
A liquid-phase chemical reaction A -* B takes place in a well-stirred tank. The concentration of A in the feed is f-AO (mol/m3), and that in the tank and outlet stream is CA (mol/nr). Neither concentration varies with time. The volume of the tank contents is V(m3) and the volumetric flow rale of the inlet and outlet streams is V (m7s). The reaction rate (the rate at which A is consumed by reaction in the lank) is given by the expression
r(mol A consumed/s) = JtVCA
where k is a constant.
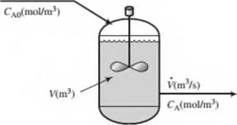
- Is this process continuous, batch, or semibatch? Is it transient or steady-state?
- What would you expect the reactant concentration CA to equal if k = 0 (no reaction)? What should it approach if k —» oo (inflnitely rapid reaction)?
- Write a differential balance on A, stating which terms in the general balance equation (accumulation = input + generation — output — consumption) you discarded and why you discarded them. Use the balance to derive the following relation between the inlet and outlet reactant concentrations:

Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Starting Out with Python (3rd Edition)
Electrical Engineering: Principles & Applications (7th Edition)
Concepts of Programming Languages (11th Edition)
Starting Out with Programming Logic and Design (5th Edition) (What's New in Computer Science)
- 2. a) Calculate the hard-sphere (HS) collision theory rate constant ks for the reaction NO(g) + C₁₂(g) Þ NOCl(g) + HS Cl(g) at 300 K in units of dm³ mols. The collision diameters of NO and Cl₂ are 370 and 540 pm, respectively. 16.0 g/mol, and mcı Use masses of m₁ = 14.0 g/mol, mo = 35.0 g/mol. N = b) Now include the line-of-centers (LOC) energy criterion to find koc in units of dm³ mol¹ s¹ using E₁ = 84.9 kJ mol LOC LOC c) Calculate the ratio of kчs and kLoc to the experimental rate constant at 300 K given A = 3.981 10° dm³ mol¹ s and E₁ = 84.9 kJ mol¹. a -1arrow_forward1 Ten (10) ml aqueous solutions of drug A (10% w/v) and drug B (25% w/v) are stored in two identical test tubes under identical storage conditions at 37°C for 3 months. If both drugs degrade by first-order, which drug will retain the highest percentage of initial concentration? (a) Drug A (b) Drug B (c) They will be the same.arrow_forward72.- The elementary reaction A + B C + D is carried out in a CSTR in which it is suspected there is bypass and dead volume. The table attached shows the step tracer concentrations at the output of the system. If the theoretical volume of the reactor is 1 m', the feeding volumetric flow rate is 0.1 m'/min, the rate constant is 0.28 m'/(kmol-min) and the feeding consists of an equimolar solution of A and B, with C = 2 kmol/m', calculate the conversion that can be expected in the system. %3D F=C t (min) 4 8. 10 14 16 18 00 C (mg/L) 1000 1333 1500 1666 1750 1800 2000 my Activate Windows Gto Settings to activalarrow_forward
- 5.10. A reaction 3A → B is carried out in a batch reactor. Molar con- centrations of A and B are monitored with time. Determine the appropriate reaction model based on the following data: 1. d 0.0 0.5 1.0 1.5 2.0 2.5 3.0 3.5 4.0 4.5 5.0 [A]. mol/m² 15.0 13.2 10.8 8.2 5.6 3.6 2.2 1.3 0.7 0.4 0.2 (B). mol/m 1.0 1.6 2.4 3.3 4.1 4.8 5.3 5.6 5.8 5.9 5.9arrow_forwardA river water sample of 25°C is contaminated with diazepam with initial concentration of 5000 ppb (or ng/L) when it secondary biodegradation rate in water, Kd = 10^1.3 (cu cm/mol-sec) at 25°C and molar mass of diazepam is 284.7g/mol. What is the half life of the diazepam in hours? Note: I need right answer with clear calculations.arrow_forward(ii) At elevated temperatures, nitrogen dioxide decomposes to nitric oxide and molecular oxygen: 2NO2(g) – → 2NO(g) + O2(g) The change in concentration of NO2 with time at 300 °C is given below: t (s) 100 200 300 400 500 [NO2]/103 (mol dm-3) 8.0 5.6 4.3 3.5 2.9 2.5 By appropriate plotting of the data and use of this graph: (a) verify that this reaction is second order with respect to NO2; (b) determine the rate constant for this reaction.arrow_forward
- Consider a gaseous mixture of A diluted in B that is required for a chemical reaction to take place within a cylindrical porous solid catalyst with radius R = 1 cm and length L = 10 cm. Once gas A reaches the catalyst surface, it enters the solid and the A->B reaction takes place, at a rate given by (-RA)= k. At the catalyst surface, that is, at r= R, the concentration of A is CAS = 1x10^-2 mol/cm. The diffusion coefficient from A to B is 0.7 cm^2/s. Determine: a) The concentration profile when k= 0, 0.001, 0.01 and 0.1 mol s cm b) The molar flux of species A when k=0, 0.001, 0.01 and 0.1 mol/s cm c) The molar flux of species A for 1 m? d) The average concentration of the system e) Establish which stage of the process is the limiting f) Graph how the concentration profile and the flux profile would be, analyze and discuss your results.arrow_forwardThe gas-phase decomposition of di-tert-butyl peroxide. (CH₂) COOC(CH3)3, is first order in the temperature range 110°C to 280°C with a first- order rate coefficient k=3.2 x 10¹ exp[-(164 kJ mol/RT³ Part A What is the value of AH for this reaction in the temperature range between 110 Cand 280 C Express your answer with the appropriate units. ΔΗ" - 164 НА kJ mol Submit Previous Answers Request Answer Xncorrect; Try Again; 4 attempts remaining ?arrow_forwardAn ocean outfall diffuser that discharges treated wastewater into the Pacific ocean is 5,000 m from a public beach. The wastewater contains 105 coliform bacteria per milliliter (coliform /mL). The wastewater discharge flow rate is 0.3 m3/s. The coliform first-order death rate in seawater is approximately 0.3 h-1. The current carries the wastewater plume toward the beach at a rate of 0.5 m/s. The ocean current may be approximated as a pipe carrying 600 m3/s of seawater. Determine the coliform concentration at the beach. Assume that the current behaves as a plug-flow reactor and that the wastewater is completely mixed in the current at the discharge point.arrow_forward
- (267) (268) 57 5 La Ce 138.905 140.116 89 90 Ac Th (227) 232.038 Increases 2. According to the collision theory, what 3 circumstances are needed for CH₁₂O, & O₂ to react? Beacting Substances must Collide, must Collide with enough energy. and must collide with the comet orientation 3. What is the activation energy for a chemical reaction? The Limont of free enering that must be added to go from the mangy fel of the Acts 1. For the Table below, use the collision theory to explain. The following equation applies to of the transition State. statements with an asterisk. *C.H12O6 (1 +6026H0 m+ 6CO2 Change in condition Increasing the temperature Increasing the concentration of CH12O6 Decreasing the concentration of 0₂ Increase the surface area by chewing up food in your mouth Decreasing the temperature Increasing the pressure in the container Increasing the volume of the container the reaction occurs in Using a catalyst (like salivary amylase) Does this increase or decrease the rate of…arrow_forwardA large, aerated lagoon is to be used to treat wastewater from a small community. The wastewater flow is 0.05 m³/s. The influent (input) wastewater contaminant concentration is 250 mg/L. The required effluent (output) concentration is 30 mg/L. It was found that the waste degrades according to first order kinetics. The degradation coefficient (k) for the reaction is 0.52 d'. You may assume that the lagoon is completely mixed and that conditions in the system are at steady state. Determine: a) The required volume of the lagoon b) The hydraulic detention time of the lagoonarrow_forwardReferring to a continuous stirred tank reactor (CSTR), determine the reactor volume required to process 1,200 L/min of 6 M reactant A in a reaction of A → B given a conversion of 90%. The reaction rate constant, k = 2.0 min-1.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





