
Derive Equation 6.5−4 for the boiling-point elevation of a dilute solution of a nonvolatile solute with mole fraction x in a solvent that has a pure-component vapor pressure
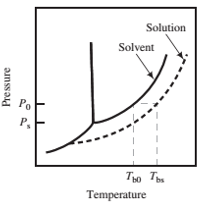
The procedure is as follows.
(a) Write the Clausius−Clapeyron equation (Equation 6.1-3) for P (the effective solvent vapor pressure at Tb0) and then for P0(the effective solvent vapor pressure at Tbs), assuming that at the low solute concentrations in question the heat of vaporization is the same at both temperatures.
Subtract the two equations. Simplify the equation algebraically, assuming that Tb0and Tbsare close enough together to say that
(b) Substitute the Raoult’s law expression (Equation 6.5−2) for
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Fundamentals of Applied Electromagnetics (7th Edition)
Artificial Intelligence: A Modern Approach
Introduction to Programming Using Visual Basic (10th Edition)
Modern Database Management
- Some nonelectrolyte solute (molar mass = 142 g/mol) was dissolved in 150. mL of a solvent (density = 0.879 g/cm3). The elevated boiling point of the solution was 355.4 K. What mass of solute was dissolved in the solvent? For the solvent, the enthalpy of vaporization is 33.90 kJ/mol, the entropy of vaporization is 95.95 J/K mol, and the boiling-point elevation constant is 2.5 K kg/mol.arrow_forwardA polymer of large molar mass is dissolved in the organic solvent m-cresol (C7H8O) at 25 °C, and the resulting solution rises to a final height of 13.4 cm above the level of the pure solvent, as solvent molecules pass through a semipermeable membrane into the solution. If the solution contains 5.24 g polymer per liter, calculate the molar mass of the polymer. Take the density of the solution to be 1.03 g cm-3.arrow_forwardThe Henry's law constant for CO2(g)CO2(g) in water at 25 ∘C25 ∘C is 29.4 bar·M−129.4 bar·M−1. Estimate the concentration of dissolved CO2(g)CO2(g) in a carbonated soft drink pressurized with 2.002.00 bar of CO2(g)CO2(g).arrow_forward
- When ethanol is added to water at 25 °C and 1 bar, the total volume of the solution changes according to the empirical function cm x - 0.36394 cm (mol EtOH) = ** V = 1002.93 cm + 56.6664 mol EtOH cm (mol EtOH) + 0.028256- where x is the number of moles of ethanol in the solution. As ethanol is added to the solution, the partial molar volume of ethanol decreases, reaches a minimum, and then increases. This behavior is shown in the figure below. Find the function for (ma) find the value x = ng 56 Ong TP How many moles of ethanol are present when the partial molar volume of ethanol in the solution is minimum? How many moles of water are present if the mole fraction of ethanol with this amount of ethanol present is 0.0952? 53 5. *- nmol 10 Is the partial molar volume of water increasing, decreasing with increasing x, at a maximum or at a minimum? Explain based on an equation based on an equation from the lecture slides. Partial melar volume, VAcm motarrow_forwardOsmotic pressures are often reported in units of atmospheres or mm Hg. The latter impliesthat the height of a column of liquid can be used as a measure of pressure. This is, in fact, thebasis of the torricellian barometer from which the units of “torr” come. The pressure, P, isrelated to the height of the column by:P = ρghwhere ρ is the density of the liquid, g is the acceleration due to gravity and h is the height. Whatwill be the height (in mm) of a column of aqueous solution with a density of 0.9987 g/mL, if theosmotic pressure of the solution at 25 °C is 1.4 mm Hg? The density of mercury at thistemperature is 13.8 g/mLarrow_forwardb) Suppose you had a solution of 6.84 g of sugar (sucrose, C12HO1) in 90.0 g of water. (i) Calculate the number of moles of sugar and water present. (H=1; C=12; 0=16) (ii) Calculate the mole fraction of the water in the solution. (iii) At 100°C, the saturated vapour pressure of water at its boiling point is 101325 Pa. Calculate the vapour pressure of the solution at that temperature. (Remember to give your answer to a suitable number of significant figures.) (iv) Would the solution of sugar in water boil at this temperature, assuming the external pressure remains at 101325 Pa? Explain your answer.arrow_forward
- Aniline, C6H5NH2, and hexane, C6H14 , form partially miscible liquid - liquid mixtures at temperatures below 69. 1 °C. When 42.8 g of aniline and 75 .2 g of hexane are mixed together at a temperature of 67.5 °C, two separate liquid phases are formed, with mole fractions of aniline of 0.308 and 0.618.(a) Determine the overall mole fraction of aniline in the mixture. (b) Use the lever rule to determine the relative amounts of the two phases.arrow_forwardAt 39.9 ◦C, a mixture of ethanol (x1 = 0.9060, p1 = 130.4 Torr) and isooctane ( p2 = 43.9 Torr) forms a vapor phase with molar fraction y1 = 0.6667 at a total pressure of 185.9 Torr. (a) Calculate the activity and activity coefficient of each component. (b) Calculate the total vapor pressure that the mixture would have if it were ideal.arrow_forwardA water carbonating plant is available for use in the home and operates by providing carbon dioxide at 5.0 atm. Estimate the molal concentration, m (in mol/kg), of the soda water it produces. The Henry's law constant for CO2 in water at 25 °C is 1650 atm. Use the following representation of Henry' law: PB = xBKB where PB is the vapor pressure of the solute; xB is the mole fraction of the solute (mole per mole); and KB is the proportionality constant.arrow_forward
- The following relation describes the volume change in a water (solvent – species 1) and salt (solute – species 2) solution: V=1.001+16.625n2 +56.092 n23/2 +119.4n22 where V is expressed in units of liters, and n2 in kmole. (1) Obtain an expression for partial V2 in terms of n2, and determine partial V2 when x2 is approching 0 in liter per kmole. (2) Obtain an expression for partial V1 one in terms of n2, and one in terms of x1.arrow_forwardPhenol and water form non-ideal liquid mixtures. When 7.32 g of phenol and 7.95 g of water are mixed together at 60 °C they form two immiscible liquid phases with mole fractions of phenol of 0.042 and 0.161. (i) Calculate the overall mole fraction of phenol in the mixture. (ii) Use the lever rule to determine the relative amounts of the two phases.arrow_forwardIn the determination of molar mass of an unknown substance by ebullioscopic constant, 30 ml of acetone (C3H6O) was placed in a test tube with thermometer and glass tubing and subjected to water bath. Upon boiling, the temperature reads 56 deg C. For the boiling point of unknown-acetone solution, you prepared the solution by mixing 1.60 g of unknown solute in the 30 mL acetone and subjected it again to water bath. The boiling temperature of the solution is 56.68 deg C. The density of acetone = 0.9849 g/mL and the Kb of acetone = 1.67 degC/kg 1. What is the value for delta Tb or the change in boiling temperature (in degrees Celsius)? 2. How many moles of solute is present in the solution given in the situation?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning



