
Concept explainers
(a)
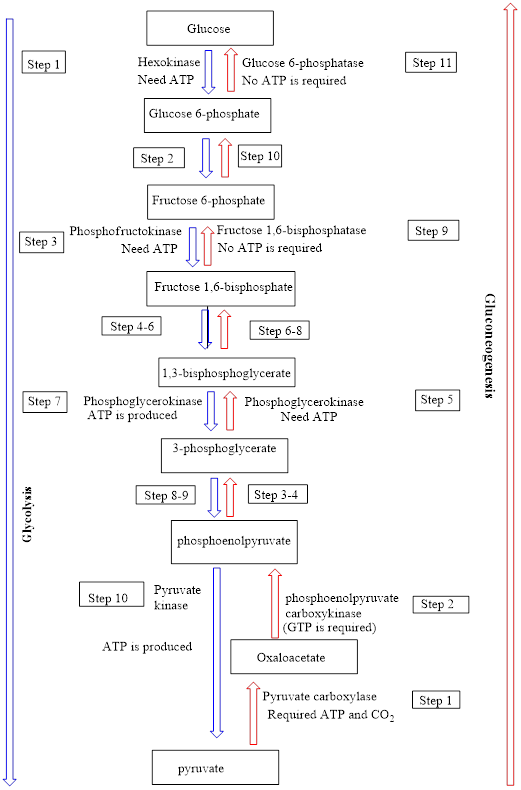
Interpretation: To identify the number of “common” steps in the gluconeogenesis and glycolysis processes in which the reactants/products are the reverse of each other and the enzyme is same.
Concept introduction: Glycolysis
In the gluconeogenesis pathway, glucose is produced from non-carbohydrate substances. The net overall equation for gluconeogenesis is as follows:
An overview of the pathways of glycolysis and gluconeogenesis is as follows:
(b)
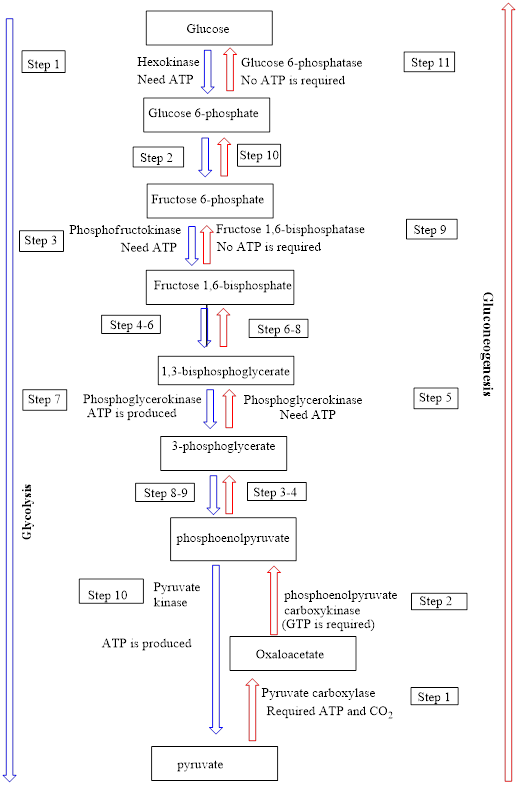
Interpretation: To identify the number of “common” steps in the gluconeogenesis and glycolysis processes in which the reactants/products are the reverse of each other and the enzyme is different.
Concept introduction: Glycolysis metabolic pathway is a ten-step enzyme catalyzed pathway in which a glucose molecule is converted to two pyruvate molecules. ATP and NADH-reduced coenzymes are produced along with pyruvate. The net overall equation for glycolysis is as follows:
In the gluconeogenesis pathway, glucose is produced from non-carbohydrate substances. The net overall equation for gluconeogenesis is as follows:
An overview of the pathways of glycolysis and gluconeogenesis is as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Glycogen synthase catalyzes a reaction that adds a glucosyl group (glucose monomer) to a glycogen molecule. In this reaction, one substrate (reactant) is glycogen, what is the other substrate (reactant)?arrow_forwardName the following enzymes:a. enzyme responsible for the cutting of the sugar on the nonreducing ends of glycogen branches b. enzyme that is only present in the liver and kidney during glycogen utilization c. enzyme that catalyzes the transfer of a two-carbon fragment from a ketose donor to an aldose acceptor in PPParrow_forwardAn experiment was performed to determine the effects of an inhibitor on the breakdown of glycogen by an enzyme. In an accompanying experiment, the inhibitor was added to the glycogen-enzyme suspension and reacted using the same experimental conditions. The data obtained from these experiments is tabulated below. Glycogen (mM) Product Formed Glycogen only (mM/min) Product Formed Glycogen and Inhibitor (mM/min) 0.5 22.6 15.9 1.0 32.1 24.9 1.5 38.1 28.2 2.0 41.2 35.1 2.5 44.7 40.0 3.0 48.5 43.3 1. How to draw Lineweaver-Burke Plots, and list step by steps of calculation 2. Determine the form of inhibition observed from these results, and explain your rationale for this form. It is uncompetitive inhibition because the Km decrease when ES complex bind to inhibitor 3. Determine the values for Km and Vmax from these results.arrow_forward
- Define the following as they apply to an enzymatic reaction. Give an example of each. a. Enzyme-limited: b. Substrate-limited:arrow_forwardIn the living cell, free energy made from one reaction can be used to drive another in an energetically unfavorable direction, provided the two reactions have a common intermediate (this is termed the principle of common intermediates). Example: In glycolysis, glucose is converted into pyruvate; in gluconeogenesis, pyruvate is converted into glucose. However, the actual AG for the formation of pyruvate from glucose is about -84 kJ/mol under typical cellular conditions. Most of the decrease in free energy in glycolysis takes place in three essentially irreversible steps catalyzed by, hexokinase, pyruvate kinase and phosphofructokinase. Using this example, (i) From gluconeogenesis, identify enzymes/steps that catalyze reactions that oppose/ bypass the irreversible steps of glycolysis (ii) Use one of the 3 opposing reactions (in glycolysis and gluconeogenesis) to demonstrate the PRINCIPLE OF COMMON INTERMEDIATESarrow_forwardSuggest a name for the enzyme that catalyzes each of the following reactions. a. transfer of a phosphate from ATP to glycerol b. addition of water to fumarate c. isomerization of glucose 6-phosphate to fructose 6-phosphatearrow_forward
- In glycogenolysis, in the reaction: glu-6-phosphate to glucose. What reaction is involved? Name the enzyme. Identify the two pathways where this enzyme is used.arrow_forwardIn the reaction: glu-1-phosphate to glu-6-phosphate, what reaction is involved? Name the enzyme. Identify the two pathways where this enzyme is used.arrow_forwarda. In the reaction: glycogen to glu-1-phosphate, what reaction is involved? Name the enzyme. b. In the reaction: glu-1-phosphate to glu-6-phosphate, what reaction is involved? Name the enzyme. Identify the two pathways where this enzyme is used.arrow_forward
- Below is a list of steps that occur after an exercising muscle cell begins degrading amino acids for ATP production. For each step, choose (A) if the process occurs in the muscle cell and (B) if the process occurs in the liver. It will be easiest to answer this question if you first put the steps in order, then decide in which organ they occur. ______ Alanine is transaminated with a-ketoglutarate.______ Branched chain amino acids are transaminated with a-ketoglutarate.______ Glutamate is deaminated.______ Glutamate is transaminated with pyruvate.______ Ketoacid skeletons resulting from branch chain amino acids enter the CAC.______ Pyruvate enters gluconeogenesis.______ Urea is formed from the nitrogen removed from the original branched chain amino acid.arrow_forwardGiven each set of information which may include common name(s) and the reaction catalyzed, you are required to identify the main class of the specific enzyme described. _____________________1. Name: alkaline phosphataseReaction: a phosphate monoester + H2O = an alcohol + phosphate_____________________2. Reaction: L-threonine = D-threonine.Other information: Inverts both chiral centers, a racemase. _____________________3. Name: glycine-N-acylaseReaction: acyl-COA + glycine = CoA + N-acylglycine_____________________ 4. Name: lysine decarboxylaseReaction: L-lysine = cadaverine + CO2 _____________________5. Name: methanol dehydrogenaseReaction: methanol + NAD+ = formaldehyde + NADH + H+ _____________________6. Name: citryl-CoA synthetaseReaction: ATP + citrate + CoA = ADP + phosphate + (3S)-citryl-CoA_____________________7. Name: D-xylulose reductaseReaction: xylitol + NAD+ = D-xylulose + NADH + H+ _____________________8. Name: cellobiose phosphorylaseReaction: cellobiose phosphate =…arrow_forwardIndicate whether the following statements are true or false:- a) The pentose phosphate pathway of glucose oxidation can lead to the release of energy. b) NAD + is a coenzyme of transketolase.arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





