temperature in...Ch. 6 - A fuel cell is an electrochemical device in which...Ch. 6 - Prob. 6.21PCh. 6 - Prob. 6.22PCh. 6 - Prob. 6.23PCh. 6 - Prob. 6.24PCh. 6 - Prob. 6.25PCh. 6 - Prob. 6.26PCh. 6 - Prob. 6.27PCh. 6 - Prob. 6.28PCh. 6 - An air conditioner is designed to bring 10.000...Ch. 6 - Prob. 6.30PCh. 6 - Prob. 6.31PCh. 6 - Prob. 6.32PCh. 6 - A gas stream containing 40.0 mole% hydrogen, 35.0%...Ch. 6 - Prob. 6.34PCh. 6 - In the manufacture of an active pharmaceutical...Ch. 6 - Prob. 6.36PCh. 6 - In the ?nal stage of the manufacturing process...Ch. 6 - Prob. 6.38PCh. 6 - A fuel gas containing methane and ethane is burned...Ch. 6 - A mixture of propane and butane is burned with...Ch. 6 - An important parameter in the design of gas...Ch. 6 - A liquid stream consisting of 12.5 mole% n-butane...Ch. 6 - Nitric acid is a chemical intermediate primarily...Ch. 6 - Prob. 6.44PCh. 6 - Sulfur trioxide (SO3) dissolves in and reacts with...Ch. 6 - State whether you would use Raoult’s law or Henrys...Ch. 6 - A gas containing nitrogen, benzene, and toluene is...Ch. 6 - Prob. 6.48PCh. 6 - Prob. 6.49PCh. 6 - A conelation for methane solubility in...Ch. 6 - Prob. 6.51PCh. 6 - The constituent partial pressures of a gas in...Ch. 6 - Prob. 6.53PCh. 6 - Prob. 6.54PCh. 6 - Prob. 6.55PCh. 6 - Prob. 6.56PCh. 6 - Prob. 6.57PCh. 6 - Prob. 6.58PCh. 6 - Nitrogen is bubbled through a liquid mixture that...Ch. 6 - Prob. 6.60PCh. 6 - Prob. 6.61PCh. 6 - Prob. 6.62PCh. 6 - The feed to a distillation column (sketched below)...Ch. 6 - Prob. 6.64PCh. 6 - Prob. 6.65PCh. 6 - Prob. 6.66PCh. 6 - Prob. 6.67PCh. 6 - Prob. 6.68PCh. 6 - Prob. 6.69PCh. 6 - Prob. 6.70PCh. 6 - A methanol-water feed stream is introduced to a...Ch. 6 - Prob. 6.72PCh. 6 - In this problem you will use a spreadsheet to...Ch. 6 - Prob. 6.74PCh. 6 - Prob. 6.75PCh. 6 - Prob. 6.76PCh. 6 - Acetaldehyde is synthesized by the catalytic...Ch. 6 - Dehydration of natural gas is necessary to prevent...Ch. 6 - A two-unit process is used to separate H2S from a...Ch. 6 - Prob. 6.80PCh. 6 - Prob. 6.81PCh. 6 - Prob. 6.82PCh. 6 - Prob. 6.83PCh. 6 - A solution containing 100 lbm KNO3/100 Ibm H2O at...Ch. 6 - A 10.0 wt% aqueous solution of sodium chloride is...Ch. 6 - Potassium dichromate (K2Cr2O7) is to be recovered...Ch. 6 - Prob. 6.87PCh. 6 - Prob. 6.88PCh. 6 - Sodium bicarbonate is synthesized by reacting...Ch. 6 - An ore containing 90 wt% MgSO4(H2O and the balance...Ch. 6 - An aqueous waste stream leaving a process contains...Ch. 6 - A solution of diphenyl (MW = 154.2) in benzene is...Ch. 6 - An aqueous solution of urea (MW = 60.06) freezes...Ch. 6 - Prob. 6.94PCh. 6 - Derive Equation 6.54 for the boiling-point...Ch. 6 - Prob. 6.96PCh. 6 - A stream of 5.00 wt% oleic acid in cottonseed oil...Ch. 6 - Benzene and hexane are being considered as...Ch. 6 - Acetone is lo be extracted with n-hexane from a...Ch. 6 - Prob. 6.100PCh. 6 - Prob. 6.101PCh. 6 - Five kilograms of a 30 wt% acetone70% water...Ch. 6 - An aqueous acetone solution is fed at a rate of...Ch. 6 - Prob. 6.104PCh. 6 - Prob. 6.105PCh. 6 - Air at 25°C and 1 atm with a relative humidity of...Ch. 6 - Prob. 6.107PCh. 6 - Prob. 6.108PCh. 6 - Various amounts of activated carbon were added to...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Consider the following system of gas-phase reactions:
B is the desired product, and X and Y are foul pollutant...
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Which solutions of the following equations will exhibit convergent behavior? Which are oscillatory? Assume zero...
Process Dynamics and Control, 4e
What will the following code display? enum Letters { Z, Y, X }; if (Z X) cout Z is greater than X. \n; else c...
Starting Out with C++ from Control Structures to Objects (8th Edition)
Write statements that can be used in a Java program to read your age, as entered at the keyboard, and display i...
Java: An Introduction to Problem Solving and Programming (7th Edition)
1-1. The floor of a heavy storage warehouse building is made of 6-in.-thick stone concrete. If the floor is a s...
Structural Analysis (10th Edition)
16. In a simple electric circuit, the current (I) must remain below 40 milliarr.ps (I < 40 mA) and must also sa...
Thinking Like an Engineer: An Active Learning Approach (4th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the free energy of mixing for a mixture of 10mol hydrogen gas and 20mol of helium gas at 50degC. Assume ideal solution. -16711.8457 cal -12261.1543 cal -16171.8457 cal -12621.1543 calarrow_forwardA mixture of A and B has a liquid-vapor phase diagram as shown in the picture which is not ideal. Assume that 1.5 mol of A and 3.5 mol of B were mixed in an isothermal vessel. Then equilibration was done, and the total vapor pressure was determined to be 50.0 torr. Solve for the number of moles of B that is present in the vapor phase. Choose your answer.a. 1.43 mol b. 1.07 mol c. 1.82 mol d. 2.43 molarrow_forwardA mixture of 87.3 mole % methanol and 12.7 mole % isopropyl alcohol is vaporized at 101.325 kPa (absolute) until 900 moles of vapor and 100 moles of liquid in equilibrium with each other are produced. This occurs in a single-stage system and the vapor and liquid are kept in contact until equilibrium is achieved. Calculate the composition of the vapor and liquid. Note: Assume the system exhibits ideal behavior (which it mostly does), e.g. Raoult’s Law applies and an Antoine’s Vapor Pressure correlation is a sufficiently accurate model of component vapor pressure.arrow_forward
- 3. The second Virial coefficient of an equimolar binary vapor mixture of methane and n-hexane is -517 cm'/mol at 50°C. What is B at the same temperature for a mixture containing 25 mol% methane and 75 mol% n-hexane. At 50°C B =-33 cm'/mol for methane, B =-1512cm'/mol for n-hexane.arrow_forwardThe table below shows temperature/composition data collected for a mixture of methylbenzene (M) and octane (O) at 1 atm. Recall that x stands for the mole fraction in the liquid and y stands for the mole fraction in the vapor in equilibrium. The boiling points for methylbenzene (M) and octane (O) are 110.60C and 125.60C, respectively. Construct the phase diagram with Temperature vs. xM. What is the composition of the vapor in equilibrium with the liquid of composition (a) xM = 0.250 and (b) xO = 0.250. T (0C) 110.9 112.0 114.0 115.8 117.3 119.0 121.1 123.0 xM 0.908 0.795 0.615 0.527 0.408 0.300 0.203 0.097 yM 0.923 0.836 0.698 0.624 0.527 0.410 0.297 0.164arrow_forward1. Sodium carbonate is recovered as decahydrate from 1000kg of 5% solution of sodium carbonate in water. If it is desired that 95% of Na2CO3 is to be recovered as decahydrate by cooling to 278 K, determine the following: a) the mass of water evaporated (b) the mass of crystals obtained (Na2CO3 ∙ 10H2O). The solubility of Na2CO3 in water at 278K is 9.0% (weight) 2. A copra batch contains 44% oil and is to be extracted with n-hexane. The extracted meal contains 0.07 kg of oil/kg oil free matter and the mass fraction of n-hexane in it is 0.05. The extract contains 49% n- hexane. If 10,000 kg of copra are to be processed, how many kg of n-hexane are required and the determine the amount of extract. 3. Absorption of sulphur dioxide is carried out in a packed tower. Gas containing 14.8% sulphur dioxide and the rest inert gases enters the tower while the leaving gases contains 1% sulphur dioxide. Water flows at the rate of 1m3/min. The tower handles 7380 m3/hr of gas at 303K and 1 bar. Find…arrow_forward
- Pure liquid A has vapor pressure 200. bar and pure liquid B has vapor pressure 100. bar. In an AB mixture with mole fraction X = 0.500, compute the mole fraction of A in the vapor.arrow_forward6. Oil reservoirs below ground frequently are in contact with underground water and, in connection with an oil drilling operation, you are asked to compute the solubility of water in a heavy oil at the underground conditions. These conditions are estimated to be 140°C and 410 bar. Experiments at 140°C and 1 bar indicate that the solubility of steam in the oil is x = 35 x 10-4 (x, is the mole fraction of steam). Assume Henry's law in the form f = H(T)x1, where H(T) is a constant, dependent only on the temperature, and f is the fu- gacity of H,O. Also assume that the vapor pressure of the oil is negligible at 140°C. Data for H,0 are given in the steam tables.arrow_forwardA mixture of phenol and water, under certain conditions of temperature and composition, forms two separate liquid phases, one rich in phenol and the other rich in water. At 30⁰ the compositions of the upper and lower layers are 70% and 9% by mass phenol, respectively. If 40kg of phenol and 60kg of water are mixed and the layers are allowed to separate at 30⁰C, what will be the weight of the two layers?arrow_forward
- The binary water-phenol diagram is given below, in molar fraction, under the total pressure Pt = 1 atm. 1) How many phases are there in the domain marked "I"? 2) From which temperature the mixture become miscible in all proportions? How do you call that temperature? 3) At a temperature T = 50 °C, in a controlled temperature container containing water. Phenol is then poured at the same temperature until the molar fraction of phenol in the mixture XC2H50H is equal to 0.05. Then, we continue to add the phenol gradually, at a constant temperature, to XC2H5OH of phenol in the mixture is XC2H5OH = 0.8. = 0.4. Then finally we stop adding phenol when the molar fraction a. How many phases are present at each one of the three mixture compositions in the container during the addition of the phenol? b. What is the composition of the phases when the fraction of phenol is XC2H5OH = 0.4? c. What is the proportions of the phases with respect to each other when XC2H5OH = 0.4? (Show your calculations)…arrow_forwardThe following temperature/composition data were obtained for a mixture of two liquids A and B at 1 atm, where x is the mole fraction in the liquid and y is the mole fraction in the vapour at equilibrium. T (°C) 110 120 130 140 XA 0.73 0.43 0.25 0.10 YA 0.96 0.78 0.60 0.36 The boiling points are 105°C for A and 150°C for B. What is the composition of the vapour in equilibrium with the liquid of composition (a) Xa = 0.35 and (b) xB = 0.25?arrow_forwardCalculate the following: (a) The bubble-point temperature of an equimolar mixture of liquid n-hexane and n-heptane at 1.0 atm and the composition (mole fractions) of the vapor in equilibrium with this mixture. (b) The dew-point temperature of a gas mixture with a molar composition of 30% n-hexane, 30% n-heptane, and 40% air at 1 atm and the composition (mole fractions) of the liquid in equilibrium with this mixture.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Mod-01 Lec-23 Degrees of freedom analysis; Author: nptelhrd;https://www.youtube.com/watch?v=c4h85JjrkzQ;License: Standard YouTube License, CC-BY
Introduction to Degrees of Freedom; Author: LearnChemE;https://www.youtube.com/watch?v=tW1ft4y5fQY;License: Standard Youtube License