
Boilers are used in most chemical plants to generate steam for various purposes, such as to preheat process streams fed to reactors and separation units. In one such process, steam and a cold process fluid are fed to a heat exchanger where enough energy is transferred from the steam to cause a large fraction of it to condense. The uncondensed steam is vented to the atmosphere, and the liquid condensate is recycled to a deaerator into which another liquid stream (makeup water) is fed. The makeup water contains some dissolved impurities and other chemicals that help prevent deposition of solids on boiler walls and heating elements, which would lead to a reduction in operating efficiency and eventually to safety hazards, possibly including explosions. The liquid leaving the deaerator is the feed to the boiler. In the boiler, most of the water in the feed evaporates to form steam, and some of the impurities in the feedwater precipitate to form solid particles suspended in the liquid (kept in suspension by the chemical additives in the makeup w ater). The liquid and suspended solids are drawn oft' as boiler blowdown, either in manual bursts or w ith a continuous blowdown system.
A diagram of the system is shown below. The symbol I is used for combined impurities and chemical additives. The makeup water contains 1.0 kg 1/2.0 X 103 kg HjO, and the ratio in the blowdown is 1.0 kg 1/3.5 X 102 kg HiO. Of the steam fed to the heat exchanger, 76% is condensed.
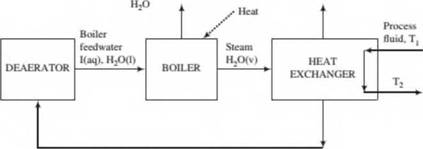
- In your own words, describe why makeup water, chemical additives to the makeup water, and blowdown are necessary in this process. Speculate on the probable disadvantage of making the 1/HsO ratio in the blowdown (I) too small, and (ii) too large.
- Assume a basis of calculation and draw and fully label a flow chart of the process. (When you draw the heat exchanger you can omit the process fluid, which plays no role in the problem.)
- Carry out a degree-of-freedom analysis and outline a solution procedure (which equations would you write in which order to calculate all of the unknow ns on the chart?).
(d, Calculate the ratio (mass of makeup water/lOOkg steam produced in boiler) and the percentage of the boiler feedwater taken off as blowdown.
(c) A proposal has been made to use highly purified water as makeup. List the benefits that would result from doing so and the most likely reason for not doing it.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Starting Out with C++: Early Objects (9th Edition)
Introduction To Finite Element Analysis And Design
Management Information Systems: Managing the Digital Firm (15th Edition)
Objects First with Java: A Practical Introduction Using BlueJ (6th Edition)
- A chemical manufacturer produces ethylene oxide by burning ethylene gas with air in the presence of catalyst. If the conditions are carefully controlled, a substantial fraction of ethylene remains unconverted and some is completely oxidized to form carbon dioxide and water. Formation of carbon monoxide is negligible. After the gases leaving the absorber is as follows: 9.6%CO2, 3.0%O2 and 6.4% ethylene. Of the ethylene entering the reactor, what percentage is converted to ethylene oxide?arrow_forwardTVA’s Kingston Fossil Plant near Cedarville, Tennessee, burns about 14,000 tons of coal a day. A typical rail car is 53 ft long from coupling to coupling, weighs 30 tons empty and can carry 100 tons of coal. How many minutes would you have to wait at a crossing for this train to pass, each day, if it was pulled by four locomotives, each 70 ft long and moving at 10 miles per hour through town? Such delays cause decreased ability to provide effective emergency response times and interference with commuters and local freight delivery affecting the local economy.arrow_forwardThe amount of oil and grease from the rivers beside mining sites are quantified by solvent-solvent extraction. The dissolved oil and grease from the organic solvent will then be dried and weighed. What type of gravimetry is described by the statement? O precipitation gravimetry O physical gravimetry thermogravimetry electrogravimetryarrow_forward
- A chemical engineer must calculate the maximum safe operating temperature of a high-pressure gas reaction vessel. The vessel is a stainless- steel cylinder that measures 33.0 cm wide and 39.6 cm high. The maximum safe pressure inside the vessel has been measured to be 6.90 MPa. For a certain reaction the vessel may contain up to 4.70 kg of chlorine pentafluoride gas. Calculate the maximum safe operating temperature the engineer should recommend for this reaction. Write your answer in degrees Celsius. Be sure your answer has the correct number of significant digits. temperature: c 0.9 Xarrow_forwardThe quantity of 83.0 g of potassium iodide (KI) is added to water to give a solution with a mass of 150. g. Assume that this solution is very dilute and thus has the same heat capacity as water. The dissolution reaction and the associated heat for potassium iodide is KI (s) -→ K+(aq) + I-(aq) qrx = DHrx = +20.3 kJ. Calculate the change in the temperature of the solution.arrow_forwardA chemical engineer must calculate the maximum safe operating temperature of a high-pressure gas reaction vessel. The vessel is a stainless-steel cylinder that measures 23.0 cm wide and 27.6 cm high. The maximum safe pressure inside the vessel has been measured to be 4.50 MPa. For a certain reaction the vessel may contain up to 0.589 kg of boron trifluoride gas. Calculate the maximum safe operating temperature the engineer should recommend for this reaction. Write your answer in degrees Celsius. Be sure your answer has the correct number of significant digits. temperature: °C Ox10 ado ?arrow_forward
- C6H12O6(aq) + 6O2(g) 6CO2(g) + 6H2O(l)ΔH = –2802.7 kJ mol –1a) Write an expression for the equilibrium constant for this reaction.b) At equilibrium, the concentration of the reactants and products are determined as [CO2] = 0.30 M, [O2] = 0.040 M and [C6H12O6] = 0.065 M. Determine the value of the equilibrium constant (Kc) and predict the whether the products or reactants will be favoured at equilibrium.c) Given that the concentrations of the reactants and products at a particular time are [CO2] = 0.65 M, [O2] = 0.020 M and [C6H12O6] = 0.055 M, determine the reaction quotient (Qc). Compare the Kc and Qc values and predict the favoured direction of the reaction.d) Explain the effect on equilibrium of:i) Increasing temperatureii) Increasing pressureiii) Decreasing the concentration of oxygeniv) Increasing the concentration of carbon dioxidev) Adding a catalystarrow_forwardPlease help me with my he. I can't seem to solve this problem. I hope you can help me This is a guide to answer the activity. HESS LAW (Law of Heat Summation) “The enthalpy change of the overall process is the sum of the enthalpy changes of the individual component steps.” - It is the standard enthalpy of Reaction, ΔHᵒ. - It can be illustrated in the following example Consider the following reactions: (a) Fe(s) + O2(g) à FeO(s)ΔHᵒ = -272 kJ (b) 2 Fe(s) + O2 (g) à Fe2O3(s)ΔHᵒ = -82.5 kJ 1. Make sure to write the balance equation. 2. The final equation requires 2 moles of FeO which means we will multiply equation (a) and its enthalpy change by 2. 3. Reverse equation (a), which means, the product goes to the reactant side, (2) (FeO(s) à Fe(s) + O2(g))ΔHᵒ = (-272 kJ)(2) = 544 kJ 2 FeO(s) à 2 Fe(s) + O2(g))ΔHᵒ = -825.5 kJ 4. Combine the final equation (a) and the initial equation (b). And then cancel the same components with same number of particles. In this…arrow_forward(b) Heat is generated by the core of a nuclear reactor, this heat is removed by pumping cooling water through the core at a rate of 45 x 106 kg/h to generate steam. Cooling water, initially at 30 °C is continuously pumped through the reactor so that the outlet temperature is 748 °C and the core temperature remains constant. Calculate the rate of heat generated by the core of a nuclear reactor (in joules per second). (The specific heat capacity of ice 2100 Jkg 'K1; water = 4190 Jkg K; steam 2010 Jkg K. The latent heat of fusion of water is 334 kJ kg: latent heat of vaporisation of water 2264 kJ kg)arrow_forward
- When the oxide of generic metal M is heated at 25.0 C, a negligible amount of M is produced. MO,(s) = M(s) + 0,(g) AG = 291.0 mol When this reaction is coupled to the conversion of graphite to carbon dioxide, it becomes spontancous. What is the chemical equation of this coupled process? Show that the reaction is in equilibrium. Include physical states and represent graphite as C(s). chemical equation: What is the thermodynamic equilbrium constant for the coupled reaction? K =arrow_forwardThe following reaction is carried out under standard conditions (25°C and 1 bar): 2CH3OH(1) + 302(g) → 2CO2(g) + 4H₂O(1) AH₁° kJ mol-1 Sᵒ J K-¹ mol-1 C6H12O6 (1) -238.4 212.0 O₂ (g) 0.0 205.1 CO₂(g) -393.5 213.7 H₂O (1) -285.8 69.95 Using the data in the table above: a) Calculate the enthalpy of reaction, ArH, for the reaction: kJ mol-¹ b) Calculate the entropy change, AS, for the reaction: 1 c) Calculate AG for the reaction: kJ mol-¹ d) Is the reaction spontaneous? Put any calculations in here: J mol-1arrow_forwardAnaerobic bacteria use the glycolysis reaction,C6H12O6(aq) → 2 CH3CH(OH)COOH(aq). as a source of energy. In the reaction, glucose, C6H12O6, is first converted to pyruvic acid, CH3COCOOH, which is then converted to lactic acid, CH3CH(OH)COOH, through the action of the enzyme lactate dehydrogenase. Use bond enthalpies and mean bondenthalpies to estimate (a) the enthalpy of formation and (b) the enthalpy of combustion of glucose. Ignore the contributions of enthalpies of fusion and vaporization.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





