
Chemistry & Chemical Reactivity
10th Edition
ISBN: 9781337399074
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 39GQ
Phosphoserine is a less-common amino acid.
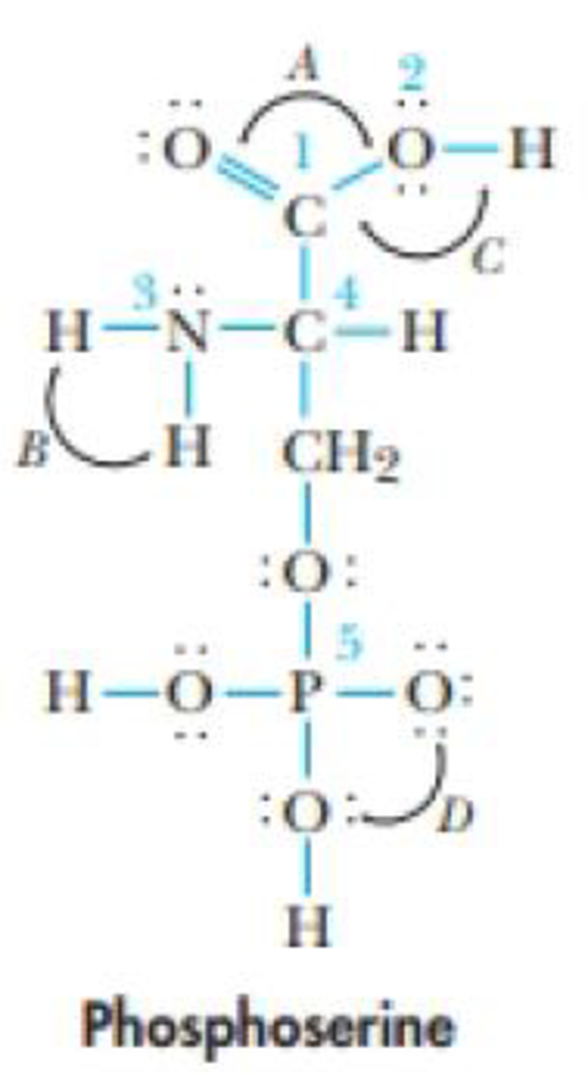
(a) Identify the hybridizations of atoms I through 5.
(b) What are the approximate values of the bond angles A, B. C, and D?
(c) What are the most polar bonds in the molecule?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Complete the electron-dot structure of caffeine, showing all lone-pair electrons, and identify the hybridization of the indicated atoms.
What is the hybridisation of Cl in ClO3-?
What is the hybridization of the central iodine atom in I3 -?
Chapter 9 Solutions
Chemistry & Chemical Reactivity
Ch. 9.1 - Use valence bond theory to describe the bonding in...Ch. 9.1 - Identify the hybridization of each underlined atom...Ch. 9.1 - Use valence bond theory to describe the bonding in...Ch. 9.2 - What is the electron configuration of the H2+ ion?...Ch. 9.2 - Could the anion Li2 exist? What is the ions bond...Ch. 9.2 - The cations O2+ and N2+ are formed when molecules...Ch. 9.3 - Photoelectron spectroscopy is s1milar to the...Ch. 9.3 - What is the energy of a photon with a wavelength...Ch. 9.3 - Using the accompanying figure, state which...Ch. 9.3 - The kinetic energy of an electron ejected from the...
Ch. 9.3 - The N2+ ions that are formed when electrons with...Ch. 9.3 - What is the empirical formula of Tynan purple?Ch. 9.3 - Butter yellow absorbs light with a wavelength of...Ch. 9.3 - Prob. 2.3ACPCh. 9 - Draw the Lewis structure for chloroform, CHCl3....Ch. 9 - Draw the Lewis structure for NF3. What are its...Ch. 9 - Draw the Lewis structure for hydroxylamine, H2NOH....Ch. 9 - Draw the Lewis structure for 1,...Ch. 9 - Draw the Lewis structure for carbonyl fluoride,...Ch. 9 - Draw the Lewis structure for acetamide, CH3CONH2....Ch. 9 - Specify the electron-pair and molecular geometry...Ch. 9 - Specify the electron-pair and molecular geometry...Ch. 9 - Prob. 9PSCh. 9 - What is the hybrid orbital set used by each of the...Ch. 9 - Draw the Lewis structures of the acid HPO2F2 and...Ch. 9 - Draw the Lewis structures of the arid HSO3F and...Ch. 9 - What is the hybridization of the carbon atom in...Ch. 9 - What is the hybridization of the carbon atoms in...Ch. 9 - What is the electron-pair and molecular geometry...Ch. 9 - What is the electron-pair and molecular geometry...Ch. 9 - Prob. 17PSCh. 9 - For each compound below, decide whether cis and...Ch. 9 - Molecular Orbital Theory (See Examples 9.49.6.)...Ch. 9 - Give the electron configurations for the ions Li2+...Ch. 9 - Calcium carbide, CaC2, contains the acetylide ion,...Ch. 9 - Platinum hexafluoride is an extremely strong...Ch. 9 - When sodium and oxygen react, one of the products...Ch. 9 - When potassium and oxygen react, one of the...Ch. 9 - Among the following, which has the shortest bond...Ch. 9 - Consider the following list of small molecules and...Ch. 9 - Prob. 27PSCh. 9 - The nitrosyl ion. NO+, has an interesting...Ch. 9 - These questions are not designated as to type or...Ch. 9 - What is the OSO angle and the hybrid orbital set...Ch. 9 - Sketch the resonance structures for the nitrite...Ch. 9 - Sketch the resonance structures for the nitrate...Ch. 9 - Sketch the resonance structures for the N2O...Ch. 9 - Compare the structure and bonding in CO2 and CO32...Ch. 9 - Numerous molecules are detected in deep space....Ch. 9 - Acrolein, a component of photochemical smog, has a...Ch. 9 - The organic compound below is a member of a class...Ch. 9 - The compound sketched below is acetylsalicylic...Ch. 9 - Phosphoserine is a less-common amino acid. (a)...Ch. 9 - Lactic acid is a natural compound found in sour...Ch. 9 - Cinnamaldehyde ocaus naturally in cinnamon oil....Ch. 9 - The ion Si2 was reported in a laboratory...Ch. 9 - The simple valence bond picture of O2 does not...Ch. 9 - Nitrogen, N2, can ionize to form N2+ or add an...Ch. 9 - Which of the homonuclear, diatomic molecules of...Ch. 9 - Which of the following molecules or ions are...Ch. 9 - Prob. 47GQCh. 9 - The structure of amphetamine, a stimulant, is...Ch. 9 - Menthol is used in soaps, perfumes, and foods. It...Ch. 9 - Prob. 50GQCh. 9 - Suppose you carry out the following reaction of...Ch. 9 - Ethylene oxide is an intermediate in the...Ch. 9 - The sulfamate ion, H2NSO3, can be thought of as...Ch. 9 - The compound whose structure is shown here is...Ch. 9 - Prob. 55ILCh. 9 - Carbon dioxide (CO2), dinitrogen monoxide (N2O),...Ch. 9 - Draw the two resonance structures that describe...Ch. 9 - Draw a Lewis structure for diimide, HNNH. Then,...Ch. 9 - Prob. 59SCQCh. 9 - Consider the three fluorides BF4, SiF4, and SF4....Ch. 9 - When two amino acids react with each other, they...Ch. 9 - What is the connection between bond order, bond...Ch. 9 - When is it desirable to use MO theory rather than...Ch. 9 - Show how valence bond theory and molecular orbital...Ch. 9 - Three of the four molecular orbitals for...Ch. 9 - Lets look more closely at the process of...Ch. 9 - Borax has the molecular formula Na2B4O5(OH)4. The...Ch. 9 - A model of the organic compound allene is shown...Ch. 9 - Prob. 69SCQCh. 9 - Prob. 70SCQCh. 9 - Bromine forms a number of oxides of varying...Ch. 9 - Prob. 72SCQCh. 9 - Urea reacts with malonic acid to produce...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Allene (1,2-propadiene), H2C=C=CH2, has two adjacent double bonds. What kind of hybridization must the central carbon have? Sketch the bonding π orbitals in allene.What shape do you predict for allene?arrow_forwardDraw the structure of CIF3 Calculate their hybridization and also determine the shapes and geometriesarrow_forwardWhat atomic or hybrid orbitals make up the bond between C and O in carbon dioxide, CO2 ?arrow_forward
- The compound 2,6-dimethylpyrazine (below) gives chocolate its odor and is used in flavorings. (a) Which atomic orbitals mix to form the hybrid orbitals of N? (b) In what type of hybrid orbital do the lone pairs of N reside? (c) Is C in CH3 hybridized the same as any C in the ring? Explain.arrow_forwardDescribe the bonding in the hydrogen cyanide molecule, HCN. Assume that N is sp-hybridized.arrow_forwardDraw the structure of PCL5 Calculate their hybridization and also determines the shapes and geometriesarrow_forward
- Acetylsalicylic acid, better known as aspirin, has the Lewisstructure (a) What are the approximate values of the bond angles labeled1, 2, and 3? (b) What hybrid orbitals are used about thecentral atom of each of these angles? (c) How many s bondsare in the molecule?arrow_forwardwhat is the geometrical shape of sp3d2 hybridization?arrow_forwardWhat is the hybridization of atom A in this compound?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY