
An ore containing 90 wt% MgSO4(H2O and the balance insoluble minerals is fed to a dissolution tank at a rate of 60,000 lbm/h along with fresh water and a recycle stream. The tank contents are heated to 120°F, causing all of the magnesium sulfate monohydrate in the ore to dissolve, forming a solution 10°F above saturation. The resulting slurry of the insoluble minerals in MgSO4 solution is pumped to a heated ?lter, where a wet ?lter cake is separated from a solids-free ?ltrate. The ?lter cake retains 5 lbm of solution per 100 lbm of solids. The ?ltrate is sent to a crystallizer in which the temperature is reduced to 50°F, producing a slurry of MgSO4(7H2O crystals in a saturated solution that is sent to another ?lter. The product ?lter cake contains all of the crystals and entrained solution in a ratio of 5 lbm solution per 100 lbm crystals. The ?ltrate from this ?lter is returned to the dissolution tank as the recycle stream.
Solubility data: Saturated magnesium sulfate solutions at 110°F and 50°F contain 32 wt% MgSO4 and 23 wt% MgSO4, respectively.
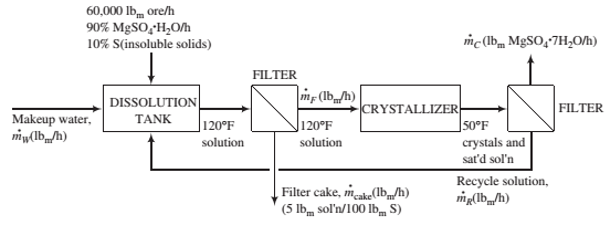
(a) Explain why the solution is ?rst heated (in the dissolution tank) and ?ltered and then cooled (in the crystallizer) and ?ltered.
(b) Calculate the production rate of crystals and the required feed rate of fresh water to the dissolution tank. (Note: Don‘t forget to include water of hydration when you write a mass balance on water.)
(c) Calculate the ratio lbm recycle/lbm makeup water.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Computer Science: An Overview (13th Edition) (What's New in Computer Science)
Prelude to Programming
Starting out with Visual C# (4th Edition)
Starting Out with C++ from Control Structures to Objects (9th Edition)
- Tungsten is extracted from the mineral scheelite (CaWO4) by roasting it with natrite (Na2CO3) at a temperature of about 700 °C. This converts the CaWO4 to water soluble sodium tungstate (NazWO4). The solid product from the roaster (called "calcine") is transferred to a leacher. In the leacher chilled water is added to dissolve the NazWO4. The chilled water dissolves only the NazWO4 in the solid calcine. The scheelite that enters the roast is mixed with gypsum (CaSO4-2H2O). This stream is 85.2%w CaWO4 and 14.8%w CaSO4-2H2O. (%w means “percent-by-weight.") Four reactions take place during this process. In the roaster: 1: CaWO4 + Na2CO3 - NazWO4 + CaO + CO2 2: CaSO4•2H2O - CaSO4 + 2H2O In the leacher: 3: СаО + H2О —- Са(ОН)2 4: CaSO4 + 2H2O - CaSO4 2H2O The first reaction does not go to completion, all the other reactions do go to completion. For the conditions shown in the process diagram on the next page calculate the conversion of the CaWO4 in the roaster.arrow_forwardA steady-state process to recover crystalline potassium chromate (K,CrOz) from an aqueous solution of this salt is required. Four thousand kilograms per hour of a solution that is one-third K,CrO, by mass is joined by a recycle stream containing 36.4% K2CrO7, and the combined stream is fed into an evaporator. The concentrated stream leaving the evaporator contains 49.4% K,CrO; this stream is fed into a crystallizer in which it is cooled (causing crystals of K,CrO, to come out of solution) and then filtered. The filter cake consists of K,CrO, crystals and a solution that contains 36.4% K,CrO, by mass; the crystals account for 95% of the total mass of the filter cake. The solution that passes through the filter, also 36.4% K,CrO,, is the recycle stream. 1- Draw the flowchart of the system and put all known information. 2- Calculate the rate of evaporation, the rate of production of crystalline K,CrO7, the feed rates that the evaporator and the crystallizer must be designed to handle, and…arrow_forwardHydrogen peroxide solutions can easily be decomposed by dust, trace amounts of metals or light. It istherefore necessary to periodically determine the purity of the peroxide solutions.This can be done via a redox titration as follows: The density of a particular surgical grade hydrogen peroxide solution is 1.11g.cm-3. Exactly 10 mL of this solution was pipetted and quantitatively transferred, along with a sufficientamount of a dilute sulphuric acid solution, into a 250.00 mL volumetric flask and made up to the mark. A 25.00 mL portion of this diluted solution is pipetted into a conical flask, and titrated with a0.372 M KMnO4 solution. The endpoint of the titration is determined when the colourless solution in the conical flask turns apersistent bright pink due to the presence of excess permanganate ions. The average titration value is 26.20 mL of the KMnO4 solution 1.Determine the percentage of the hydrogen peroxide in the solution sample.2. Naturally occurring manganese…arrow_forward
- We want to determine the purity of alum sample bought in a local pharmacy in Miagao. A Chem 28 student determined the purity gravimetrically of a sample of alum, K2SO4:Al2(SO4)3:24H20. A 1.5879-g sample was dissolved and the aluminum precipitated as Al(OH)3. The precipitate was collected by filtration, washed, and ignited to give a product Al203 weighing 0.1572 g. Show your calculation to determine the purity of the alum sample.arrow_forwardb) A plot of land was explored and found to contain 1,000,000 grams of gold. The average goldconcentration was found to be 0.8 grams per ton. A mine was set up on it and sluice boxes were used as the main gold recovery units and recovered200,000 g of gold from 80% of the ore. The concentration ratio of the sluice boxes was 10,000.The field was mined again. On this occasion bowl concentrators were used. The feed used wasestimated to be 800,000 tons and the concentration ratio of this unit was 20,000. 750,000 grams ofgold was recovered. Calculate the following: i) the Quantity and grade concentrate from the sluice boxes .................. ..................... ii) the quantity and grade of the concentrate from the bowl concentrator plant. .............. iii) the quantity and grade of the material remaining at this location...................................arrow_forwardA pipet is used to transfer 3.00 mL of a 2.00 M stock solution in flask “S” to a 25.00-mL volumetric flask “A,” which is then diluted with DI H2O to the calibration mark. The solution is thoroughly mixed. Next, 3.00 mL of the solution in volumetric flask “A” is transferred by pipet to a 50.00-mL volumetric flask “B” and then diluted with DI H2O to the calibration mark. Calculate the molarity of the solution in volumetric flask “B.”arrow_forward
- To each digestion flask 200 mL aliquots of 10 different drinking water samples were added. Silver was extracted by a co-precipitation procedure and collected by filtration. The precipitate was digested in 200 μl hot concentrated nitric acid and diluted to 5 mL in a dilute phosphate solution. Standards were prepared according to the following table. The samples were analyzed by graphite furnace AAS. Calculate the concentration of silver in each sample. Are there any problems? How will you address each of the identified problems?arrow_forward1)Please express the normal salt (NaCl) concentration in body fluid into molarity (mM).2) Commercial fuming Sulphuric acid (Oleum-H2S2O6) is 99.9%. solution. Please convert it into molarity.3) Find out the Volume (dm3) of product (gas) at RTP when 0.58 M, 150 mL NaOH (aq.) reacts with 350 mL, 0.25 NH4Cl.4) The above reaction has the product Ammonia, which when dissolved in 650 mL ethanol makes an alkaline ethanolic solution. Find its molarity (M) 5) Calculate the adult dose as per the BW of the baby. (Child dose-50 mg and the BW of the baby is 48 lb (British pound) (1lb=0.453 Kg)arrow_forwardTwo process streams are mixed to form a single stream. Only the flow in the mixed stream is known. A soluble salt is added to one of the original streams at a steady rate. Samples taken of this stream show it to be 4.76 % w salt. Samples from the combined stream show 0.62% w salt. What is the ratio of the flows in the two original streams?arrow_forward
- Hydroxyapatite, Ca,(PO4),(OH)₂, has a solubility constant of Ksp = 2.34 x 10-59, and dissociates according to Cao (PO)(OH)₂ (s)10 Ca2 (aq) + 6PO (aq) + 2OH (aq) Solid hydroxyapatite is dissolved in water to form a saturated solution. What is the concentration of Ca2+ in this solution if [OH] is fixed at 3.40 x 10-4 M? [Ca²+] = Marrow_forwardHydroxyapatite, Ca,(PO)(OH)₂, has a solubility constant of K-2.34 x 10-59, and dissociates according to Cao (PO)(OH)₂ (s) = 10 Ca² (aq) + 6PO (aq) + 2OH(aq) Solid hydroxyapatite is dissolved in water to form a saturated solution. What is the concentration of Ca²+ in this solution if JOH is fixed at 8.30 x 10M? |Ca²1 Marrow_forward4. (cont.) (e) CO2 (g) + H20 (1) → 5. Balance the following equations; write the ionic equations and the corresponding balanced net ionic equation with the proper phase symbols (if different from the ionic equation). H2SO4 (aq) + NaOH (ag) > Na2SO4 (aq) + H20 (1) (a) (b) Al203 (s) + HCl (aq) → AIC13 (aq) + H20 (1) (c) H2SO3 (aq) КОН (aq) > K2SO3 (aq) + H20 (1) (d) HC2H3O2 (aq) + Ca(OH)2 (aq) →. Ca(C2H3O2)2 (aq) +. H20 (1) 55arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
