
Dehydration of natural gas is necessary to prevent the formation of gas hydrates, which can plug valves and other components of a gas pipeline, and also to reduce potential corrosion problems. Water removal can be accomplished as shown in the following schematic diagram:
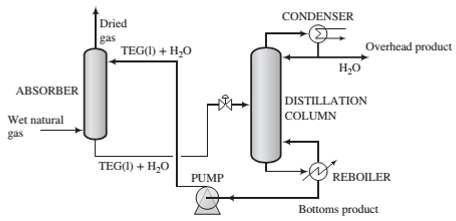
Natural gas containing
(a) Draw and completely label a ?owchart of this process. Calculate the mass ?ow rate (lbm/day) and volumetric ?ow rate (ft3/day) of the overhead product from the distillation column.
(b) The greatest possible amount of dehydration is achieved if the gas leaving the absorption column is in equilibrium with the solvent entering the column. If the Henry's law constant for water in TEG at 90°F is 0.398 psia/mol fraction, what is the maximum allowable mole fraction of water in the solvent fed to the absorber‘?
(c) A column of in?nite height would be required to achieve equilibrium between the gas and liquid at the top of the absorber. For the desired separation to be achieved in practice, the mole fraction of water in the entering solvent must be less than the value calculated in Part (b). Suppose it is 80% of that value and the ?ow rate of TEG in the recirculating solvent is 37 lbmTEG/lbmwater absorbed in the column. Calculate the ?ow rate (lbm/day) of the solvent stream entering the absorber and the mole fraction of water in the solvent stream leaving the absorber.
(d) What is the purpose of the distillation column in the process? (Hint: Think about how the process would operate without it.)
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Process Dynamics and Control, 4e
Starting Out with C++ from Control Structures to Objects (9th Edition)
Starting Out with Java: From Control Structures through Objects (7th Edition) (What's New in Computer Science)
Starting Out with Programming Logic and Design (4th Edition)
Management Information Systems: Managing the Digital Firm (15th Edition)
- 6-35 Describe how we would prepare the following solutions: (a) 280. mL of a 27% v/v solution of ethanol C2H6O, in water (b) 435 mL of a 1.8% v/v solution of ethyl acetate, C4H8O2 in water (c) 1.65 L of an 8.00% v/v solution of benzene C6H6, in chloroform, CHCI3arrow_forwardA sulfuric acid spill occurred on the highway after a truck containing barrels of sulfuric acid swerved out of control from a truck tire blowout. Luckily no one got hurt in the event but there is a concern that the sulfuric acid spill may affect local ground water supply. A sample of the ground water in that area was recovered to assess the extent of contamination of the ground water supply. In order to determine the concentration of sulfuric acid in the sample, 25.00 mL of the ground water sample was titrated with 0.133 M NaOH and you get the following data: Sodium hydroxide (initial buret reading) 0.66 mL Sodium hydroxide (final buret reading) 17.42 mL Calculate the molarity of sulfuric acid in the ground water sample. Enter numerical answer. Report in 3 sig figs. Show workarrow_forwardFollowing the irrigation of salted soils, 10,000 L of salty irrigation runoff was filtered by a reverse osmosis (RO) treatment plant generating 7,000 L of pure water for return to a local river. Following RO treatment, the brine (left-over salt solution) concentration was 1.95 M NaCl. What was the hydrostatic pressure (atm) required by the RO plant to filter this volume of the salty irrigation runoff at a temperature of 20.0 oC?arrow_forward
- You are a NASA engineer. You are the chief engineer for the Apollo 13 mission to the moon. The astronauts are running out of oxygen and need to get rid of the excess carbon dioxide. You know that sodium hydroxide has been suggested as a means of removing carbon dioxide from the spacecraft cabin. The filter which they had been using is fully saturated and no longer works. You remember that the astronauts have a 3,000 g container of sodium hydroxide (NaOH) on the ship. You also know that sodium hydroxide can be used to remove carbon dioxide according to the following reaction: NaOH + co,-→ Na,Co, + H,0 How many grams CO, can be removed from the ship with the 3,000 g of NaOH? - The astronauts have 2 days left before they land on earth. You know that the astronauts will emit roughly 1,000 grams of CO, each day. Is there enough sodium hydroxide in the cabin to cleanse the cabin air of the carbon dioxide, or are the astronauts doomed? Again be sure to show all your work.arrow_forwardThe calcium (AW= 40.08 g/mol) from a sample of limestone weighing 607.4 mg was precipitated as calcium oxalate hydrate (CaC204.H20, FW 146.12 g/mol) and ignited to calcium carbonate (CaCO3, FW= 100.09 g/mol) weighing 246.7 mg. (c) If the precipitate had been ignited at a higher temperature giving calcium oxide (CaO, FW= 56.08 g/mol), what would be its weight?arrow_forward25.0mL of a 0.515 M K2S solution is mixed with 30.0 mL of 0.833 M HNO3 acid solution to give the following reaction: K2S(aq) + 2HNO3(aq) → 2KNO3(aq) + H2S(g) H2S is an unwanted by-product in a pulp and paper industry. To capture H2S gas, it is bubbled through a NaOH solution to produce Na2S with a yield of 94%. H2S(g) + 2NaOH (aq) → Na2S (aq) + 2H2O(l) The mass of H2S(g) that was processed (in kg) if 10.76 kg of Na2S was collected isarrow_forward
- (ans: -7.50 x 10 ) one mole of sucrose). Imol %3D 9reaction=n AH %3D %3D (ans: -5.64 x 10 kJ/mol) Calcium chloride and sodium hydroxide react according to the equation: CaCl(aq) + 2 NaOH(aq) → Ca(OH),(s) +2 NaCl(aq) What volume of 2.00 M NaOH would be needed to react with 50.0 mL of 1.50 M CaCl,? a, 017 (ans: 75.0 mL) b) When 50.0 mL of 1.50 M CaCl, solution and 80.0 mL of 2.00 M NAOH solution (i.e., 5.0 mL more than calculated in part (a)) both initially at 22.3°C are mixed in a coffee-cup calorimeter with a calorimeter constant of 12.8 J/ C, the temperature fell to 20.0°C. If the density of the reaction mixture is 1.03 g/mL and its specific heat 3.94 J/g°C, calculate the heat absorbed hv the reaction. (ans: 1.24 x 103) c) Calculate AH (in kJ per mole of Ca(OH)2 formed) for the reaction. (ans: 1.6 x 10' kJ/mol)arrow_forward5) While making an inventory of the Chemistry Laboratory, Din Djarin found a dusty amber bottle hidden in one of the cabinets and the bottle has the following label: el "Compound R with empirical formula C3H302. (empirical Mw = 71 g/mol), in 50.0 g of ethanol" A preliminary test concluded that this solution boils at 79.03°C and after evaporating the ethanol it is found that there are 5.5 g of compound R. a) Calculate the molality of the solution. (Tb of alcohol = 78.4 °C, Kb = 1.22 °C/m). mol m= kg 01402 Mol b) Calculate the molar mass of the solute. c) What is its molecular formula?. b) qu e/ he el e larrow_forwardChlorine gas was first prepared in 1774 by the oxidation of NaCl with MnO₂: 2NaCl(s) + 2H2SO4 (1) + MnO2 (s)→→ Na2SO4(s) + MnSO4(s) + 2H₂O(g) + Cl₂(g) Assume that the gas produced is saturated with water vapor at a partial pressure of 28.7 mm Hg and that it has a volume of 0.597 L at 27 °C and 755 mm Hg pressure.arrow_forward
- A water contains 50.40 mg · L−1 as CaCO3 of carbon dioxide, 190.00 mg · L−1 as CaCO3 of Ca2+ and 55.00 mg · L−1 as CaCO3 of Mg2+. All of the hardness is carbonate hardness. Using the stoichiometry of the lime soda ash softening equations, what is the daily sludge production (in dry weight, kg · day−1) if the plant treats water at a rate of 2.935 m3 · s−1? Assume that the effluent water contains no carbon dioxide, 30.0 mg · L−1 as CaCO3 of Ca2+ and 10.0 mg · L−1 as CaCO3 of Mg2+. Be sure to calculate the mass of CaCO3 and Mg(OH)2 sludge produced each day.arrow_forwardThe binary compound M2X has a solubility of 9.4 × 10−7 mol/L and a molar mass, ℳ = 126.04 g/mol. Calculate the Ksp for this compound. Report your answer to TWO significant figures. Enter your answer in scientific notation using the appropriate boxes. Remember, a number like 1.6, in scientific notation is 1.6 × 100. Note: Your answer is assumed to be reduced to the highest power possible.arrow_forward✓- a Gasoline vaporizes in the carburetor of an automobile engine 2Hg (1) + O₂ (g) --> - 2HgO (s) seperating solute particles throughout a solvent SO₂(g) + CaO(s) -----> CaSO3(s) freezing water to form ice BaF2 (s)--> Ba2+ (aq) + 2F (aq) PCL3(g) + Cl₂(g) ----> PCL5 (g) melting ice to form water Raindrops form in a cloud | C₂H4(g) + H₂(g) ---> - C₂H6 (g) Ni(s) + 2 HCl(aq) -----> H₂(g) + NiCl₂(aq) Sugar dissolves in coffee CO₂(g) + H₂O(1) ----> H₂CO3(aq) 2NO₂ (g) - dissolution of solid KCl in water mixing of two gases into one container boiling water to form steam Frost forms on the windshield of your car --> N₂O4 (g) CaO(s) + 2 HCl(g) ---> CaCl₂(s) + H₂O(l) Moisture condenses on the outside of a cold glass ir is pumped into a tire Sub question a. As0arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning



