
Acetaldehyde is synthesized by the catalytic dehydrogenation of ethanol:
Fresh feed (pure ethanol) is blended with a recycle stream (95 mole% ethanol and 5% acetaldehyde), and the combined stream is heated and vaporized, entering the reactor at 280°C. Gases leaving the reactor are cooled to -40°C to condense the acetaldehyde and unreacted ethanol. Off-gas from the condenser is sent to a scrubber, where the uncondensed organic compounds are removed and hydrogen is recovered as a by-product. The condensate from the condenser, which is 45 mole% ethanol, is sent to a distillation column that produces a distillate containing 99 mole% acetaldehyde and a bottoms product that constitutes the recycle blended with fresh feed to the process. The production rate of the distillate is 1000kg/h. The pressure throughout the process may be taken as 1 atm absolute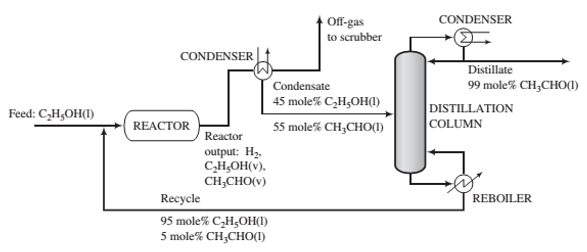
(a) Calculate the molar ?ow rates (kmol/h) of the fresh feed, the recycle stream, and the hydrogen in the off-gas. Also determine the volumetric ?ow rate (m3/h) of the feed to the reactor. (Suggestion: Use Raoult’s law in the analysis of the condenser.)
(b) Estimate (i) the overall and single—pass conversions of ethanol and (ii) the rates (kmol/h) at which ethanol and acetaldehyde are sent to the scrubber.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Introduction to Programming Using Visual Basic (10th Edition)
Materials for Civil and Construction Engineers (4th Edition)
Modern Database Management
Java: An Introduction to Problem Solving and Programming (8th Edition)
- The n-butane is converted into isobutane in an isomerization reactor, which operates isothermally at 149°C. Suppose that the reactor is fed with a mixture containing 93% mole of n-butane, 5% isobutane and 2% HCl at 149°C and that a 40% conversion of n-butane is achieved. How much heat in KJ/m3 (STP) of the feed, should be supplied or extracted from the reactor?arrow_forwardButane (C4H10)(C4H10) and dry air are fed to a combustion reactor at a steady rate of 45.9 mol/s45.9 mol/s butane and 2500.0 mol/s2500.0 mol/s dry air. Assume the butane reacts completely to produce CO2CO2 and H2O.H2O. The reactants enter the reactor at 25 ∘C25 ∘C and the products leave the reactor at 1110.4 ∘C.1110.4 ∘C. Calculate the percent excess air. Using the information below, find the heat interaction. Species ΔHf at 298 K(kJ/mol) CP (kJ/kmol·K) O2 0 3.5R N2 0 3.5R C4H10 -125.5 12R CO2 -393.5 3.5R H2O -241.8 3.5Rarrow_forwardethylene oxide is prepared by oxidation of ethylene. 100Kmol of ethylene and 100Kmol of O2 are charged to a reactor.The percent conversion of ethylene is 85 and percent yield of C2H4O is 94.12. Calculate the composition of product steam leaving the reactor .The reactions taking place are: C2H4+0.5O2=C2H4O C2H4+3O2= 2CO2+2H2Oarrow_forward
- Consider the following simultaneous reactions: N2 + 3H2 → 2NH3(a) H2 + CO2 → CO + H2O (b) 550 mols are fed into the reactor, initially containing 67.4% H2, 21.3% N2 and the remainder CO2. The mixture at the outlet has 16% NH3 and 5% H2O (molar percentages). a- Calculate the degrees of advancement of reactions (a) and (b).arrow_forwardfor the reactor below:N2(g)+ 3H2(g) -> 2Nh3(g)Kc= 9.60 at 300degrees celcius Calculate Kp for the reactor at the same temperaturearrow_forwardA vehicle airbag system works by using a series of chemical reactions. When sensors in the system detect a collision, a heat source instantly causes the decomposition of sodium azide at 300oC. This first reaction produces sodium metal and nitrogen gas. The formation of the nitrogen gas inflates the airbag, but unfortunately, the other product, sodium metal, is extremely dangerous and highly reactive. The sodium metal must undergo an immediate secondary reaction to transform into a safer substance. That’s where potassium nitrate gets involved. In the secondary reaction, sodium metal reacts with the potassium nitrate to form potassium oxide, sodium oxide, and additional nitrogen gas. This secondary generation of nitrogen gas also fills the airbag. Excess gas from the secondary reaction gets vented from e airbag. Finally, in the third and fourth reactions, the sodium oxide and potassium oxide react with silicon dioxide separately to product harmless and stable silicate glasses sodium…arrow_forward
- Titanium (Ti) is an important material in manufacturing airplane engines and frames. It is obtained from titanium tetrachloride (TiCl4), prepared from titanium dioxide, carbon, and chlorine through the following reaction: 3TiO2(s) + 4C(s) +6C2(g) -------à 3TiC4(g) 2CO2(g) + 2CO(g) a. If the reactor contains 11.34 g C, 13.56 g Cl2, and 8.3 g TiO2, identify the limiting reagent. b. Compute the theoretical yield of TiCl4. After the actual experiment, the reaction produces 13.48 g TiCl4. What is the percent yield of TiCl4?arrow_forward12:06 A * A * ll 68%i Zoom v Leave • REC Breakout Rooms GROUP WORK An iron ore was analyzed by dissolving a 1.1324 g sample in concentrated HCI. The resulting solution was diluted with water and the iron (III) was precipitated as the hydrous oxide Fe,O3.xH,O by the addition of NH3. After filtration and washing, the residue was ignited at a high temperature to give 0.5394 g of pure Fe,03 (159.69g/mol). Calculate (a) % Fe (55.847g/mol) and (b) % Fe,04(231.54g/mol) in the sample. •. 28 Unmute Start Video Share Participants More IIarrow_forwardA 51.0-LL reactor at 1600 KK is charged with 50.0 gg of NiO(s)NiO(s) and 1.40 atmatm of CO(g)CO(g). After equilibrium is reached, what is the partial pressure of CO2(g)CO2(g) in the reactor?arrow_forward
- The oxidation of ethylene to produce ethylene oxide proceeds according to the equation2C 2H 4 +O 2 ! 2C 2H 4OThe feed to a reactor contains 100 kmol C 2H 4 and 100 kmol O 2. Which reactant is limiting?arrow_forwardAktiv Chemistry → C < + app.101edu.co b Answered: Complete the balanX M Your Chemistry answer is ready X 1 Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs, write NR after the reaction arrow. Be sure to include the proper phases for all species within the reaction. ₁ + ■ Reset + Cu U 2 3 Cu₂SO (aq) + Na PO₂(aq) - 2 □2 ☐ 3 OC ) 4 NR Time's Up! P a Amazon.com: LINSY HOME Cor X a Amazon.com: bulletin board fo X LO 5 05 + 0²+ 07 6 7 8 9 0 = (s) O 3+ Na 4+ • x H₂O ☐9 口。 (1) (g) (aq) S Delete Update: Submitarrow_forwardYour careless, clumsy lab mate has accidentally allowed carbon monoxide to leak out of a reactor, causing your lab’s CO monitor alarm to sound. After you quickly put on the appropriate Personal Protective Equipment, you find the leak and seal it. Then, you close your lab door, shut the windows, and call OSHA to report the hazard.When OSHA technicians arrive, they begin ventilating the 3000-ft3laboratory by pumping 25 ft3/min of fresh air into the room and removing the contaminated air at the same rate. They put a shop fan in the laboratory, such that the air is well-mixed. They also install a digital CO composition monitor, which currently reads 2 mol%. OSHA tells you that the laboratory is safe when there is less than 35 ppm of CO in the lab.How long must you wait to get back to work?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





