
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
4th Edition
ISBN: 9781119192107
Author: BULLARD
Publisher: JOHN WILEY+SONS,INC.-CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 6, Problem 6.96P
Interpretation Introduction
Interpretation:
To determine amount of xylene transfers to the sulfolane phase.
Concept introduction:
Extraction is a process of separating different components of a mixture based on their solubility. When the vapor pressure of different components is of the same range, they cannot be separated by the distillation column. Extraction is used for the similar boiling point range compounds.
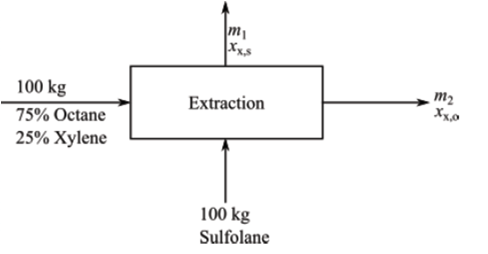
Flowchart for the process is as follows:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The separation of aromatic compounds from paraffins is essential in producing many polyesters that are used in a variety of products. When aromatics and paraffins have the same number of carbon atoms, they often have similar vapor pressures, which makes them difficult to separate by distillation. Extraction is a viable alternative, as illustrated by the following simple system. Sulfolane (an industrial solvent) and octane may be considered completely immiscible. At 25°C, the ratio of the mass fraction of xylene in the octane-rich phase to the mass fraction of xylene in the sulfolanerich phase is 0.25. One hundred kg of pure sulfolane are added to 100 kg of a mixture containing 75 wt% octane and 25% xylene, andthe resulting systemis allowedto equilibrate. How much xylenetransferstothe sulfolane phase?
33. 1.18 g aniline liquid is mixed with 1.00 g benzene liquid at 25 oC. Calculate the mole fraction of aniline in the vapor phase. At 25 oC, the vapor pressure of pure benzene is 30.5 kPa and the vapor pressure of pure aniline is 28.0 kPa.
molar masses: benzene = 78.00 g/mole and aniline = 93.12 g/mole
a.0.821b.0.233c.0.522d.0.476
In an organic chemistry lab, some camphor crystals were synthesized but there were some traces
of isoborneol also present in the crystals which lowered the melting point of the camphor crystals
from 179 °C to 169 °C. Treating it as a solid-solid solution with isoborneol as the solute and
camphor as the solvent, find the mass percentages of camphor and isoborneol in the solution.
The freezing point depression constant for camphor is 40 °C kg mol1. The molecular weights of
camphor and isoborneol are 152.2 g/mol and 154.3 g/mol respectively.
O A. 96.14 % and 3.86 % respectively
B. 3.80 % and 96.20 % respectively
O C. 3.67 % and 96.33 % respectively
O D. 96.29 % and 3.71 % respectively
O E. 99.75 % and 0.25 % respectively
Chapter 6 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Ch. 6 - Ten mL of pure liquid water in a cylinder with a...Ch. 6 - A quantity of methyl acetate is placed in an open,...Ch. 6 - Ethyl alcohol has a vapor pressure of 20.0 mm Hg...Ch. 6 - Prob. 6.4PCh. 6 - Prob. 6.5PCh. 6 - Prob. 6.6PCh. 6 - Prob. 6.7PCh. 6 - Prob. 6.8PCh. 6 - Prob. 6.9PCh. 6 - Prob. 6.10P
Ch. 6 - Prob. 6.11PCh. 6 - Prob. 6.12PCh. 6 - Prob. 6.13PCh. 6 - Air at 50% relative humidity is cooled...Ch. 6 - Prob. 6.15PCh. 6 - Prob. 6.16PCh. 6 - Air at 90°C and 1.00 atm (absolute) contains 10.0...Ch. 6 - When fermentation units are operated with high...Ch. 6 - When you step out of a shower, the temperature in...Ch. 6 - A fuel cell is an electrochemical device in which...Ch. 6 - Prob. 6.21PCh. 6 - Prob. 6.22PCh. 6 - Prob. 6.23PCh. 6 - Prob. 6.24PCh. 6 - Prob. 6.25PCh. 6 - Prob. 6.26PCh. 6 - Prob. 6.27PCh. 6 - Prob. 6.28PCh. 6 - An air conditioner is designed to bring 10.000...Ch. 6 - Prob. 6.30PCh. 6 - Prob. 6.31PCh. 6 - Prob. 6.32PCh. 6 - A gas stream containing 40.0 mole% hydrogen, 35.0%...Ch. 6 - Prob. 6.34PCh. 6 - In the manufacture of an active pharmaceutical...Ch. 6 - Prob. 6.36PCh. 6 - In the ?nal stage of the manufacturing process...Ch. 6 - Prob. 6.38PCh. 6 - A fuel gas containing methane and ethane is burned...Ch. 6 - A mixture of propane and butane is burned with...Ch. 6 - An important parameter in the design of gas...Ch. 6 - A liquid stream consisting of 12.5 mole% n-butane...Ch. 6 - Nitric acid is a chemical intermediate primarily...Ch. 6 - Prob. 6.44PCh. 6 - Sulfur trioxide (SO3) dissolves in and reacts with...Ch. 6 - State whether you would use Raoult’s law or Henrys...Ch. 6 - A gas containing nitrogen, benzene, and toluene is...Ch. 6 - Prob. 6.48PCh. 6 - Prob. 6.49PCh. 6 - A conelation for methane solubility in...Ch. 6 - Prob. 6.51PCh. 6 - The constituent partial pressures of a gas in...Ch. 6 - Prob. 6.53PCh. 6 - Prob. 6.54PCh. 6 - Prob. 6.55PCh. 6 - Prob. 6.56PCh. 6 - Prob. 6.57PCh. 6 - Prob. 6.58PCh. 6 - Nitrogen is bubbled through a liquid mixture that...Ch. 6 - Prob. 6.60PCh. 6 - Prob. 6.61PCh. 6 - Prob. 6.62PCh. 6 - The feed to a distillation column (sketched below)...Ch. 6 - Prob. 6.64PCh. 6 - Prob. 6.65PCh. 6 - Prob. 6.66PCh. 6 - Prob. 6.67PCh. 6 - Prob. 6.68PCh. 6 - Prob. 6.69PCh. 6 - Prob. 6.70PCh. 6 - A methanol-water feed stream is introduced to a...Ch. 6 - Prob. 6.72PCh. 6 - In this problem you will use a spreadsheet to...Ch. 6 - Prob. 6.74PCh. 6 - Prob. 6.75PCh. 6 - Prob. 6.76PCh. 6 - Acetaldehyde is synthesized by the catalytic...Ch. 6 - Dehydration of natural gas is necessary to prevent...Ch. 6 - A two-unit process is used to separate H2S from a...Ch. 6 - Prob. 6.80PCh. 6 - Prob. 6.81PCh. 6 - Prob. 6.82PCh. 6 - Prob. 6.83PCh. 6 - A solution containing 100 lbm KNO3/100 Ibm H2O at...Ch. 6 - A 10.0 wt% aqueous solution of sodium chloride is...Ch. 6 - Potassium dichromate (K2Cr2O7) is to be recovered...Ch. 6 - Prob. 6.87PCh. 6 - Prob. 6.88PCh. 6 - Sodium bicarbonate is synthesized by reacting...Ch. 6 - An ore containing 90 wt% MgSO4(H2O and the balance...Ch. 6 - An aqueous waste stream leaving a process contains...Ch. 6 - A solution of diphenyl (MW = 154.2) in benzene is...Ch. 6 - An aqueous solution of urea (MW = 60.06) freezes...Ch. 6 - Prob. 6.94PCh. 6 - Derive Equation 6.54 for the boiling-point...Ch. 6 - Prob. 6.96PCh. 6 - A stream of 5.00 wt% oleic acid in cottonseed oil...Ch. 6 - Benzene and hexane are being considered as...Ch. 6 - Acetone is lo be extracted with n-hexane from a...Ch. 6 - Prob. 6.100PCh. 6 - Prob. 6.101PCh. 6 - Five kilograms of a 30 wt% acetone70% water...Ch. 6 - An aqueous acetone solution is fed at a rate of...Ch. 6 - Prob. 6.104PCh. 6 - Prob. 6.105PCh. 6 - Air at 25°C and 1 atm with a relative humidity of...Ch. 6 - Prob. 6.107PCh. 6 - Prob. 6.108PCh. 6 - Various amounts of activated carbon were added to...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The blue line on the diagram illustrates the effect of using fractional distillation to separate a mixture of hexane (C6H14) and heptane (C7H16). If one starts a fractional distillation with a 0.20 mole fraction of hexane, what is the approximate mole fraction of hexane in the vapor phase after two evaporation condensation cycles? Figure B A temperaturecomposition diagram for hexane (C6H14)-heptane (C7H16) mixtures.arrow_forwardCarbon tetrachloride (CCl4) and benzene (C6H6) form ideal solutions. Consider an equimolar solution of CCl4 and C6H6 at 25C. The vapor above the solution is collected and condensed. Using the following data, determine the composition in mole fraction of the condensed vapor. Substance Gfo C6H6(l) 124.50 kJ/mol C6H6(g) 129.66 kJ/mol CCI4(l) 65.21 kJ/mol CCI4,(g) 60.59 kJ/molarrow_forwardA solution is prepared by dissolving 40.00 g of MgCl2 (f.w. = 95.211 g mol–1), a non-volatile solute, in enough water (m.w. = 18.02 g mol–1) to result in exactly 2 L of solution at 25 °C. Assume the density of the solution is that of pure water (dsolution = 1.000 g mL–1). The ebullioscopic constant (Kb) for water is 0.513 °C m–1. The cryoscopic constant (Kf) for water is 1.86 °C m–1. The vapor pressure of pure water is 0.0313 atm. Determine the freezing point of the solution.arrow_forward
- A solution is prepared by dissolving 40.00 g of MgCl2 (f.w. = 95.211 g mol–1), a non-volatile solute, in enough water (m.w. = 18.02 g mol–1) to result in exactly 2 L of solution at 25 °C. Assume the density of the solution is that of pure water (dsolution = 1.000 g mL–1). The ebullioscopic constant (Kb) for water is 0.513 °C m–1. The cryoscopic constant (Kf) for water is 1.86 °C m–1. The vapor pressure of pure water is 0.0313 atm. Determine the osmotic pressure in atm.arrow_forwardYou have a solution that contains 4.0g of oxalic acid dissolved in 100ml of water. this solution is placed in a separatory funnel, and then 100ml of diethyl ether is added to the separatory funnel and an extraction is performed. (Diethyl ether and water are immiscible solvents.) Use the following constant to calculate how much oxalic acid is in each solvent after extraction. Solubility of Oxalic acid in water=9.5g/100ml Solubility of Oxalic acid in ether=16.9g/100mlarrow_forward33. 1.18 g aniline liquid is mixed with 1.00 g benzene liquid at 25 oC. Calculate the mole fraction of aniline in the vapor phase. At 25 oC, the vapor pressure of pure benzene is 30.5 kPa and the vapor pressure of pure aniline is 28.0 kPa. molar masses: benzene = 78.00 g/mole and aniline = 93.12 g/mole Group of answer choices 0.522 0.233 0.821 0.476arrow_forward
- synthetic ion exchange resins. These resins are organic polymeric networks that contain functional groups that are permanently attached to the resin. One such functional group is sulfonic acid, -SO3H. When placed in water, the resin (being a large organic molecule) will not dissolve. However, the ionic functional groups become hydrated and the resin will swell as it takes up waters of hydration. The resin may then be thought of as a solid solution mixture with the functional groups being "dissolved" but the polymer remaining as a solid. The hydronium ion (H3O+) associated with acidic functional groups like sulfonic acid will dissociate just as it would if the acid were free of the resin. If other cations such as Cu2+ or Na+ are added to the resin, an ion-exchange reaction can occur: . Note that one H+ is released for each positive charge bound to the resin. Since the resin exchanges a hydrogen ion for a sodium ion in this reaction, sulfonic acid resins are called cation-exchange…arrow_forwardWhen 1 mole of water is added to an infinitely large amount of an aqueous methanol solution having a mole fraction of methanol of .40, the volume of the solution increased by 17.35cm^3 and 25C. When 1 mole of methanol is added to such a solution, the volume increases by 39.01 cm^3. Calculate the volume of a solution containing .40 mole of methanol and .60 mole of water. What is the volume of .6 mole of water plus the volume of .4 mole of methanol before they are mixed? The densities of water and methanol at 25C are .998g/cm^3 and .791g/cm^3 respectivley.arrow_forwardSolute S has a distribution constant of 4.9 between water (phase 1) and hexane (phase 2). A 89.0 mL solution of S in water is extracted five times with 14.0 mL of hexane. Calculate the fraction of S remaining in the aqueous phase. fraction remaining:arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning