
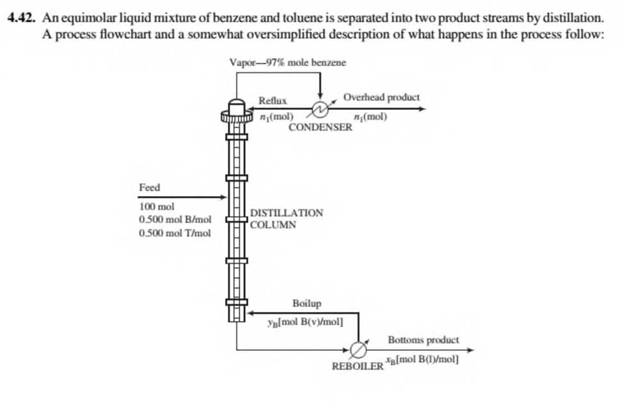
Inside the column a liquid stream flows downward and a vapor stream rises. At each point in the column some of the liquid vaporizes and some of the vapor condenses. The vapor leaving the top of the column, w hich contains 97 mole% benzene, is completely condensed and split into tw o equal fractions: one is taken off as the overhead product stream, and the other (the reflux) is recycled to the top of the column. The overhead product stream contains 89.2% of the benzene fed to the column. The liquid leaving the bottom of the column is fed to a partial reboiler in which 45% of it is vaporized. The vapor generated in the reboiler (the boilup) is recycled to become the rising vapor stream in the column, and the residual reboiler liquid is taken off as the bottom product stream. The compositions of the streams leaving the reboiler are governed by the relation
where Vb and .rB are the mole fractions of benzene in the vapor and liquid streams, respectively.
- Take a basis of 1IX) mol fed to the column. Draw and completely label a flowchart, and for each of four systems (overall process, column, condenser, and reboiler), do the degree-of-freedom analysis and identify a system with which the process analysis might appropriately begin (one with zero degrees of freedom).
- Write in order the equations you would solve to determine all unknown variables on the flowchart, circling the variable for which you would solve in each equation. Do not do the calculations in this part.
- Calculate the molar amounts of the overhead and bottoms products, the mole fraction of benzene in the bottoms product, and the percentage recover)' of toluene in the bottoms product (100 x moles toluene in bottoms/mole toluene in feed).
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Process Dynamics and Control, 4e
Engineering Mechanics: Statics & Dynamics (14th Edition)
Electric Circuits (10th Edition)
Fundamentals of Heat and Mass Transfer
Artificial Intelligence: A Modern Approach
- To prepare a delicious banana milk on an industrial scale, there is an experimental process that consists of seven unit operations. A stream of banana (whose components are fruit, peel, and moisture) is fed to a peeler, where 99.9% of the peel is removed. This stream should become pulp, so it is mixed with an additive Q to avoid pulp oxidation. This mixture is then sent to an extruder press wherein a stream of a 50:50 mixture of additives Q and R is added. The resulting flow of 30 kg/h has a composition of 2% additive R and 5% additive Q. This flow is then fed to an agitated mixer. To the same mixer is fed a dairy stream of 250 kg/h that contains 70% milk (the rest is water) and a sweet aqueous stream that contains 30% sugar. For each 25 kg/h of dairy flow, 2 kg/h of the sweet flow is fed. The stream that leaves the agitated mixer has 5% fruit and 0.005% peel and enters a series of two thermal processes in which each operation allows 5% of the water and 2% of the milk to evaporate. The…arrow_forward0 A too kmal/h stream that is 97 msle % Carbon tetrachlorice Ccc4) and 3 % Carbon disullide ccs) is to be recovered Prom the bottom of a distillation column. The feed to the column is 16 mole % CS, and 84 % Cc4, and 2 % of the cc4 enterirg the column is contained in the overhead stream leaving H top of 'the column- O a flow charf of the pocess and do the degree-of- Drae and label freedom analysis- Clculate the mass and mle fracklons of Ccl4 in the overhend stranm, and determine He malar Piow rates of cc4 & CS, in the overhend and feel streamsarrow_forwardA hydrocarbon stream composed of 40 mol% n-butane and 60 mol% n-octane is fed at 100 kmol/h to a flash vessel operating at 5 bar and 145°C. The liquid outlet stream of the flash vessel is then sent to a distillation column with a partial condenser to further separate the components. 90% of the butane in the feed to the column is recovered in the overhead vapor product (distillate), along with small amounts of octane. The bottoms product contains octane which accounts for 75% of the octane in the fresh hydrocarbon feed (to the flash vessel). The condenser and reboiler operating temperatures are 136.7°C and 165.7°C, respectively. Determine the compositions of the liquid outlet stream of the condenser and the vapor outlet stream of the reboiler.arrow_forward
- Q3/ A 100 mole feed containing equimolar amounts of methanol and water is mixe with 10 moles of a 40 mole% aqueous methanol stream. The mixture enters a distillation column that creates two streams. A top stream exits that contains 70 mole% methanol and balance with water. The bottom stream, which is 70 moles, enters a second distillation column. A top stream exits the second column as a 50% methanol and 50% water. The two top streams exiting the distillation column have the same flow rate. Calculate all unknown streams variables.arrow_forwardA mixture containing 42 wt% benzene (B) and 58 wt% toluene (T) is fed to a distillation column at a flow rate of 100 kg per minute. The product steam leaving the top of the column (the overhead product) contains 90 wt% benzene and 85 wt% of the total benzene fed to the column exits in this overhead product stream. Calculate the mass flow rate and mass composition of the product stream leaving the bottom of the column. Calculate the volumetric flow rate of the overhead product, assuming that it exits the distillation column as a vapour stream at 82C and 1 atm (abs). Physical Property Data (S.G.= specific gravity) from table B-1: Benzene S.G. = 0.879 MW = 78.11 g/mol Toluene S.G. = 0.866 MW = 92.13 g/mol Water Density = 1.00 kg/L MW = 18.02 g/mol R = 0.08206 L atm/mol Karrow_forwardA gaseous fuel containing methane and ethane is burned with excess air. The fuel enters the furnace at 25°C and 1 atm, and the air enters at 200°C and 1 atm. The stack gas leaves the furnace at 800°C and 1 atm and contains 5.32 mole% CO2, 1.60% CO, 7.32% O₂, 12.24% H₂O, and the balance ₂. 1. Calculate the molar percentages of methane and ethane in the fuel gas and the percentage excess air fed to the reactor. 2. Calculate the heat (kJ) transferred from the reactor per cubic meter of fuel gas fed. 3. A proposal has been made to lower the feed rate of air to the furnace. State advantages and a drawback of doing so.arrow_forward
- 6. Assume you have 1kg of an alloy consisting of 85 wt% Sn and 15 wt% Pb at a temperature of 184 °C. How much Sn (in kg) needs to be added to solidify the entire mixture without lowering the temperature? Composition (at% Sn) 20 40 60 80 100 327°C Temperature (°C) 300 200 100 (Pb) a + L 18.3 20 183°C a + B Liquid 61.9 40 60 Composition (wt% Sn) B+L 80 232°C 97.8 B al 600 500 400 300 200 100 100 (Sn) Temperature (°F)arrow_forwardDetermine the volume (L) of hydrogen needed (based on room temperature 25 °C and l atm) to hydrotreat 1 liter crude oil with 1.8 wt % S to a sulfur content of 0.2 wt%. The oil has a specific gravity of 0.8 (compared to water). The stoichiometry follows the following reaction: Oil-S + 2H2¬→O¡I-H2 + H2Sarrow_forwardIn a process producing KNO3 salt, 1000 kg/h of a feed solution containing 10 wt% KNO3 is fed to an evaporator, which evaporates some water at 422 K to produce a 50 wt% KNO3 solution. This is then fed to a crystallizer at 311 K, where crystals containing 98 wt% KNO3 are removed. The saturated solution containing 37.5 wt% KNO3 is recycled to the evaporator. Calculate the amount of the recycle stream R in kg/h and the product stream of crystals P in kg/h.arrow_forward
- In a chemical production plant, cyclohexane is made by the reaction of benzene and hydrogen. Reaction is as follows: CHs + H2 → CoH12 The complete process of producing cyclohexane uses a reactor and a separator. Using the process shown below and assuming 20 % excess hydrogen in the fresh feed, find the ratio of the recycle stream to the fresh feed stream, the composition of the final product stream, and the composition of the stream leaving the reactor. The overall conversion of benzene is 92% and the single-pass conversion is 21.5 %. The recycle stream is composed of 23.9 % benzene and the remaining hydrogen. Fresh Feed REACTOR SEPARATOR Product Recyclearrow_forwardQ4/in a two stage process, acetic acid (A)is extracted from water (W) into hexanol (H) in a liquid-liquid extraction vessel and the extract is subsequently separated by distillation. Assume that water is completely insoluble in hexanol. A mixture of 18wt% acetic acid and the balance water is feed to liquid-liquid extraction vessel. Pure hexanol is feed to the column to extracted the acetic acid. The water-rich stream leaving the vessel is 99.5wt% water and the balance acetic acid. The hexanol-rich extract from the vessel is feed to a distillation column. The composition of the distillate is 96wt% acetic acid and the balance hexanol. The bottom stream contains 97.2wt% hexanol and recovers 95% of the hexanol feed to the liquid-liquid extraction vessel. Calculate the percentage of acetic acid in the process feed that is recovered in the distillate stream. Distillation Liq-liq Column Extractionarrow_forwardAn evaporation-crystallization process is used to obtain solid potassium sulfate from an aqueous solution of this salt. The fresh feed to the process contains 19.6% wt K2SO4 The wet filter cake consists of solid K2SO4 crystals and 40 % wt K2SO4 solution, in a ratio 10 kg crystals/kg solution. The filte a 40% solution is recycled to join the fresh food. Of the water fed to the evaporator, 45% is evaporated The evaporator has a maximum capacity of 175 kg/hr water evaporales Calculate a) The maximam production rate of solid K2SO4 b) The rate at which fresh feed must be supplied to achieve this production rate c) The ratio kg recycle kg fresh feedarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





