
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
4th Edition
ISBN: 9781119192107
Author: BULLARD
Publisher: JOHN WILEY+SONS,INC.-CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 6, Problem 6.101P
Interpretation Introduction
Interpretation:
To estimate the composition and mass of all phases of the mixture.
Concept introduction:
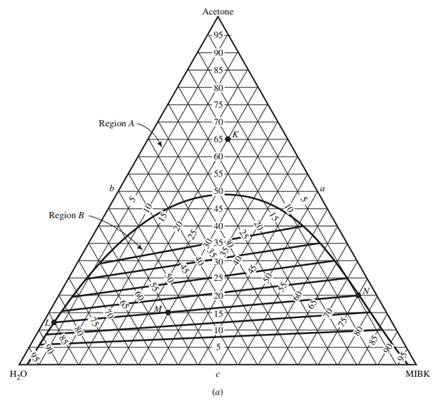
The behavior of partially miscible three-component systems may be represented on a triangular phase diagram which is as follows:
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Calculate the percentage change in Kx for the reaction CH3OH(g) + NOCl(g) ⇋ HCl(g) + CH3NO2(g) when the total pressure is increased from 1.0 bar to 2.0 bar at constant temperature.
A mixture of isobutylene ((CH3)2CCH2, 0.400 bar partial pressure) and
HCI (0.600 bar partial pressure) is heated at 500.0 K. The equilibrium
constant K for the gas-phase thermal decomposition of tert-butyl chloride
((CH3)3CCI) is 3.45 at 500.0 K.
(CH3);CCI(g) = (CH3),CCH2(g) + HCI(g)
Based on your ICE table, set up the expression for K for the
decomposition of (CH3);CCI.
(CH3);CCI(g) = (CH3)2CCH2(g) + HCI(g)
1
Do not combine or simplify terms.
K =
3.45
5 RESET
(P(CH);CCH;)
(P(CH,);CCH,)?
(PHCI)
(Рнс)
(P(CH,),cCi)
(P(CH,),CCI)?
2(Рнс)
2(P(CH3),CCH;)
2(P(CH,),CCI)
2(P(CH,),CCI)?
2(Рнс)?
2(P(CH3),C=CH,)?
6.) Since all the products have the same molecular weight, you can closely approximate the density of
the product mixture by averaging the densities of the products. Using the average product density,
determine the theoretical yield in mL when 1.5 mL of 3,3-dimethyl-2-butanol and 7 drops of H2SO4
(conc) are heated together at 70°C.
Chapter 6 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Ch. 6 - Ten mL of pure liquid water in a cylinder with a...Ch. 6 - A quantity of methyl acetate is placed in an open,...Ch. 6 - Ethyl alcohol has a vapor pressure of 20.0 mm Hg...Ch. 6 - Prob. 6.4PCh. 6 - Prob. 6.5PCh. 6 - Prob. 6.6PCh. 6 - Prob. 6.7PCh. 6 - Prob. 6.8PCh. 6 - Prob. 6.9PCh. 6 - Prob. 6.10P
Ch. 6 - Prob. 6.11PCh. 6 - Prob. 6.12PCh. 6 - Prob. 6.13PCh. 6 - Air at 50% relative humidity is cooled...Ch. 6 - Prob. 6.15PCh. 6 - Prob. 6.16PCh. 6 - Air at 90°C and 1.00 atm (absolute) contains 10.0...Ch. 6 - When fermentation units are operated with high...Ch. 6 - When you step out of a shower, the temperature in...Ch. 6 - A fuel cell is an electrochemical device in which...Ch. 6 - Prob. 6.21PCh. 6 - Prob. 6.22PCh. 6 - Prob. 6.23PCh. 6 - Prob. 6.24PCh. 6 - Prob. 6.25PCh. 6 - Prob. 6.26PCh. 6 - Prob. 6.27PCh. 6 - Prob. 6.28PCh. 6 - An air conditioner is designed to bring 10.000...Ch. 6 - Prob. 6.30PCh. 6 - Prob. 6.31PCh. 6 - Prob. 6.32PCh. 6 - A gas stream containing 40.0 mole% hydrogen, 35.0%...Ch. 6 - Prob. 6.34PCh. 6 - In the manufacture of an active pharmaceutical...Ch. 6 - Prob. 6.36PCh. 6 - In the ?nal stage of the manufacturing process...Ch. 6 - Prob. 6.38PCh. 6 - A fuel gas containing methane and ethane is burned...Ch. 6 - A mixture of propane and butane is burned with...Ch. 6 - An important parameter in the design of gas...Ch. 6 - A liquid stream consisting of 12.5 mole% n-butane...Ch. 6 - Nitric acid is a chemical intermediate primarily...Ch. 6 - Prob. 6.44PCh. 6 - Sulfur trioxide (SO3) dissolves in and reacts with...Ch. 6 - State whether you would use Raoult’s law or Henrys...Ch. 6 - A gas containing nitrogen, benzene, and toluene is...Ch. 6 - Prob. 6.48PCh. 6 - Prob. 6.49PCh. 6 - A conelation for methane solubility in...Ch. 6 - Prob. 6.51PCh. 6 - The constituent partial pressures of a gas in...Ch. 6 - Prob. 6.53PCh. 6 - Prob. 6.54PCh. 6 - Prob. 6.55PCh. 6 - Prob. 6.56PCh. 6 - Prob. 6.57PCh. 6 - Prob. 6.58PCh. 6 - Nitrogen is bubbled through a liquid mixture that...Ch. 6 - Prob. 6.60PCh. 6 - Prob. 6.61PCh. 6 - Prob. 6.62PCh. 6 - The feed to a distillation column (sketched below)...Ch. 6 - Prob. 6.64PCh. 6 - Prob. 6.65PCh. 6 - Prob. 6.66PCh. 6 - Prob. 6.67PCh. 6 - Prob. 6.68PCh. 6 - Prob. 6.69PCh. 6 - Prob. 6.70PCh. 6 - A methanol-water feed stream is introduced to a...Ch. 6 - Prob. 6.72PCh. 6 - In this problem you will use a spreadsheet to...Ch. 6 - Prob. 6.74PCh. 6 - Prob. 6.75PCh. 6 - Prob. 6.76PCh. 6 - Acetaldehyde is synthesized by the catalytic...Ch. 6 - Dehydration of natural gas is necessary to prevent...Ch. 6 - A two-unit process is used to separate H2S from a...Ch. 6 - Prob. 6.80PCh. 6 - Prob. 6.81PCh. 6 - Prob. 6.82PCh. 6 - Prob. 6.83PCh. 6 - A solution containing 100 lbm KNO3/100 Ibm H2O at...Ch. 6 - A 10.0 wt% aqueous solution of sodium chloride is...Ch. 6 - Potassium dichromate (K2Cr2O7) is to be recovered...Ch. 6 - Prob. 6.87PCh. 6 - Prob. 6.88PCh. 6 - Sodium bicarbonate is synthesized by reacting...Ch. 6 - An ore containing 90 wt% MgSO4(H2O and the balance...Ch. 6 - An aqueous waste stream leaving a process contains...Ch. 6 - A solution of diphenyl (MW = 154.2) in benzene is...Ch. 6 - An aqueous solution of urea (MW = 60.06) freezes...Ch. 6 - Prob. 6.94PCh. 6 - Derive Equation 6.54 for the boiling-point...Ch. 6 - Prob. 6.96PCh. 6 - A stream of 5.00 wt% oleic acid in cottonseed oil...Ch. 6 - Benzene and hexane are being considered as...Ch. 6 - Acetone is lo be extracted with n-hexane from a...Ch. 6 - Prob. 6.100PCh. 6 - Prob. 6.101PCh. 6 - Five kilograms of a 30 wt% acetone70% water...Ch. 6 - An aqueous acetone solution is fed at a rate of...Ch. 6 - Prob. 6.104PCh. 6 - Prob. 6.105PCh. 6 - Air at 25°C and 1 atm with a relative humidity of...Ch. 6 - Prob. 6.107PCh. 6 - Prob. 6.108PCh. 6 - Various amounts of activated carbon were added to...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The decomposition reaction, C(9) = A(g) + 2B(g) has Ke 26 at 298 K. What is the value of K, for this system at 298 K? K, = | %3!arrow_forwardSolid NH4I ammonium iodide (which is always considered to be in excess) is introduced into an initially empty container at a constant temperature of 427°C. An equilibrium is quickly established and the total pressure (PT) is then 0.9 bar. NHẠI (s) 2 NH3(g) + HI(g) Express the equilibrium constant K' of this reaction as a function of PT a. Express the equilibrium constant K' of this reaction as a function of PT- The answer is PNH, PHI - - ( 2 2 P K' || po2 p°2 2P°, Can you explain why we get (p/2)^2?arrow_forwardA mixture of isobutylene ((CH3)2CCH2, 0.400 bar partial pressure) and HCI (0.600 bar partial pressure) is heated at 500.0 K. The equilibrium constant K for the gas- phase thermal decomposition of tert- butyl chloride ((CH3)3CCI) is 3.45 at 500.0 K. bar (CH3);CCI(g) = (CH3)2CCH2(g) + HCI(g) 1 3 What is the partial pressure of isobutylene ((CH3)2CCH2) at equilibrium? 4 C 8. 9. +/- х 100arrow_forward
- A mixture of isobutylene ((CH3)2CCH2, 0.400 bar partial pressure) and HCI (0.600 bar partial pressure) is heated at 500.0 K. The equilibrium constant K for the gas- phase thermal decomposition of tert- butyl chloride ((CH3)3CCI) is 3.45 at 500.0 K. bar (CH3);CCI(g) = (CH3)2CCH2(g) + HCI(g) 1 3 What is the partial pressure of hydrochloric acid (HCI) at equilibrium? 4 C 8. 9. +/- х 100arrow_forwardA mixture of isobutylene ((CH3)2CCH2, 0.400 bar partial pressure) and HCI (0.600 bar partial pressure) is heated at 500.0 K. The equilibrium constant K for the gas- phase thermal decomposition of tert- butyl chloride ((CH3)3CCI) is 3.45 at 500.0 K. bar (CH3);CCI(g) = (CH3)2CCH2(g) + HCI(g) 1 3 You have calculated the equilibrium partial pressures to be: 4 C tert-butyl chloride,CH3);CCI = (x) hydrochloric acid, HCI = (0.600 - x) isobutylene, (CH3)2CCH2 = (0.400 - x) What is the partial pressure of tert- butyl chloride ((CH3)3CCI) at equilibrium? 8. 9. +/- х 100arrow_forwardFor the reaction PCl5(g) <=> PCl3(g) + Cl2(g), the equilibrium constantKc = 1.1 * 10-2 at 400 K. What is the equilibrium constant for the reactionPCl3(g) + Cl2(g) <=> PCl5(g) at 400 K?arrow_forward
- Using the Kf and Kb equations with electrolytes A certain liquid X has a normal boiling point of 110.90 °C and a boiling point elevation constant K=1.66 °C-kg mol -1 .A solution is prepared by dissolving some ammonium sulfate ((NH SO,) in 150.g of X. This solution boils at 112.2 °C. Calculate the mass of (NH), SO, that was dissolved. Round your answer to 2 significant digits. ? Explanation Check ©2022 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center | étv Aarrow_forwardA mixture of isobutylene ((CH3)2CCH2, 0.400 bar partial pressure) and HCI (0.600 bar partial pressure) is heated at 500.0 K. The equilibrium constant K for the gas- phase thermal decomposition of tert- butyl chloride ((CH3);CCI) is 3.45 at 500.0 K. (CH3);CCI(g) = (CH3)2CCH2(g) + HCI(g) 1 3 Calculate the value of Kc 4 6. C 7 8 9 +/- х 100arrow_forwardThe following system is at equilibrium in a closed vessel: 2 NH3(g) + 3 I2(g) ⇆ N2(g) + 6 HI(g) Various stresses are applied to the system as illustrated in the chart. Drag the appropriate label from the list below that indicates how the system responds to each stress. Each label can be used more than once.arrow_forward
- You are a NASA engineer. You are the chief engineer for the Apollo 13 mission to the moon. The astronauts are running out of oxygen and need to get rid of the excess carbon dioxide. You know that sodium hydroxide has been suggested as a means of removing carbon dioxide from the spacecraft cabin. The filter which they had been using is fully saturated and no longer works. You remember that the astronauts have a 3,000 g container of sodium hydroxide (NaOH) on the ship. You also know that sodium hydroxide can be used to remove carbon dioxide according to the following reaction: NaOH + co,-→ Na,Co, + H,0 How many grams CO, can be removed from the ship with the 3,000 g of NaOH? - The astronauts have 2 days left before they land on earth. You know that the astronauts will emit roughly 1,000 grams of CO, each day. Is there enough sodium hydroxide in the cabin to cleanse the cabin air of the carbon dioxide, or are the astronauts doomed? Again be sure to show all your work.arrow_forwardThe production of 1,3-butadiene can be carried out by the dehydrogenation of n-butane: C 4H 10 (9) H 2C=CHHC=CH 2 (g) + 2 H 2 (g) The reaction is carried out at 925 K and 0.5 bar pressure. Starting with pure n-butane gas it comes to equilibrium. At 925 K the equilibrium constant (K) of the reaction is 0.30. Assuming ideal gases, calculate the fractional conversion of n-butane at equilibrium. 0.29 0.41 0.52 0.63 0.71 0.85 OOarrow_forwardConsider the following reactions, write the expressions for Kc and Kp and determine their units. Unit for concentration should be mol/dm3 and for pressure kPa. d) Ca3(PO4)2 (s) = 3 Ca²*(aq) + 2 PO4³ (aq) e) 2C2H6(g) + 7O2(g) = 4CO2(g) + 6H2O (g)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning