
Inside a distillation column (see Problem 4.8), a downward-flowing liquid and an upward-flowing vapor maintain contact with each other. For reasons we will discuss in greater detail in Chapter 6. the vapor stream becomes increasingly rich in the more volatile components of the mixture as it moves up the column, and the liquid stream is enriched in the less volatile components as it moves down. The vapor leaving the lop of the column goes to a condenser. A portion of the condensate is taken off as a product (the overhead product), and the remainder (the reflux) is returned to the top of the column to begin its downward journey as the liquid stream. The condensation process can be represented as shown below:
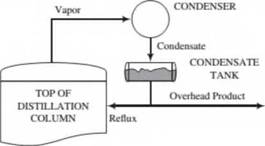
A distillation column is being used to separate a liquid mixture of ethanol (more volatile) and water (less volatile). A vapor mixture containing 89.0 moleSt ethanol and the balance water enters the overhead condenser at a rate of 100 lb-mole/h. The liquid condensate has a density of 49.0 lbnl/ft’. and
the reflux ratio is 3 lbm reflux/lbm overhead product. When the system is operating at steady state, the tank collecting the condensate is half full of liquid and the mean residence time in the tank (volume of liquid/volumetrie flow rate of liquid) is 10.0 minutes. Determine the overhead product volumetric flow rate (ft3/min) and the condenser tank volume (gal).
Trending nowThis is a popular solution!
Learn your wayIncludes step-by-step video

Chapter 4 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Introduction to Java Programming and Data Structures, Comprehensive Version (11th Edition)
Absolute Java (6th Edition)
Fundamentals of Heat and Mass Transfer
Starting Out with Java: From Control Structures through Objects (6th Edition)
- Write the balanced NET ionic equation for the reaction when aqueous SrBr₂ and aqueous K₂SO₄ are mixed in solution to form solid SrSO₄ and aqueous KBr. Be sure to include the proper phases for all species within the reaction.arrow_forwardA mixture combining 60 mL ethanol (C₂H6O) with 75 mL water (H₂O) is prepared at 280 K. The partial molar volumes of ethanol (E) and water (W) when mixed in these proportions (right column below) and in pure form (left column below) are ethanol water pure substance molar volume 58.2 mL/mol 18.00 mL/mol partial molar volume 55.3 mL/mol 17.7 mL/mol Determine the volume of the ethanol-water mixture. [Hint: You will need to determine the moles of each component.]arrow_forwardMethanol (CH3OH) is produced in the reaction of carbon dioxide and hydrogen via the reaction and process given below: CO2+3H2→CH3OH+H2O The fresh feed to the process contains hydrogen, carbon dioxide, and 0.400 mol% inerts (I). The reactor effluent passes through a condenser that removes essentially all of the methanol and water formed and none of the reactants or inerts. The unreacted reactants and inerts are recycled to the reactor. To avoid buildup of the inerts in the system, a purge stream is withdrawn from the recycle. 100 mol/h of feed to the reactor (not the fresh feed to the process) contains 29.5 mol% CO2, 68.0 mol% H2, and 2.50 mol% inerts. The single-pass conversion of hydrogen is 46.71%. What is the recycle-to-fresh feed ratio?arrow_forward
- Write the balanced NET ionic equation for the reaction when aqueous Cs3PO4 and aqueous AgNO3 are mixed in solution to form solid Ag3PO4 and aqueous CSNO3. Be sure to include the proper phases for all species within the reaction.arrow_forwardA geochemist in the field takes a small sample of the crystals of mineral compound X from a rock pool lined with more crystals of X. He notes the temperature of the pool, 24. C, and caps the sample carefully. Back in the lab, the geochemist dissolves the crystals in 3.00 L of distilled water. He then filters this solution and evaporates all the water under vacuum. Crystals of X are left behind. The researcher washes, dries and weighs the crystals. They weigh 45.0 g. Using only the information above, can you calculate yes the solubility of X in water at 24." C no If you said yes, calculate it. Be sure your answer has a unit symbol and 3 signiicant digits.arrow_forwardConsider the Solubility Curve for this Problem: Analysis for KClO3 & NH4Cl MW (KClO3) = 122.55 g/mol , MW (NH4Cl) = 53.49 g/mol i) What is the temperature at which KClO3 & NH4Cl both have the same solubility? Round to nearest degree. +/- 1° T = °C ii) What is the solubility (g per 100g H2O) at this temperature. Round to nearest gram. +/- 1 g s (KClO3) = g/100g H2O s (NH4Cl) = g/100g H2O iii) What is the molar solubility for KClO3 at this temperature? Assume the density of the solution is 1.21 g/cc. Round off to 2 sig. figs. |s| KClO3 = M iv) Calculate the Ksp for each of these chemicals at this temperature. Ksp = v) What is the unit of Ksp for KClO3? Unit for Ksp =arrow_forward
- 4.0g of potassium hydrogen tartrate was added to 300mL distilled water. The temperature of the solution is 23.1C. The liquid was filtered and 50mL of the filtered solution was transferred to 250mL beaker, two drops of phenolpthalein was added to the 250mL beaker. The concentration of NaOH is 1.0 M that is filled in the 2mL graduate pipette, single drops of NaOH was added to the 250mL beaker until the solution turns pink and the potassium hydrogen tartrate reach the endpoint. The datd of four trials was collected. Please answer the following questions 4) calculate ksp for potassium hydrogen tartrate for each trial and average ksp for thr experiment 5) calculate the percent error in you value of ksp using reference that is found as the theoretical value. 6) why was the temperature of the saturated solution recorded?arrow_forward4.0g of potassium hydrogen tartrate was added to 300mL distilled water. The temperature of the solution is 23.1C. The liquid was filtered and 50mL of the filtered solution was transferred to 250mL beaker, two drops of phenolpthalein was added to the 250mL beaker. The concentration of NaOH is 1.0 M that is filled in the 2mL graduate pipette, single drops of NaOH was added to the 250mL beaker until the solution turns pink and the potassium hydrogen tartrate reach the endpoint. The datd of four trials was collected. Please answer the following questions 1) calculate the total volume and moles of NaOH required to reach the endpoint for each trial. 2) calculate the molar solubility of potassium hydrogen tartate ( in mol/L) for each trial. 3) calculate the average molar solubility of potassium hydrogen tartate for the four trials. What is the average solubility of potassium hydrogen tartrate in g/L? 4) calculate ksp for potassium hydrogen tartrate for each trial and average ksp for thr…arrow_forwardProcedure part 1: Once the distillation is set up, 20.00 g of cyclohexanol is massed out and 3.00 ml of concentrated phosphoric acid is added as a catalyst to the boiling flask. Two boiling chips are added and the distillation can begin. The cooling water for the condenser is turned on and the flask is heated gently so that the temperature of the vapor does not exceed 135oC. The heating is continued until all but 1 -2 ml of residue remain in the boiling flask and it turns a dark yellow color. At this point the heat is turned off and the remaining liquid is placed into a waste container. In the receiving flask at the opposite end of the distillation apparatus, you will notice that the product appears in two layers. Question 1: Based on what we learned about the density of organic compounds, which layer is the organic layer (top or bottom)? Cyclohexanol + water Question 2: Looking at the reaction listed at the start of the lab, what two compounds are making the two…arrow_forward
- 50.0 mL of 0.400 M NaOH (ag) was added to 20.0 mL of 0.500 M H2SO4 (ag) in a calorimeter of heat capacity 39.0 J/K. The temperature of the resulting solution rose by 3.60 K. Find AH° in kJ/mol for the neutralization of H2SO4 (ag) with NaOH (ag). Specific heat of aqueous mixture = 4.184 J/g.K. Density of mixture = 1.030 g/mL Carrow_forwardA geochemist in the field takes a 9.0 mL sample of water from a rock pool lined with crystals of a certain mineral compound X. He notes the temperature of the pool, 15. C, and caps the sample carefully. Back in the lab, the geochemist first dilutes the sample with distilled water to 400. mL.. Then he filters and evaporates all the water under vacuum. Crystals of X are left behind. The researcher washes, dries and weighs the crystals. They weigh 0.045 g. Using only the information above, can you calculate the solubility of Xin water at 15.° C7 If you said yes, calculate it. Be sure your answer has a unit symbol and 2 significant digits. yes O no 0 0.9 H ロ・ロ X 0 Garrow_forwardSupposed you are in a chemistry laboratory and your CHEF102B teacher gave you a 500 ml Erlenmeyer flask containing 200 ml of heterogenous liquid mixture. This liquid mixture consists of sodium chloride, water, benzoic acid, is made by combining Mixture A and Mixture B. Mixture A (aqueous mixture): sodium chloride is dissolved in distilled water. Mixture B (organic mixture): benzoic acid, phenol and aniline is dissolved in petroleum ether (a nonpolar solvent). Your task is to separate the components of the heterogenous mixture and identify the components.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





